Electronegativity and Polarity Electronegativity The ability of an

Electronegativity and Polarity
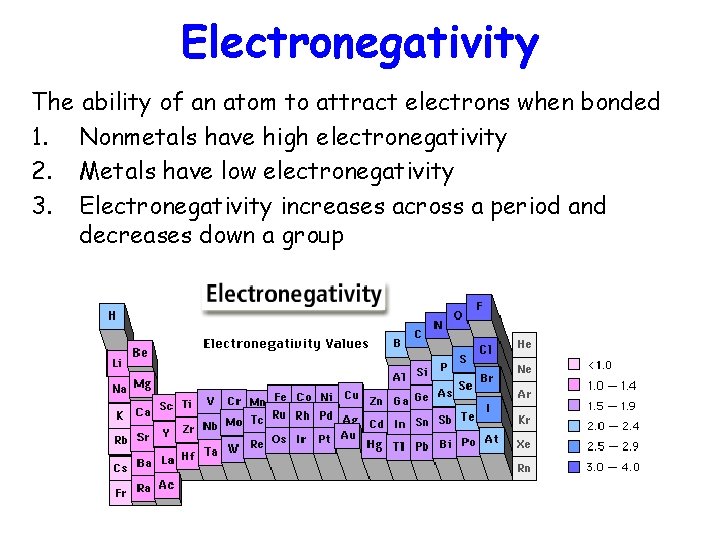
Electronegativity The ability of an atom to attract electrons when bonded 1. Nonmetals have high electronegativity 2. Metals have low electronegativity 3. Electronegativity increases across a period and decreases down a group

Nonpolar Covalent Bond • When electrons are shared between 2 atoms, a covalent bond is formed. • If the atoms are identical, e. g. Cl 2, the electrons are shared equally (nonpolar)
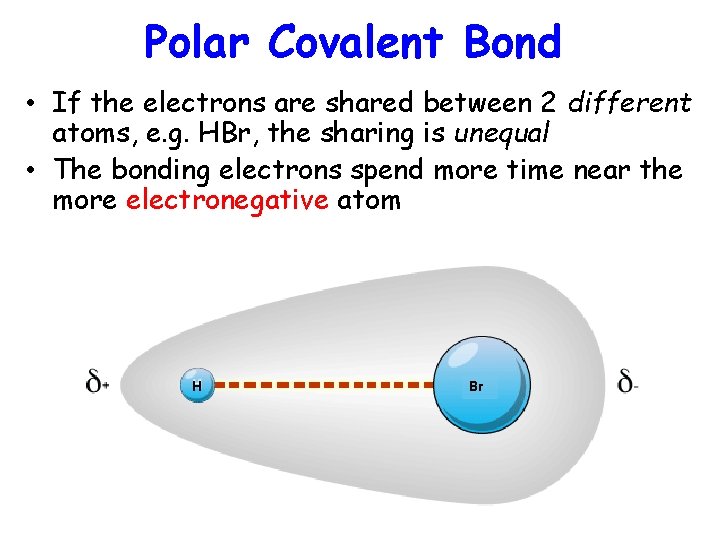
Polar Covalent Bond • If the electrons are shared between 2 different atoms, e. g. HBr, the sharing is unequal • The bonding electrons spend more time near the more electronegative atom H Br
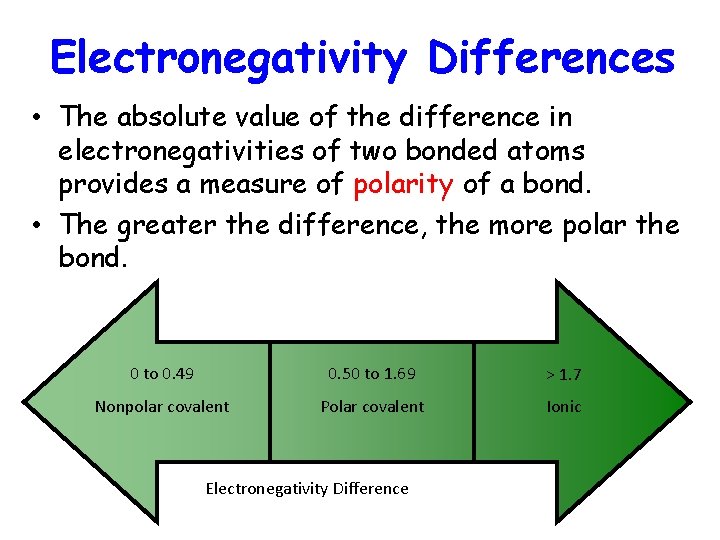
Electronegativity Differences • The absolute value of the difference in electronegativities of two bonded atoms provides a measure of polarity of a bond. • The greater the difference, the more polar the bond. 0 to 0. 49 0. 50 to 1. 69 > 1. 7 Nonpolar covalent Polar covalent Ionic Electronegativity Difference
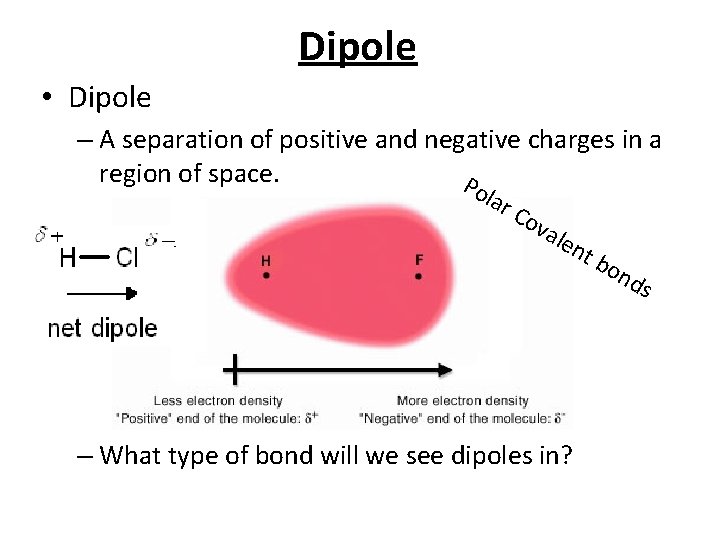
Dipole • Dipole – A separation of positive and negative charges in a region of space. P ola r. C ova len – What type of bond will we see dipoles in? tb on ds

Practice • Using the periodic table at the back of your text, determine whether the following molecules are non-polar covalent, or ionic bonds. • • HCl Na. Cl CCl 4 NH 3
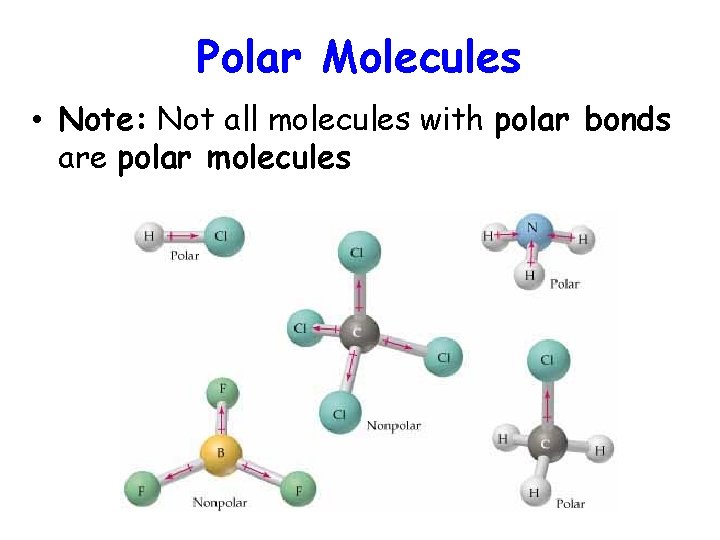
Polar Molecules • Note: Not all molecules with polar bonds are polar molecules
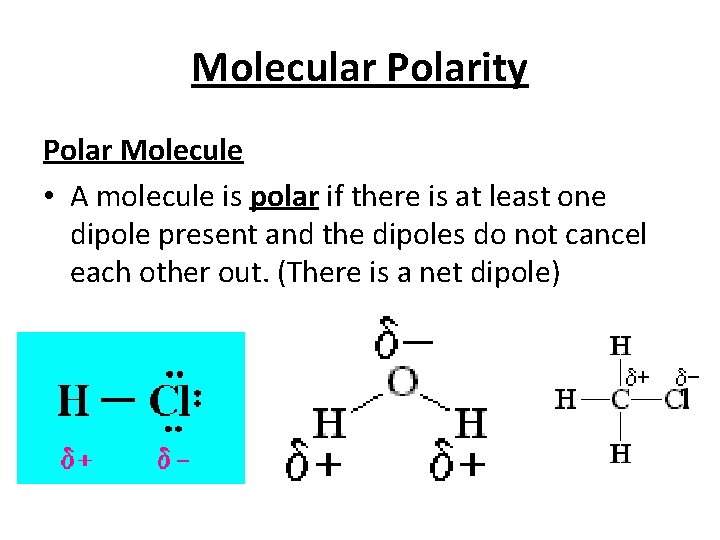
Molecular Polarity Polar Molecule • A molecule is polar if there is at least one dipole present and the dipoles do not cancel each other out. (There is a net dipole)

Molecular Polarity • Non- Polar Molecule – A molecule that has ONLY non-polar bonds OR - A bond dipole sum of zero.

Molecular Forces in. TRAmolecular Forces – Ionic bonds, covalent bonds, metallic bonding are all examples of bonds within a molecule in. TERmolecular Forces • Forces between different molecules • Weaker than in. TRAmolecular forces, but still very important
- Slides: 11