Electronic and vibronic spectroscopy of crown ether water

Electronic and vibronic spectroscopy of crown ether water complexes: benzo-15 -crown-5 (B 15 C) and 4’-aminobenzo-15 -crown (ABC) B 15 C-(H 2 O)n ABC-(H 2 O)n V. Alvin Shubert and Timothy S. Zwier Purdue University, Department of Chemistry, West Lafayette, IN 47907
Motivations • Crown ethers long noted for ability to selectively bind substrates, especially cations • Much work has focused on structure and binding energy of crown-cation complex in solution • However, oxygen-rich pocket is ideally suited to binding other types of substrates, including water • We can study the binding of water to crown ethers in the absence of ions using jet-cooled gas phase spectroscopy • As a first step, we present the IR and UV spectra of water clusters of 4’-aminobenzo-15 -crown-5 and benzo-15 -crown-5 ethers
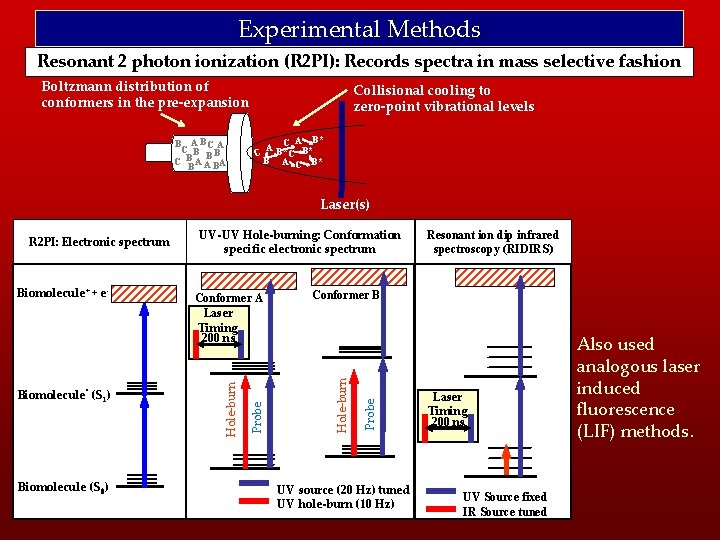
Experimental Methods Resonant 2 photon ionization (R 2 PI): Records spectra in mass selective fashion Boltzmann distribution of conformers in the pre-expansion B A BC A C B B B C B A A BA B Collisional cooling to zero-point vibrational levels A B*C B* C C B A C B* Laser(s) Biomolecule (S 0) Resonant ion dip infrared spectroscopy (RIDIRS) Probe Conformer B Hole-burn Biomolecule* (S 1) Conformer A Laser Timing 200 ns Probe Biomolecule+ + e- UV-UV Hole-burning: Conformation specific electronic spectrum Hole-burn R 2 PI: Electronic spectrum UV source (20 Hz) tuned UV hole-burn (10 Hz) Laser Timing 200 ns UV Source fixed IR Source tuned Also used analogous laser induced fluorescence (LIF) methods.

Computational Methods • Build water clusters from optimized monomers (see monomer presentation, FD 07) • Place water in position such that it can form two H-bonds to crown oxygens • Optimize with DFT B 3 LYP/6 -31+G(d), ultrafine grid and tight convergence options using the GAUSSIAN 03 suite of programs • For uniquely optimized structures, perform frequency calculations
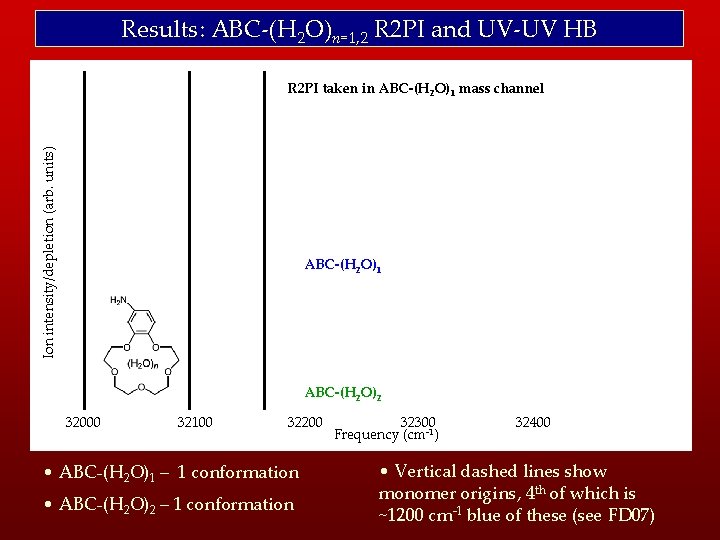
Results: ABC-(H 2 O)n=1, 2 R 2 PI and UV-UV HB Ion intensity/depletion (arb. units) R 2 PI taken in ABC-(H 2 O)1 mass channel ABC-(H 2 O)1 ABC-(H 2 O)2 32000 32100 32200 • ABC-(H 2 O)1 – 1 conformation • ABC-(H 2 O)2 – 1 conformation 32300 Frequency (cm-1) 32400 32500 • Vertical dashed lines show monomer origins, 4 th of which is ~1200 cm-1 blue of these (see FD 07)
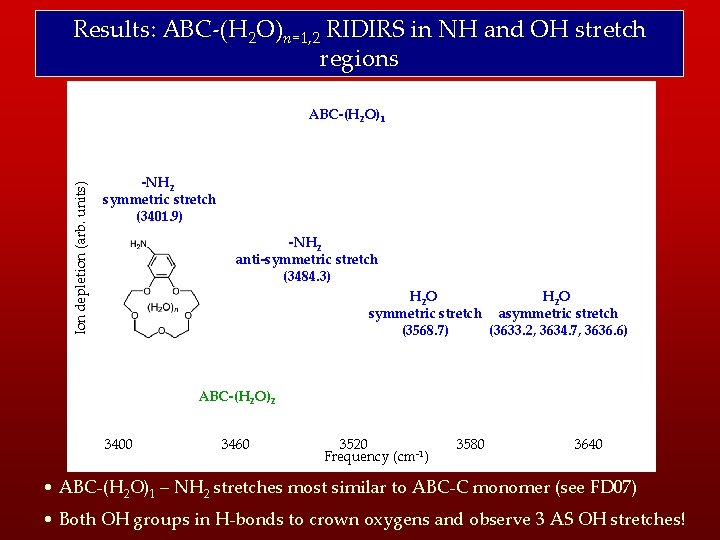
Results: ABC-(H 2 O)n=1, 2 RIDIRS in NH and OH stretch regions Ion depletion (arb. units) ABC-(H 2 O)1 -NH 2 symmetric stretch (3401. 9) -NH 2 anti-symmetric stretch (3484. 3) H 2 O symmetric stretch asymmetric stretch (3568. 7) (3633. 2, 3634. 7, 3636. 6) ABC-(H 2 O)2 3400 3460 3520 Frequency (cm-1) 3580 3640 • ABC-(H 2 O)1 – NH 2 stretches most similar to ABC-C monomer (see FD 07) • Both OH groups in H-bonds to crown oxygens and observe 3 AS OH stretches!
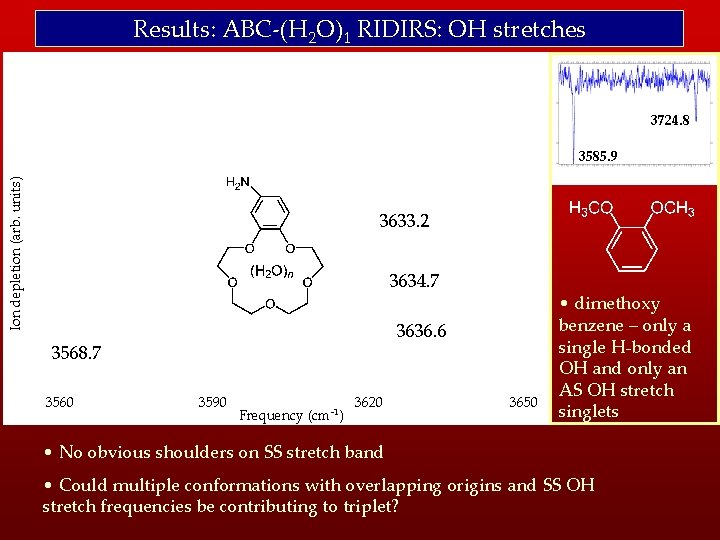
Results: ABC-(H 2 O)1 RIDIRS: OH stretches 3724. 8 Ion depletion (arb. units) 3585. 9 3633. 2 3634. 7 3636. 6 3568. 7 3560 3590 Frequency (cm-1) 3620 3650 • dimethoxy benzene – only a single H-bonded OH and only an AS OH stretch singlets • No obvious shoulders on SS stretch band • Could multiple conformations with overlapping origins and SS OH stretch frequencies be contributing to triplet?
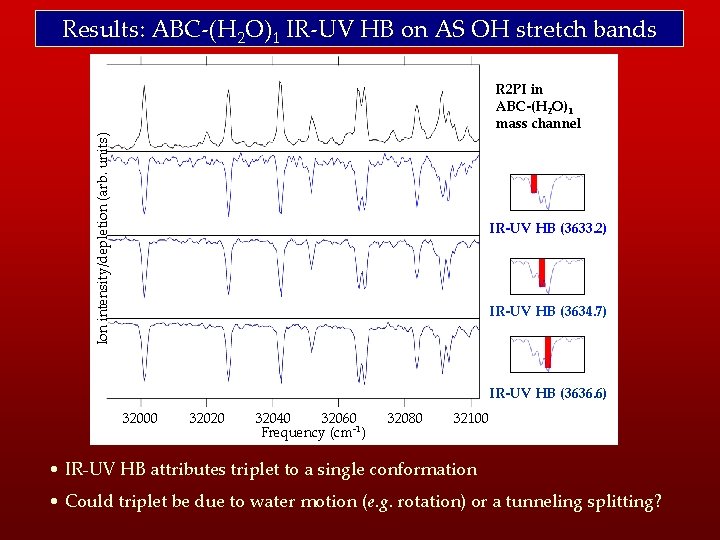
Results: ABC-(H 2 O)1 IR-UV HB on AS OH stretch bands Ion intensity/depletion (arb. units) R 2 PI in ABC-(H 2 O)1 mass channel IR-UV HB (3633. 2) IR-UV HB (3634. 7) IR-UV HB (3636. 6) 32000 32020 32040 32060 Frequency (cm-1) 32080 32100 • IR-UV HB attributes triplet to a single conformation • Could triplet be due to water motion (e. g. rotation) or a tunneling splitting?
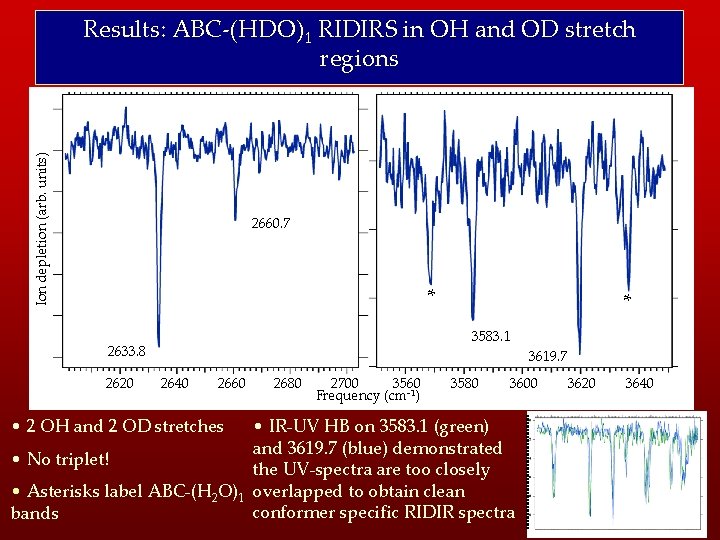
Ion depletion (arb. units) Results: ABC-(HDO)1 RIDIRS in OH and OD stretch regions 2660. 7 * 3583. 1 2633. 8 2620 * 3619. 7 2640 2660 2680 2700 3560 Frequency (cm-1) 3580 3600 • IR-UV HB on 3583. 1 (green) and 3619. 7 (blue) demonstrated • No triplet! the UV-spectra are too closely • Asterisks label ABC-(H 2 O)1 overlapped to obtain clean conformer specific RIDIR spectra bands • 2 OH and 2 OD stretches 3620 3640
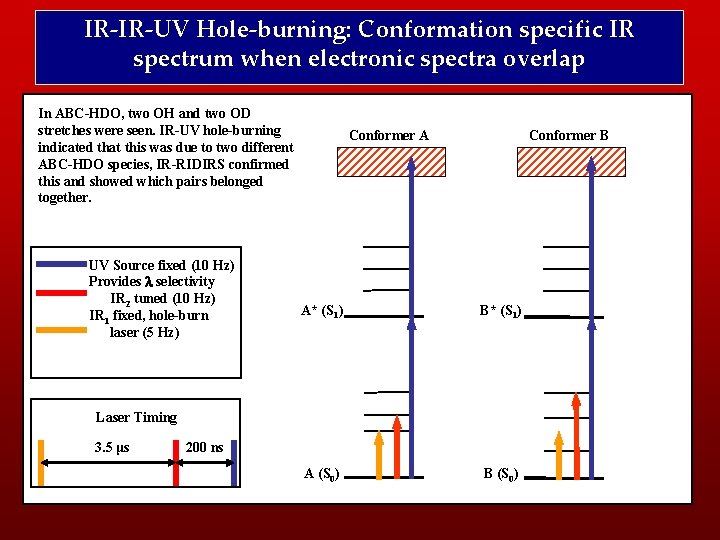
IR-IR-UV Hole-burning: Conformation specific IR spectrum when electronic spectra overlap In ABC-HDO, two OH and two OD stretches were seen. IR-UV hole-burning indicated that this was due to two different ABC-HDO species, IR-RIDIRS confirmed this and showed which pairs belonged together. UV Source fixed (10 Hz) Provides l selectivity IR 2 tuned (10 Hz) IR 1 fixed, hole-burn laser (5 Hz) Conformer A Conformer B A* (S 1) B* (S 1) A (S 0) B (S 0) Laser Timing 3. 5 μs 200 ns
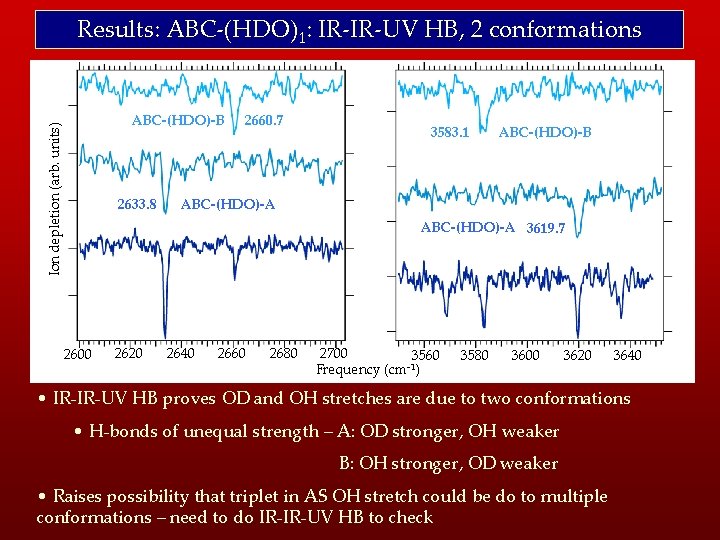
Results: ABC-(HDO)1: IR-IR-UV HB, 2 conformations Ion depletion (arb. units) ABC-(HDO)-B 2633. 8 2660. 7 3583. 1 ABC-(HDO)-B ABC-(HDO)-A 3619. 7 2600 2620 2640 2660 2680 2700 3560 -1 Frequency (cm ) 3580 3600 3620 3640 • IR-IR-UV HB proves OD and OH stretches are due to two conformations • H-bonds of unequal strength – A: OD stronger, OH weaker B: OH stronger, OD weaker • Raises possibility that triplet in AS OH stretch could be do to multiple conformations – need to do IR-IR-UV HB to check
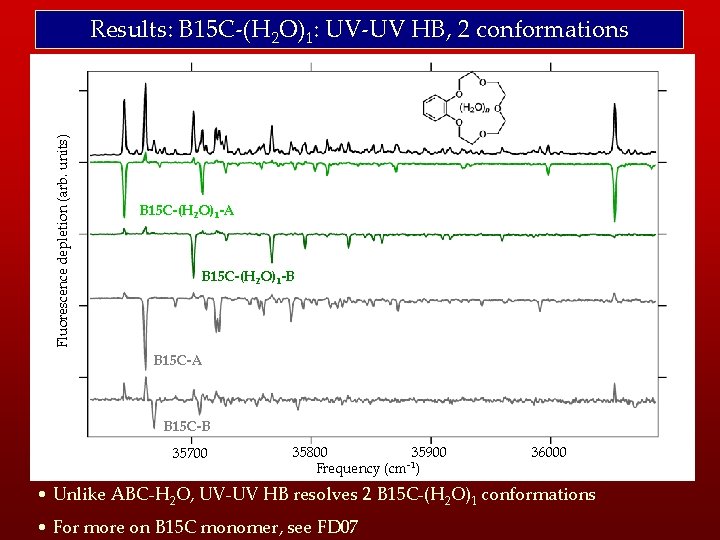
Fluorescence depletion (arb. units) Results: B 15 C-(H 2 O)1: UV-UV HB, 2 conformations B 15 C-(H 2 O)1 -A B 15 C-(H 2 O)1 -B B 15 C-A B 15 C-B 35700 35800 35900 -1 Frequency (cm ) 36000 • Unlike ABC-H 2 O, UV-UV HB resolves 2 B 15 C-(H 2 O)1 conformations • For more on B 15 C monomer, see FD 07
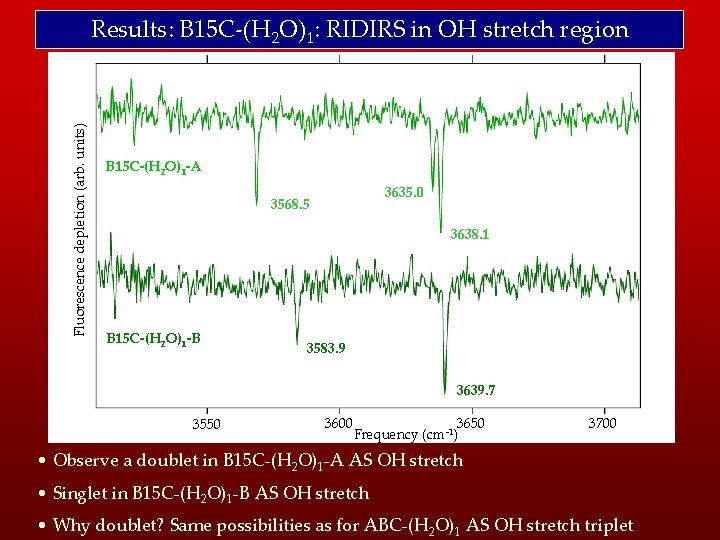
Fluorescence depletion (arb. units) Results: B 15 C-(H 2 O)1: RIDIRS in OH stretch region B 15 C-(H 2 O)1 -A 3635. 0 3568. 5 3638. 1 B 15 C-(H 2 O)1 -B 3583. 9 3639. 7 3550 3600 3650 Frequency (cm-1) 3700 • Observe a doublet in B 15 C-(H 2 O)1 -A AS OH stretch • Singlet in B 15 C-(H 2 O)1 -B AS OH stretch • Why doublet? Same possibilities as for ABC-(H 2 O)1 AS OH stretch triplet
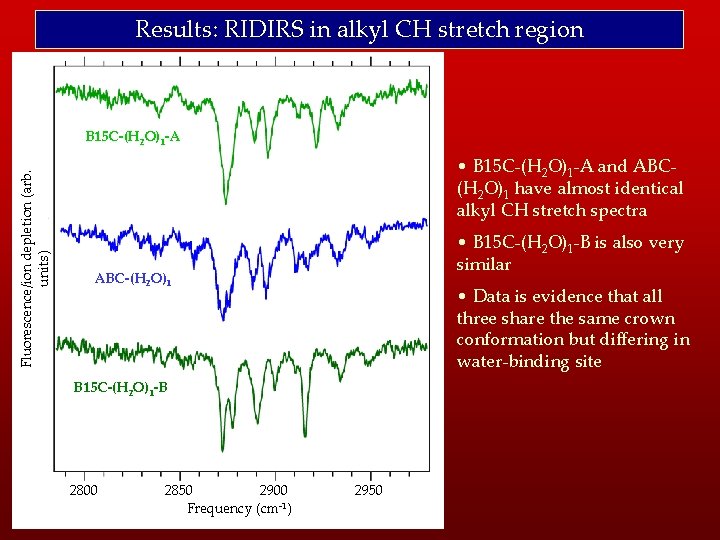
Results: RIDIRS in alkyl CH stretch region Fluorescence/ion depletion (arb. units) B 15 C-(H 2 O)1 -A • B 15 C-(H 2 O)1 -A and ABC(H 2 O)1 have almost identical alkyl CH stretch spectra • B 15 C-(H 2 O)1 -B is also very similar ABC-(H 2 O)1 • Data is evidence that all three share the same crown conformation but differing in water-binding site B 15 C-(H 2 O)1 -B 2800 2850 2900 Frequency (cm-1) 2950
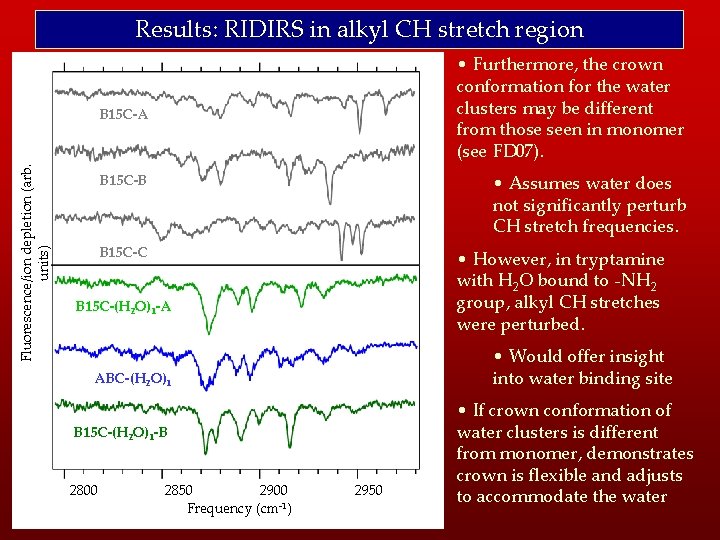
Results: RIDIRS in alkyl CH stretch region • Furthermore, the crown conformation for the water clusters may be different from those seen in monomer (see FD 07). Fluorescence/ion depletion (arb. units) B 15 C-A • Assumes water does not significantly perturb CH stretch frequencies. B 15 C-B B 15 C-C • However, in tryptamine with H 2 O bound to -NH 2 group, alkyl CH stretches were perturbed. B 15 C-(H 2 O)1 -A • Would offer insight into water binding site ABC-(H 2 O)1 B 15 C-(H 2 O)1 -B 2800 2850 2900 Frequency (cm-1) 2950 • If crown conformation of water clusters is different from monomer, demonstrates crown is flexible and adjusts to accommodate the water
Conclusions • ABC-(H 2 O)1 : 1 conformer; B 15 C-(H 2 O)1: 2 conformers • Crown conformation same in all three water clusters (alkyl CH stretch) • Triplet/doublet observed in AS OH stretch of ABC-(H 2 O)1/B 15 C-(H 2 O)1 • In ABC-(H 2 O)1, disappears upon substitution with HDO • Two conformers associated with two different strength H-bonds • Tunneling splitting? – observed intensity ratio is reverse of that predicted by spin statistics • Water motion, rotation? • Multiple overlapped conformations? Future Work • Perform IR-IR-UV HB on AS OH stretch peaks (for both ABC and B 15 C-A) • Measure water binding energies • Study higher order water clusters

Acknowledgements • Zwier group: Prof. Timothy S. Zwier Jasper R. Clarkson Esteban E. Baquero Tracy Legreve Nathan Pillsbury Josh Newby William H. James III Chirantha Rodrigo Ching Ping Liu Christian Müller Josh Sebree • Funding: National Science Foundation • Computing Resources Information and Technology at Purdue (ITa. P), Rosenbaum Computing Center (RCC)
- Slides: 17