Electronegativity and Bonding Electronegativity and Bonding Electronegativity EN
Electronegativity and Bonding

Electronegativity and Bonding • Electronegativity (EN) is a measure of an atom’s ability to attract electrons away from other atoms. • Atoms with high electronegativity values (EN’s) are the best electron attractors and atoms with low electronegativity values (EN’s) are the worst electron attractors. • The electronegativity scale is a relative (arbitrary) scale based on the element fluorine, because fluorine is the best of the best electron attractors

Electronegativity and Bonding • NOTICE: Most noble gases do not have EN values assigned because they do not attract electrons. The reason will be explained later.

Electronegativity and Bonding Ex. (1) As atomic number increases in group-1, the EN _____________. decreases Ex. (2) As atomic number increases in period-3, the EN _____________. increases Ex. (3) Which of the following atoms would be the worst electron attractors? (1) Li (2) Ba (3) S (4) Sn

Electronegativity and Bonding • Electronegativity difference (ΔEN) can be used to predict the type of bonding present in a compound. Nonpolar Covalent Bonding 0 Polar Covalent Bonding 1. 7 Ionic Bonding ∆EN • Using the Periodic Table to predict bonding type is a more reliable method.

Electronegativity and Bonding • Electronegativity difference (ΔEN) indicates the degree of polarity in a chemical bond. • The larger the electronegativity difference (ΔEN) is between two bonded elements, the greater the bond polarity will be. ØH 2 O has a more polar bond than CO 2. ΔEN H 2 O = 1. 4 ΔEN CO 2 = 0. 9
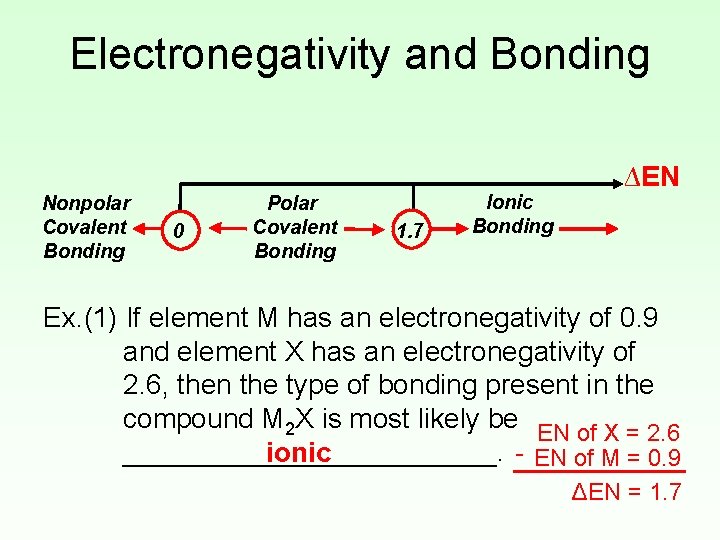
Electronegativity and Bonding Nonpolar Covalent Bonding 0 Polar Covalent Bonding 1. 7 Ionic Bonding ∆EN Ex. (1) If element M has an electronegativity of 0. 9 and element X has an electronegativity of 2. 6, then the type of bonding present in the compound M 2 X is most likely be EN of X = 2. 6 - EN of M = 0. 9 ____________. ionic ΔEN = 1. 7
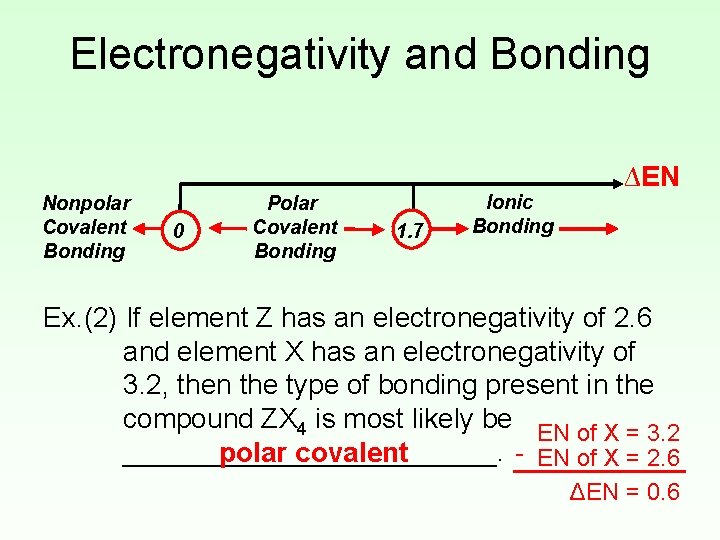
Electronegativity and Bonding Nonpolar Covalent Bonding 0 Polar Covalent Bonding 1. 7 Ionic Bonding ∆EN Ex. (2) If element Z has an electronegativity of 2. 6 and element X has an electronegativity of 3. 2, then the type of bonding present in the compound ZX 4 is most likely be EN of X = 3. 2 - EN of X = 2. 6 ____________. polar covalent ΔEN = 0. 6
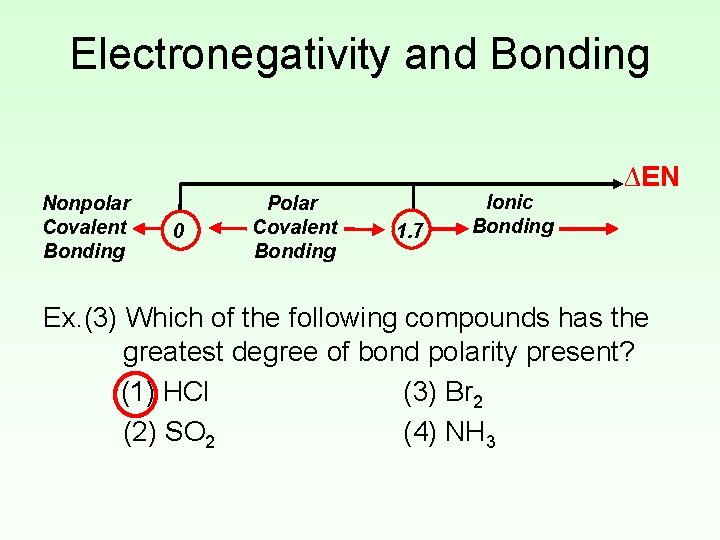
Electronegativity and Bonding Nonpolar Covalent Bonding 0 Polar Covalent Bonding 1. 7 Ionic Bonding ∆EN Ex. (3) Which of the following compounds has the greatest degree of bond polarity present? (1) HCl (3) Br 2 (2) SO 2 (4) NH 3
- Slides: 9