Electronegativity Electronegativity Ability of an atom to attract

Electronegativity
Electronegativity Ability of an atom to attract electrons towards itself in a chemical bond
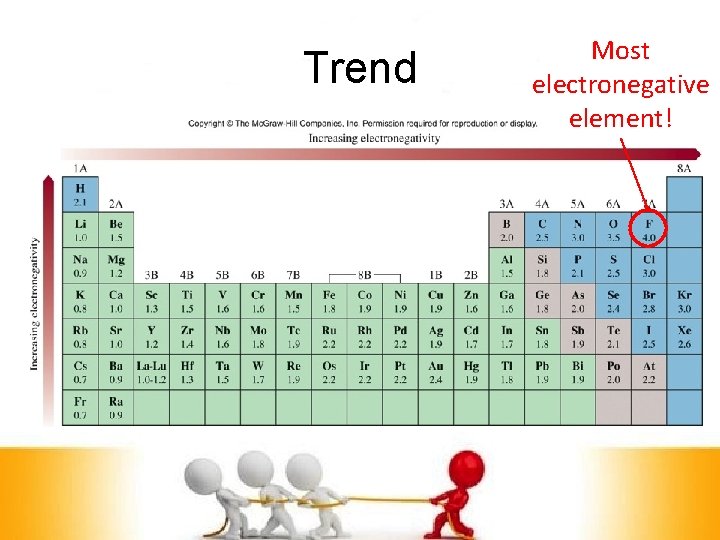
Trend Most electronegative element!

F > O > Cl, N > Br > C, S, I > H, P • • • Frank Owes Claire Nine Brownies • • • Cause She Is a Hungry Person v Put a comma in every third element v These elements separated by commas are equal in electronegativity v Memorize these!!!
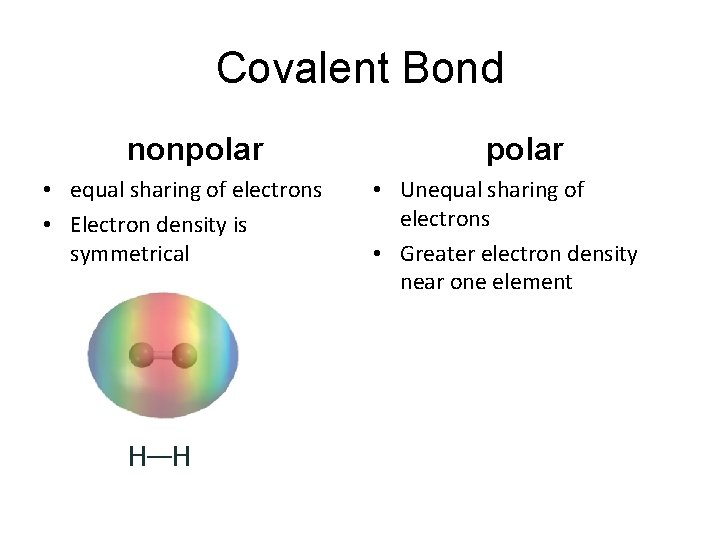
Covalent Bond nonpolar • equal sharing of electrons • Electron density is symmetrical H—H polar • Unequal sharing of electrons • Greater electron density near one element
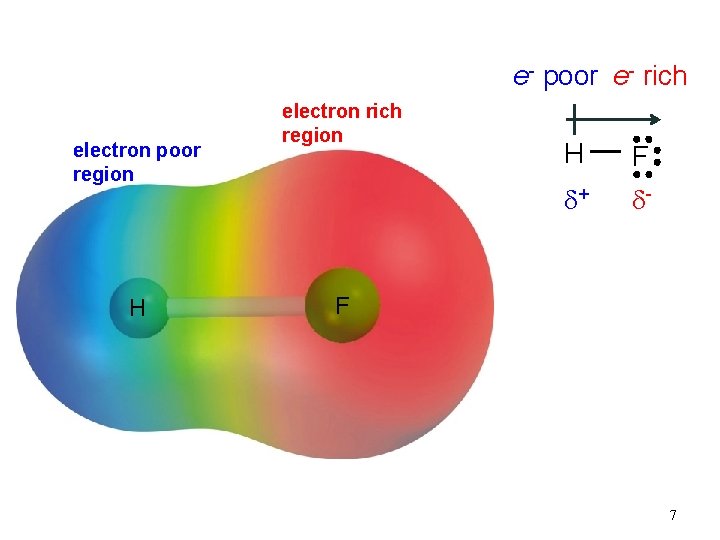
e- poor e- rich electron poor region H electron rich region H F d+ d- F 7
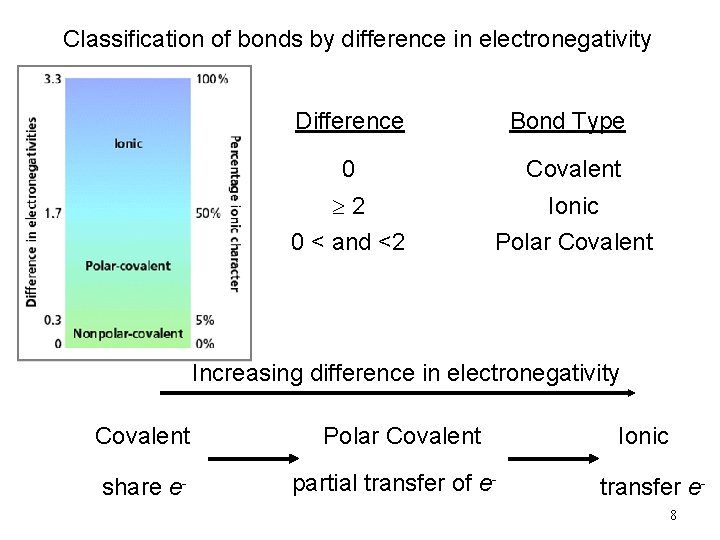
Classification of bonds by difference in electronegativity Difference Bond Type 0 Covalent 2 0 < and <2 Ionic Polar Covalent Increasing difference in electronegativity Covalent share e- Polar Covalent partial transfer of e- Ionic transfer e 8
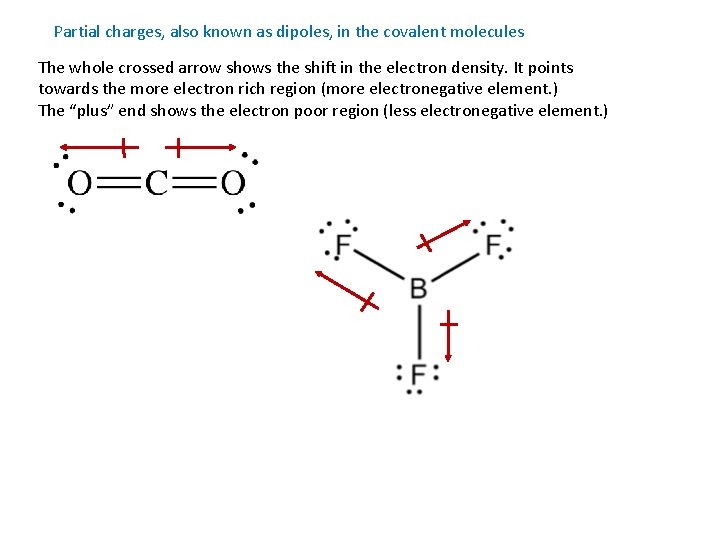
Partial charges, also known as dipoles, in the covalent molecules The whole crossed arrow shows the shift in the electron density. It points towards the more electron rich region (more electronegative element. ) The “plus” end shows the electron poor region (less electronegative element. )
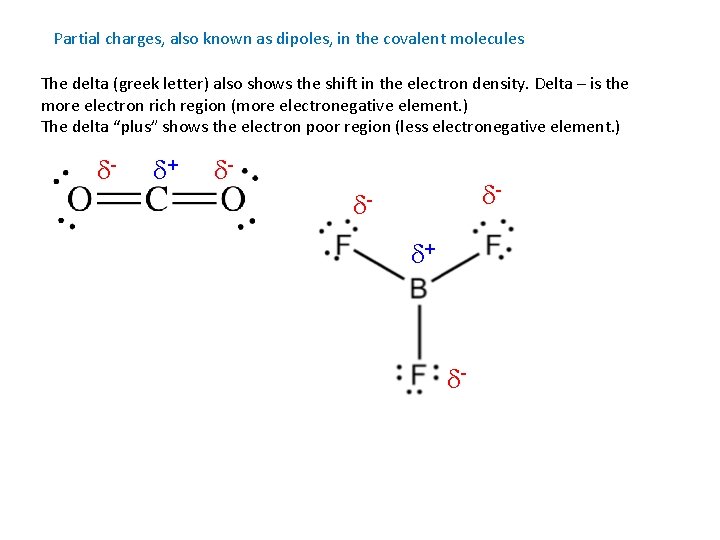
Partial charges, also known as dipoles, in the covalent molecules The delta (greek letter) also shows the shift in the electron density. Delta – is the more electron rich region (more electronegative element. ) The delta “plus” shows the electron poor region (less electronegative element. ) d- d+ d- d- dd+ d-
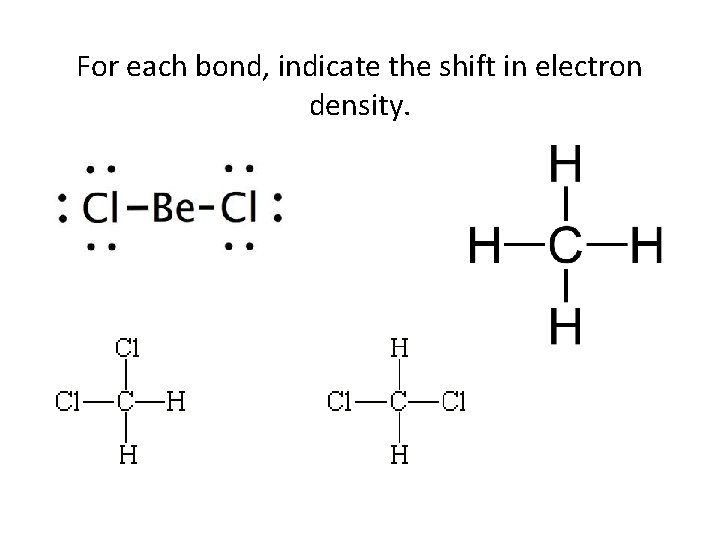
For each bond, indicate the shift in electron density.

Practice Which of the following bonds is polar covalent, which is non polar covalent, and which is ionic? Ionic a. The bond in Cs. Cl Polar covalent b. The bond in H 2 S c. The NN bond in H 2 NNH 2 Non Polar covalent
- Slides: 12