Electron Transport Oxidative Phosphorylation Dr Kevin Ahern Biological

Electron Transport & Oxidative Phosphorylation Dr. Kevin Ahern
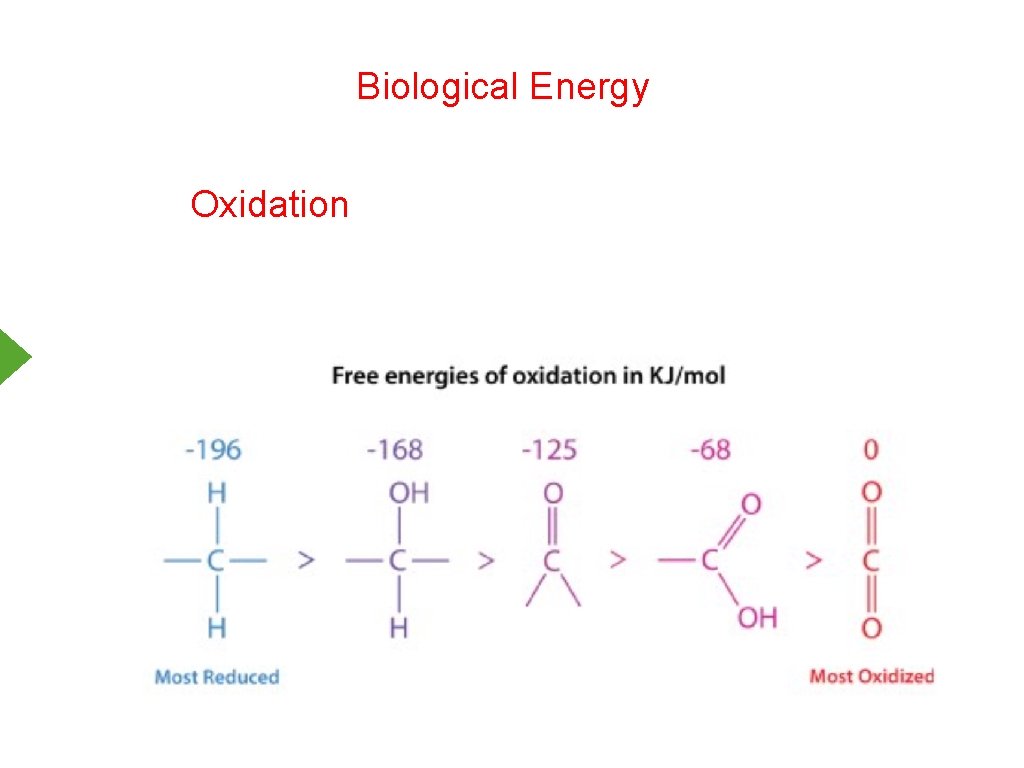
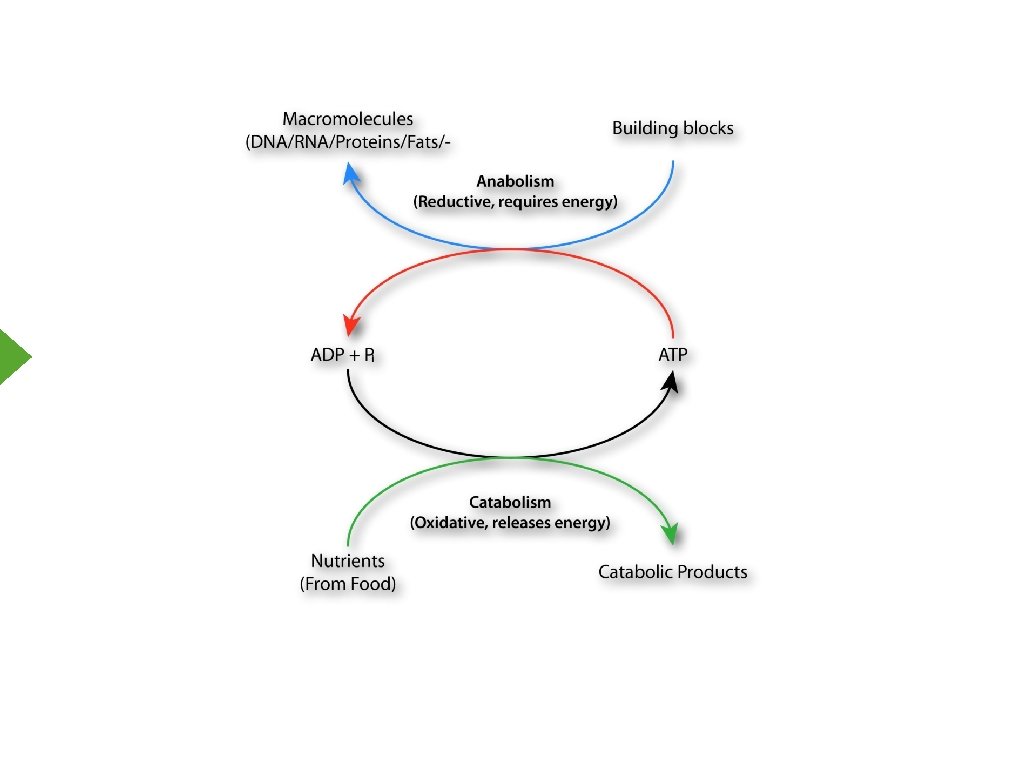
Biological Energy Oxidation
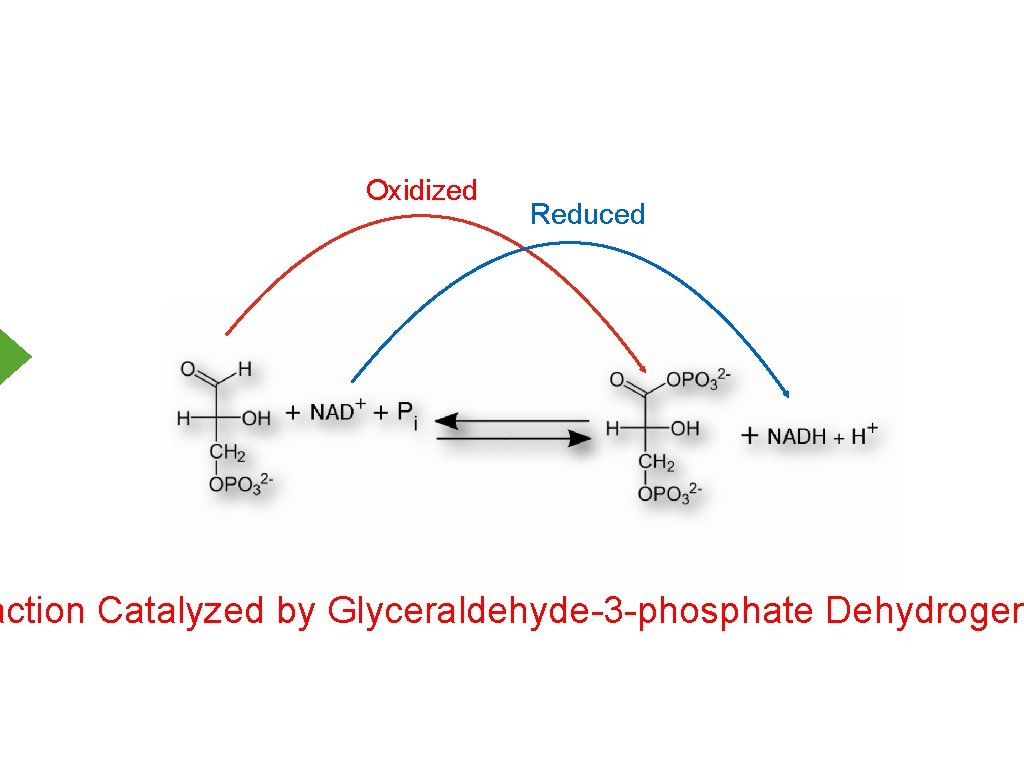
Oxidized Reduced action Catalyzed by Glyceraldehyde-3 -phosphate Dehydrogen
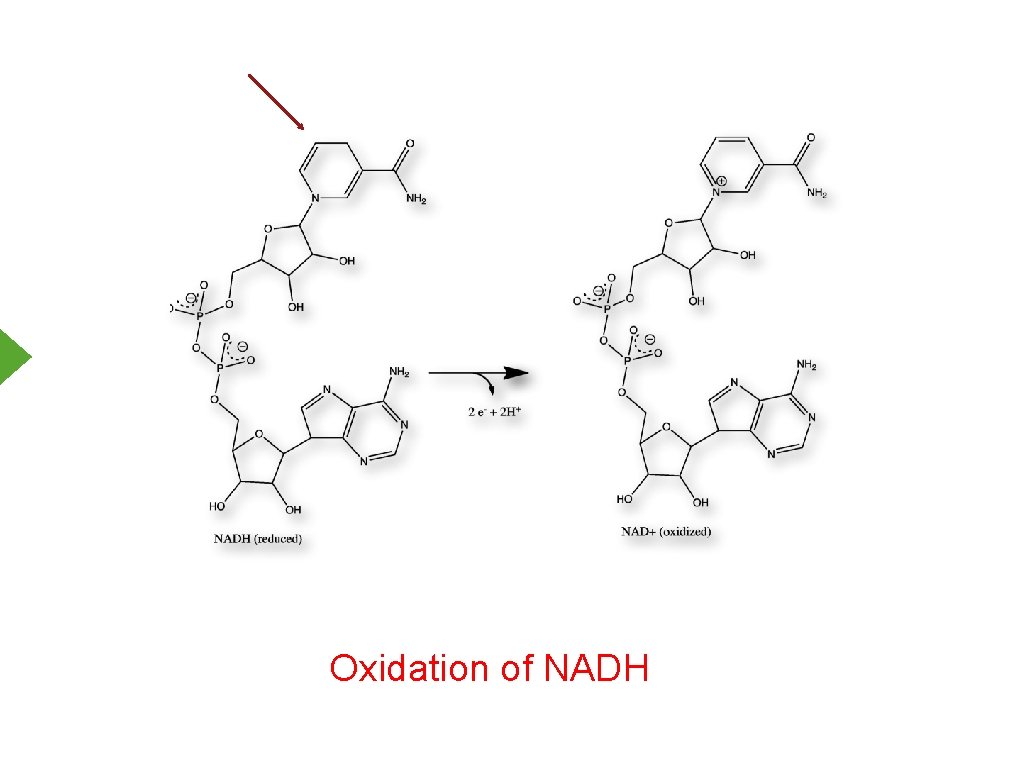
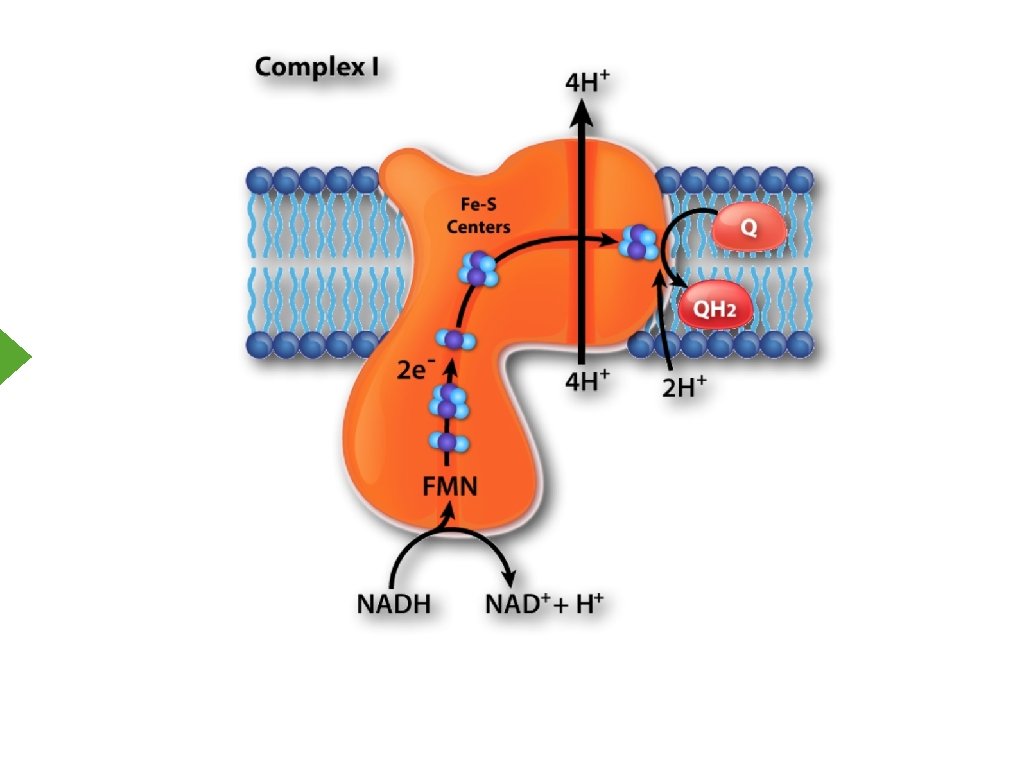
Oxidation of NADH
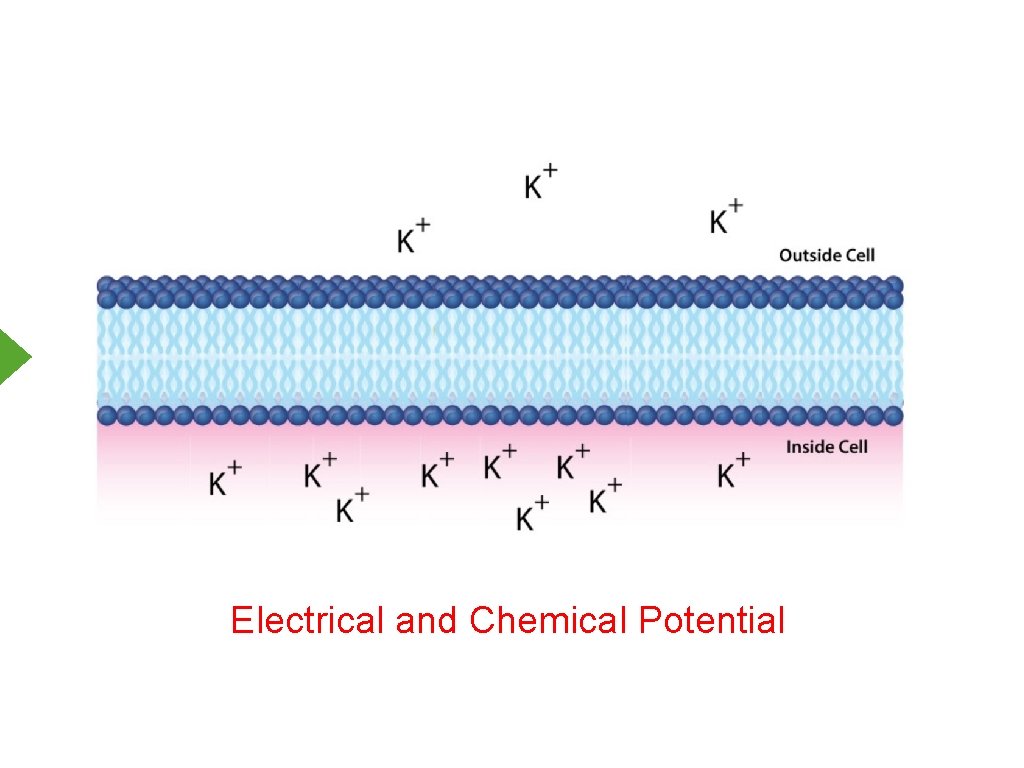
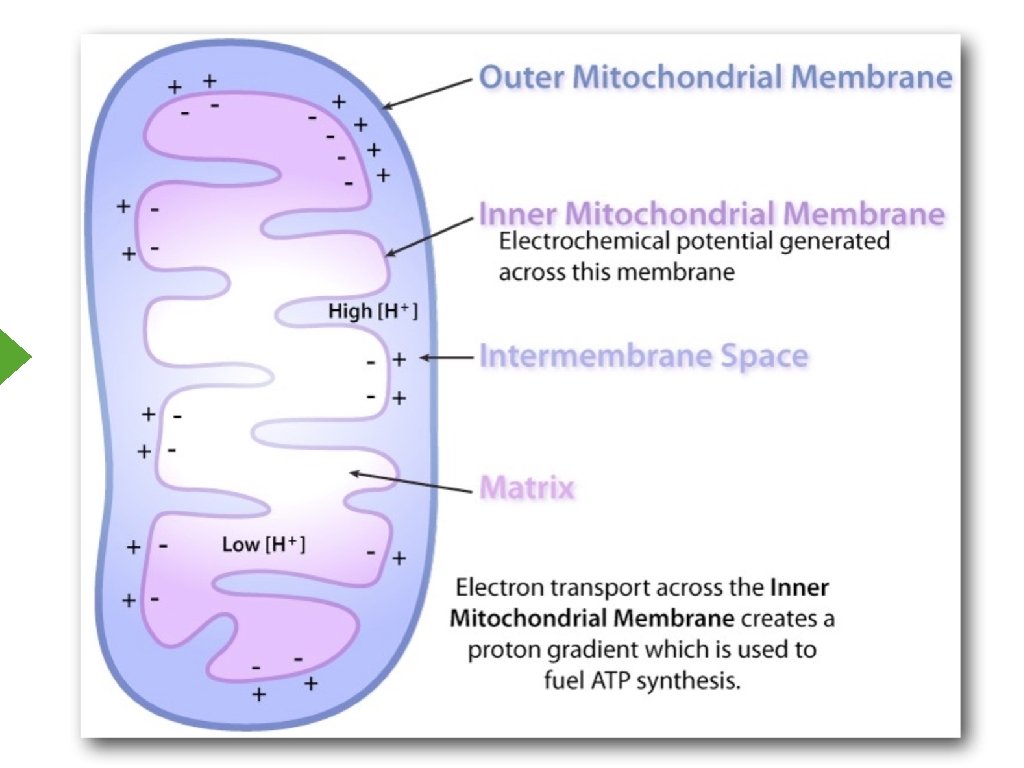
Electrical and Chemical Potential
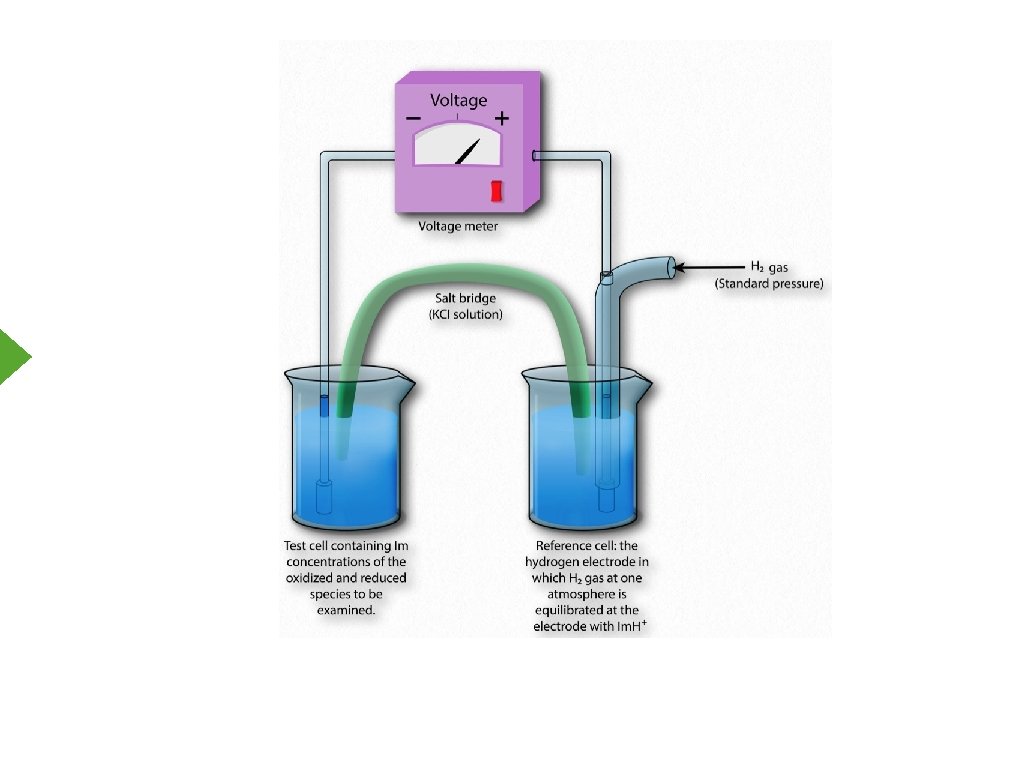
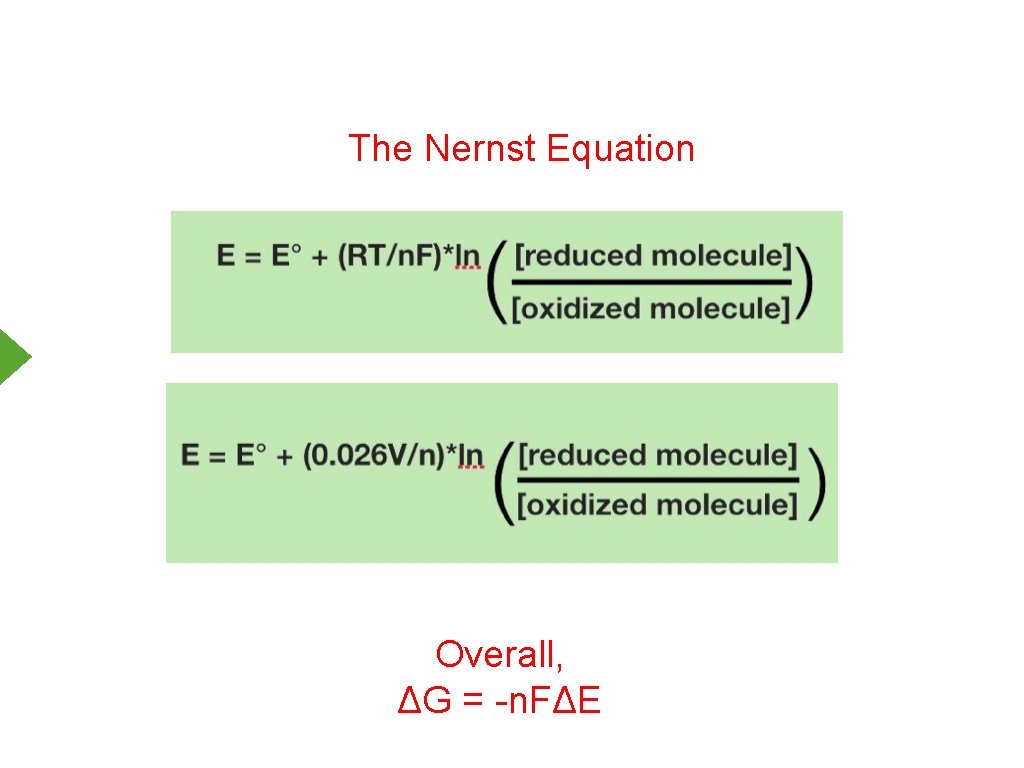
The Nernst Equation Overall, ΔG = -n. FΔE
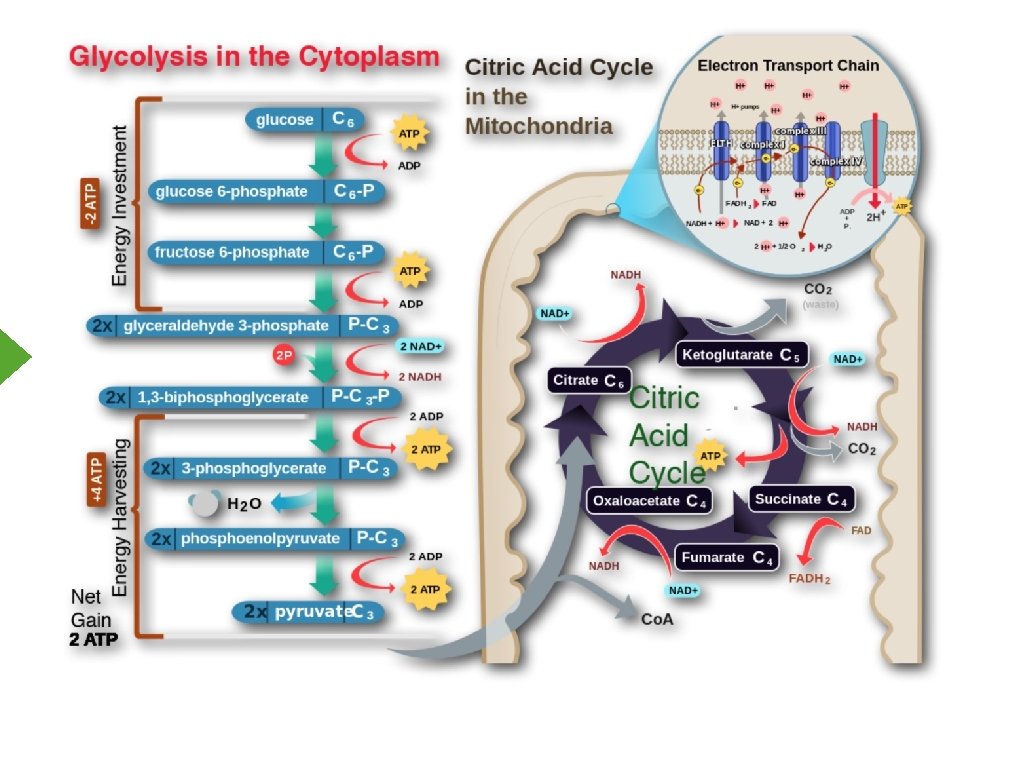
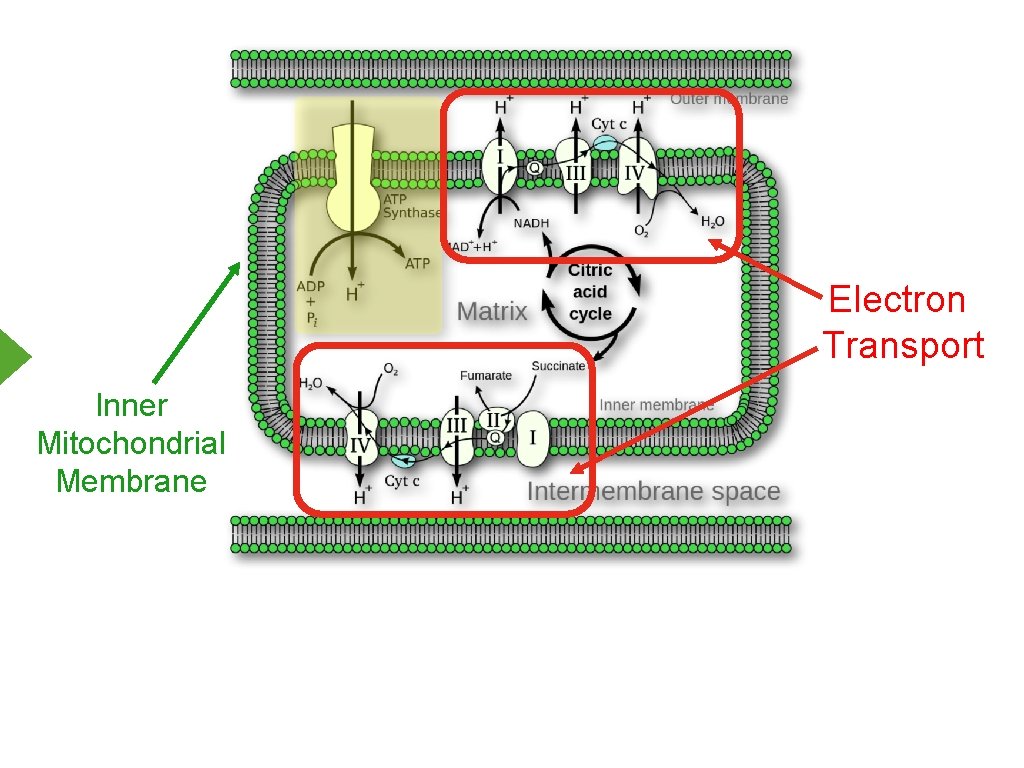
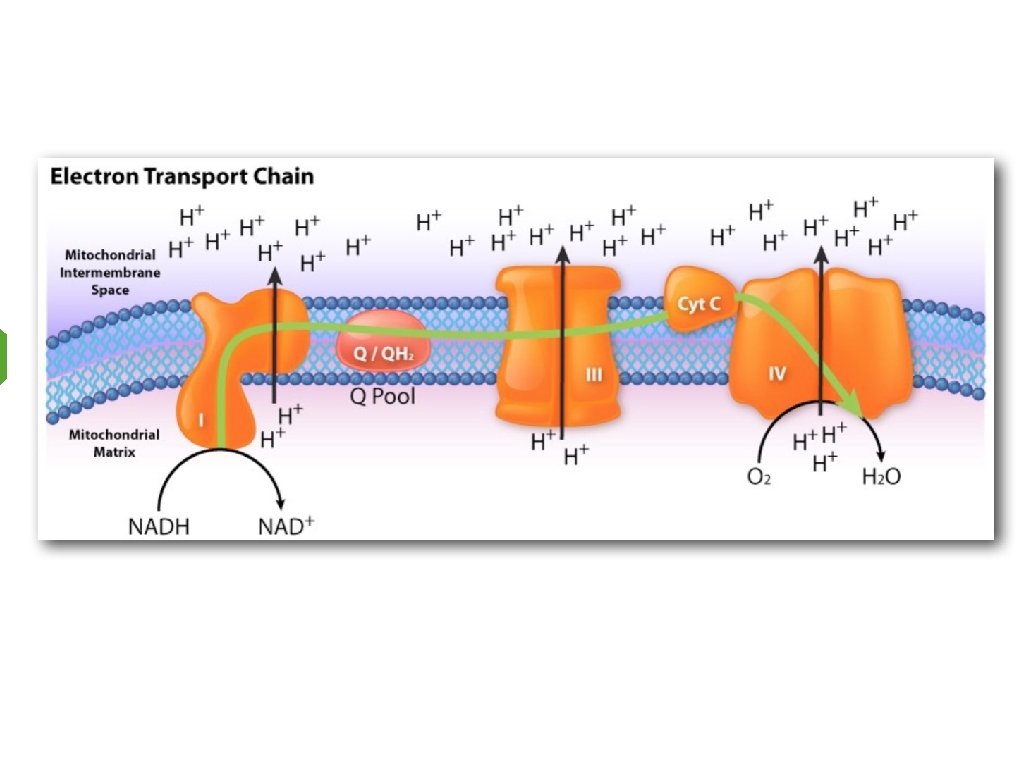
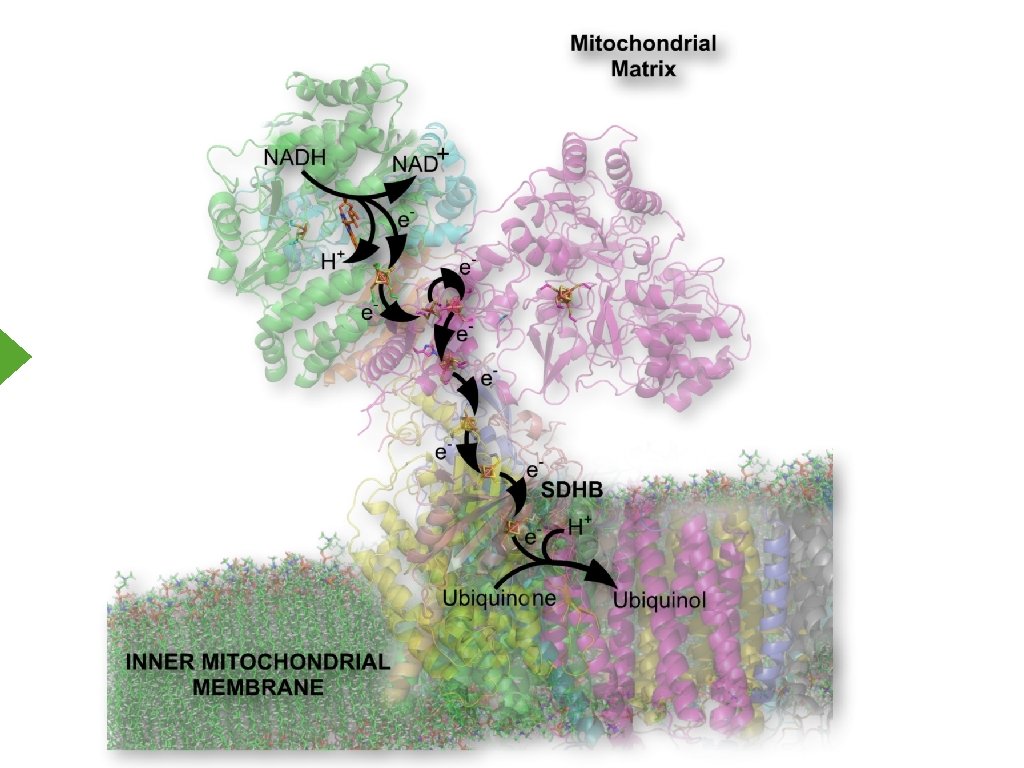
Electron Transport Inner Mitochondrial Membrane
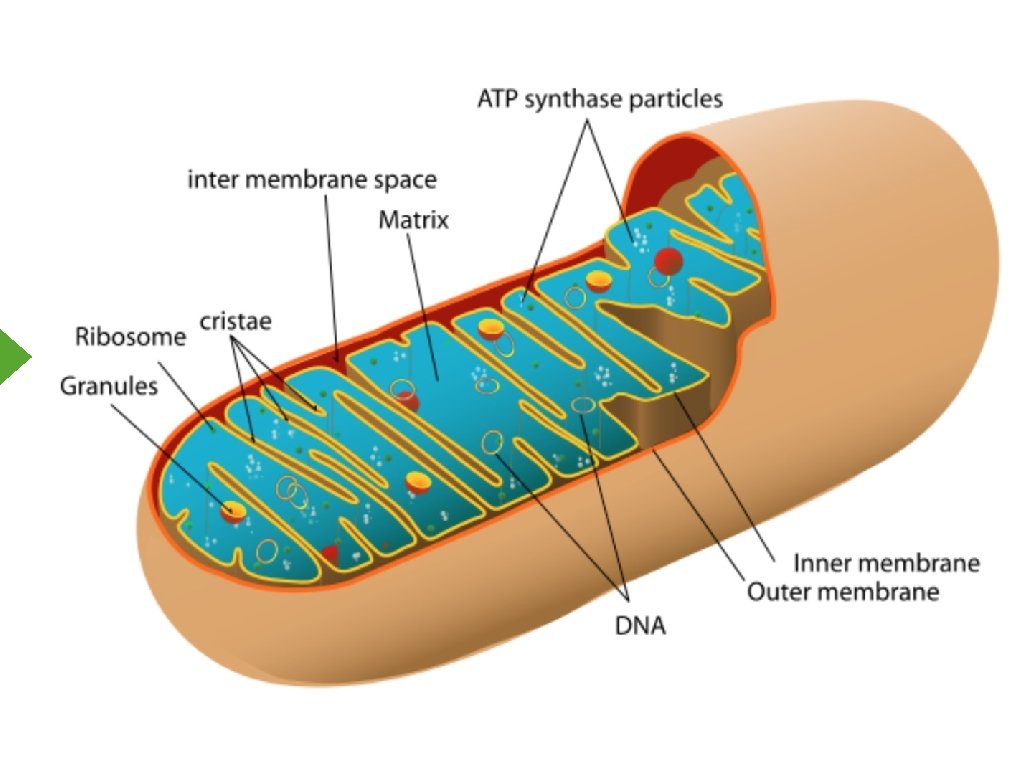
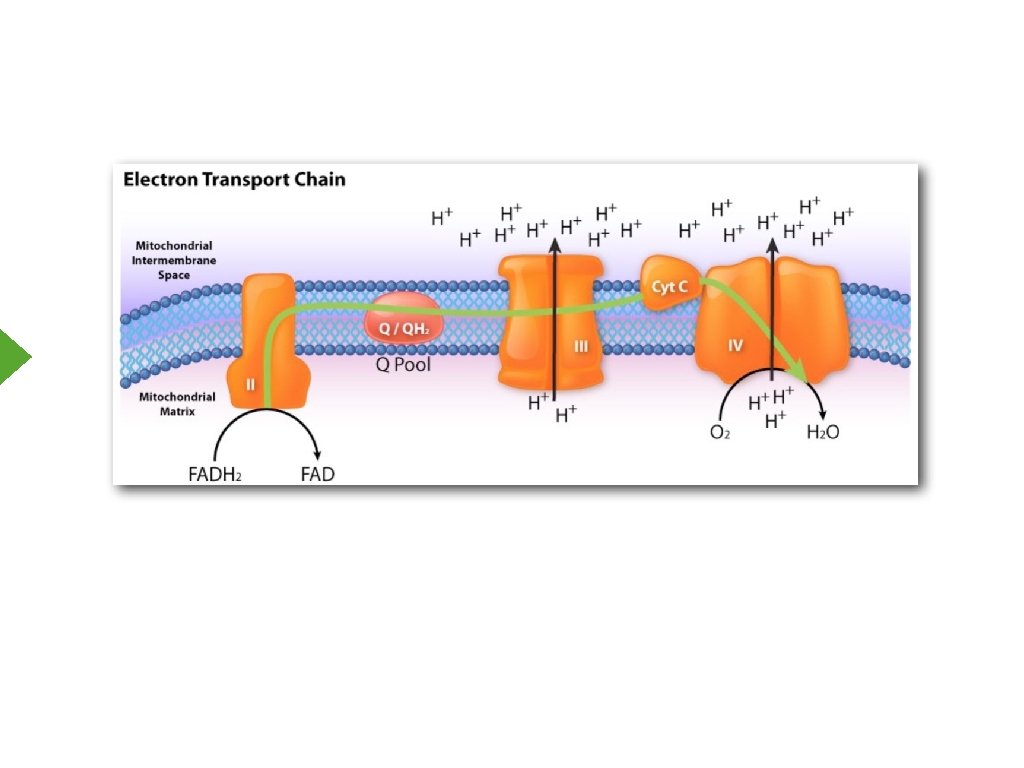
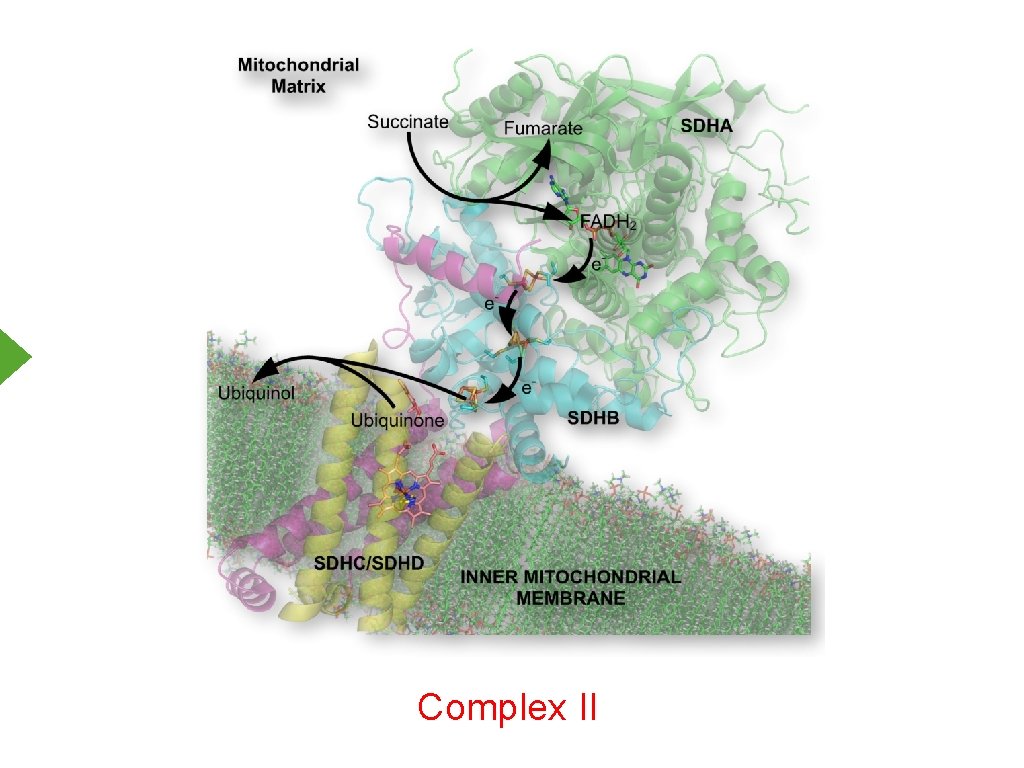
Complex II
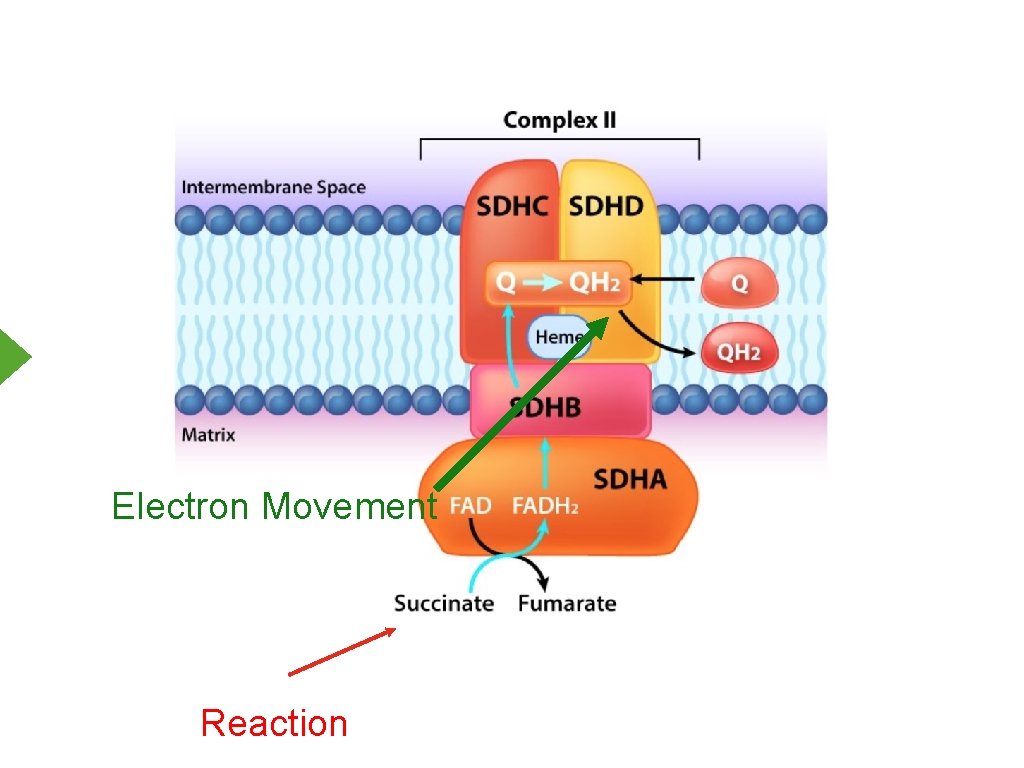
Electron Movement Reaction
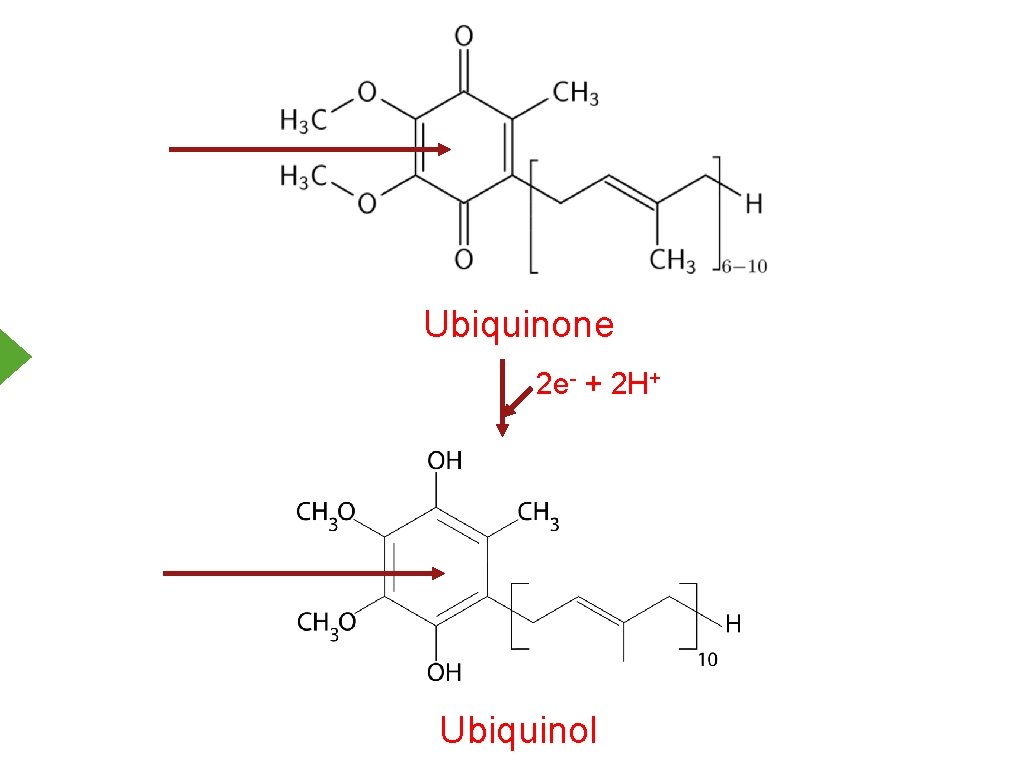
Ubiquinone 2 e- + 2 H+ Ubiquinol
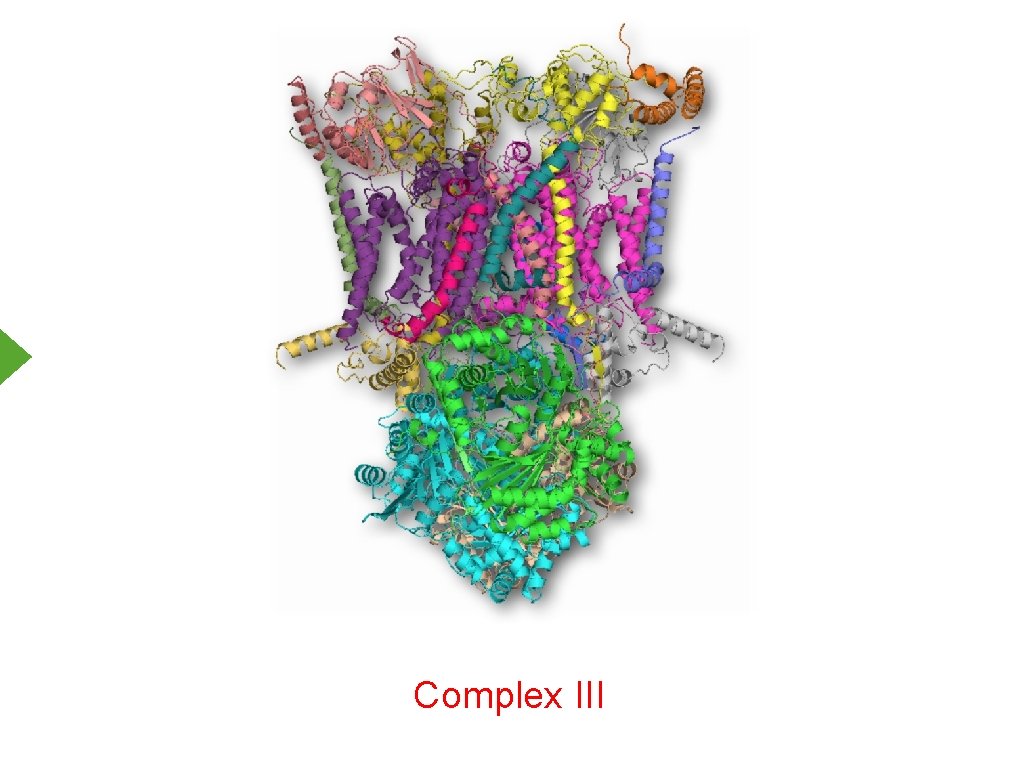
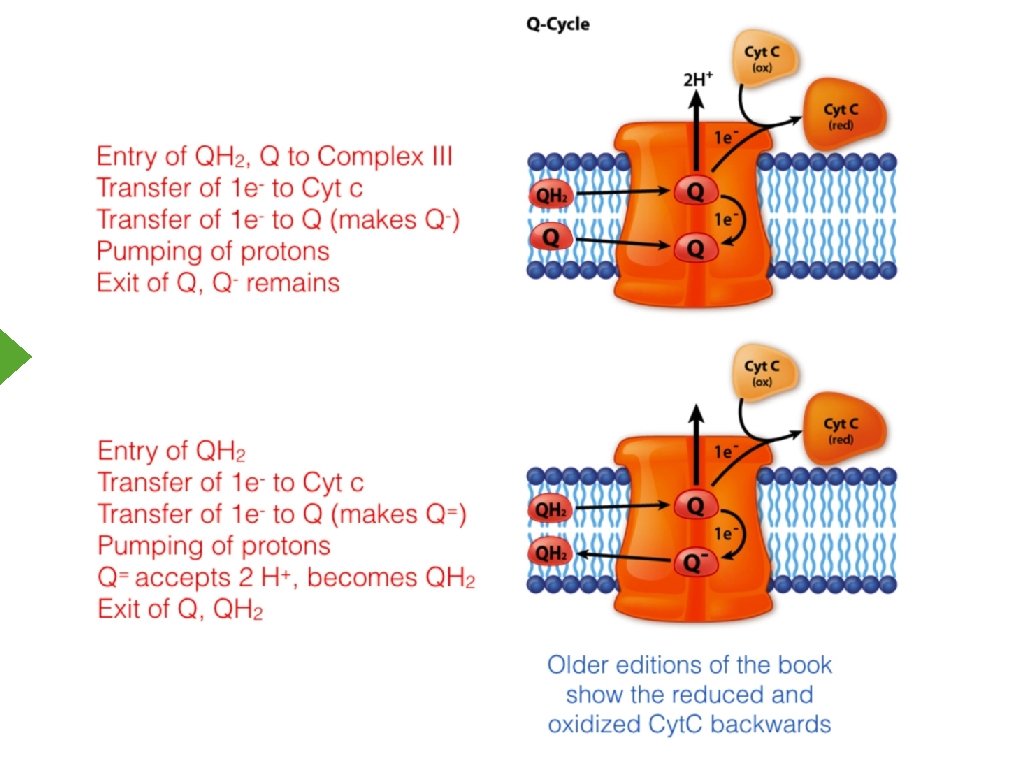
Complex III
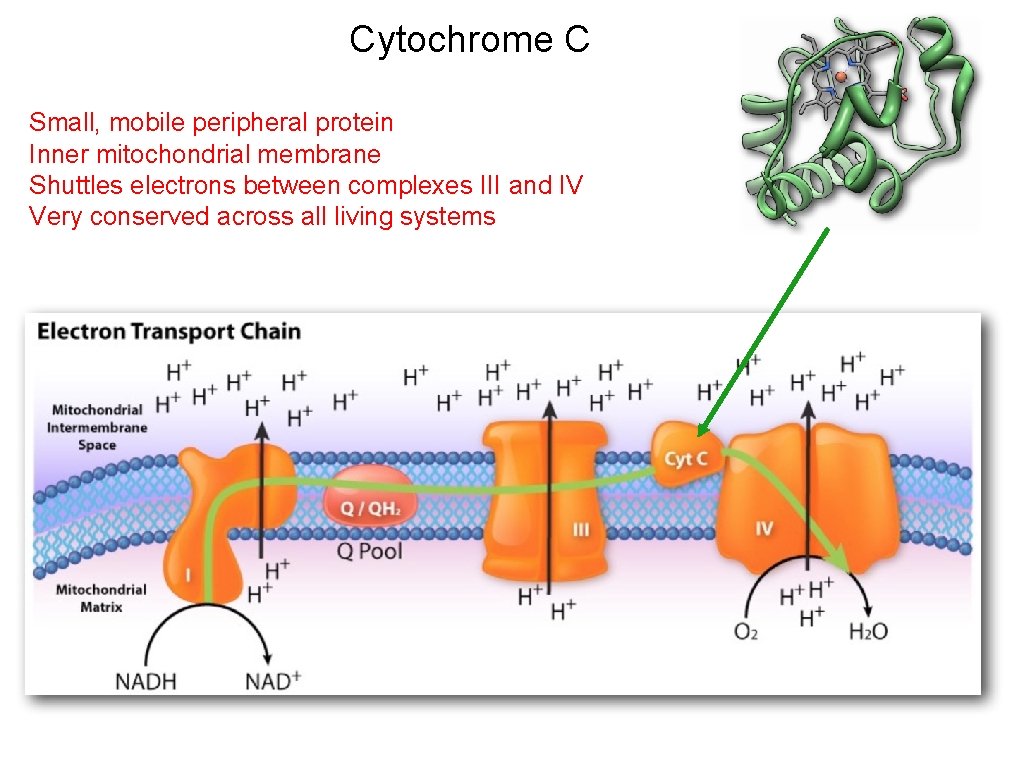
Cytochrome C Small, mobile peripheral protein Inner mitochondrial membrane Shuttles electrons between complexes III and IV Very conserved across all living systems
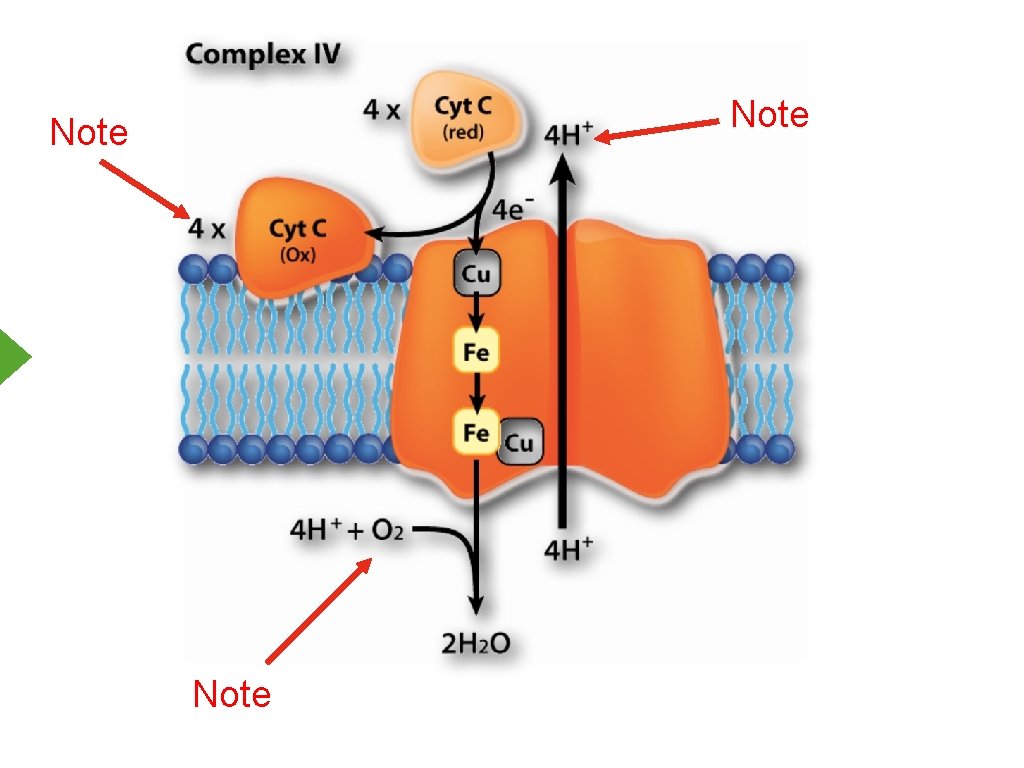
Note
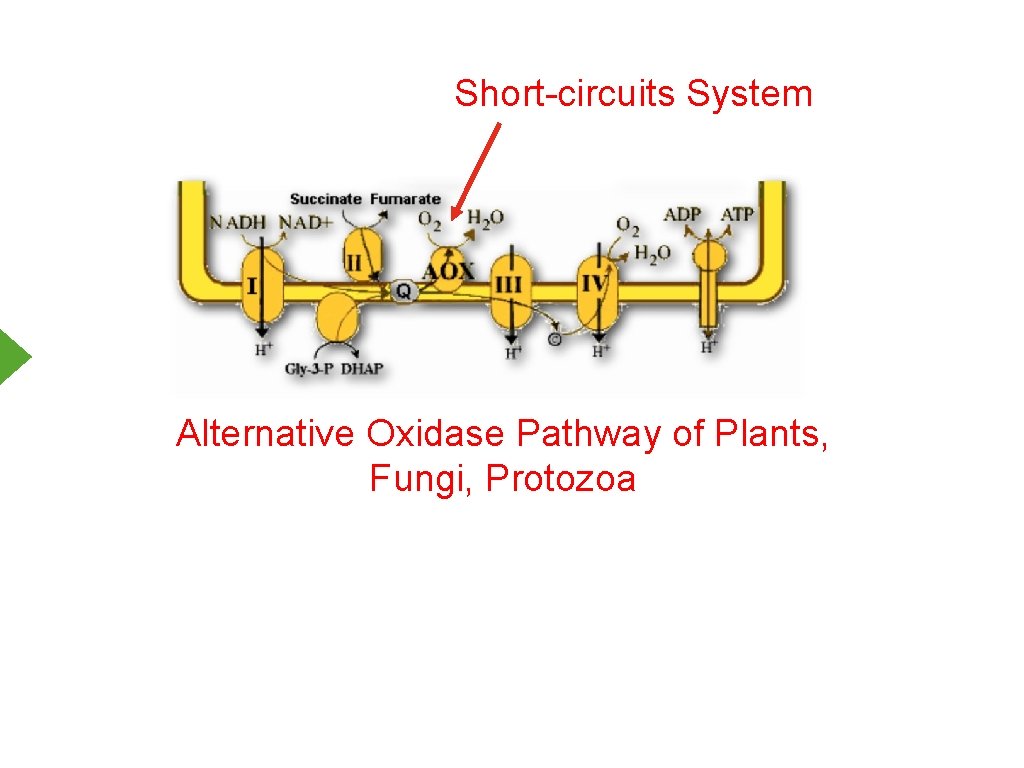
Short-circuits System Alternative Oxidase Pathway of Plants, Fungi, Protozoa

Requirements for Electron Transport 1. Electron carriers (NADH/FADH 2) 2. Oxygen
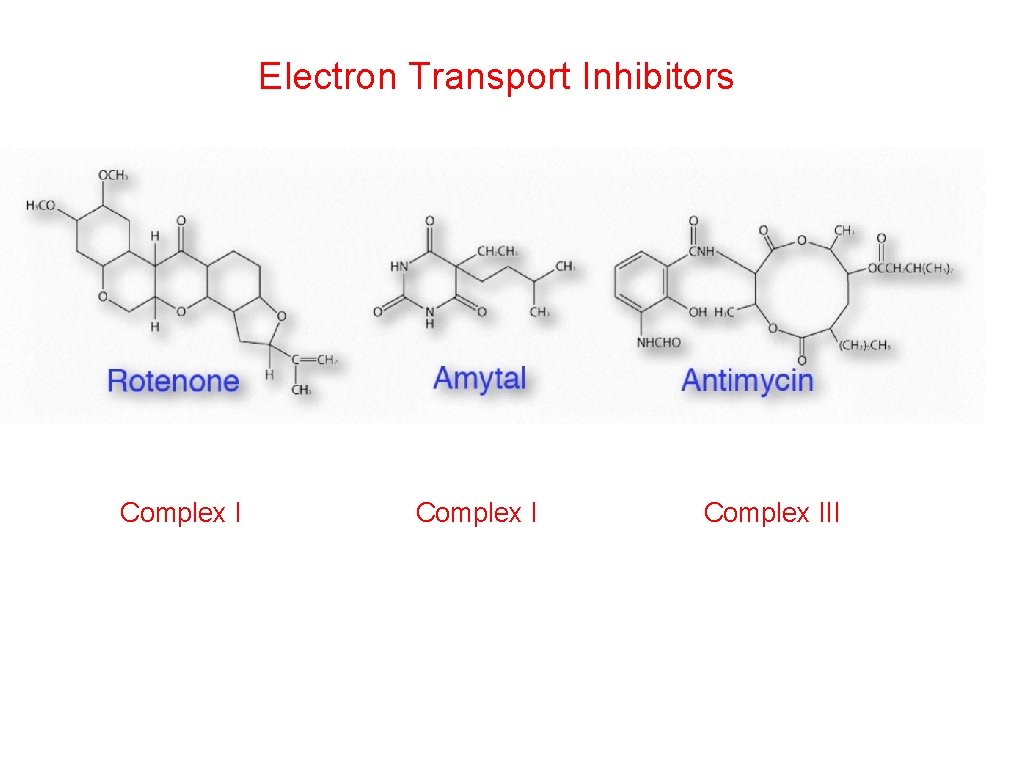
Electron Transport Inhibitors Complex III
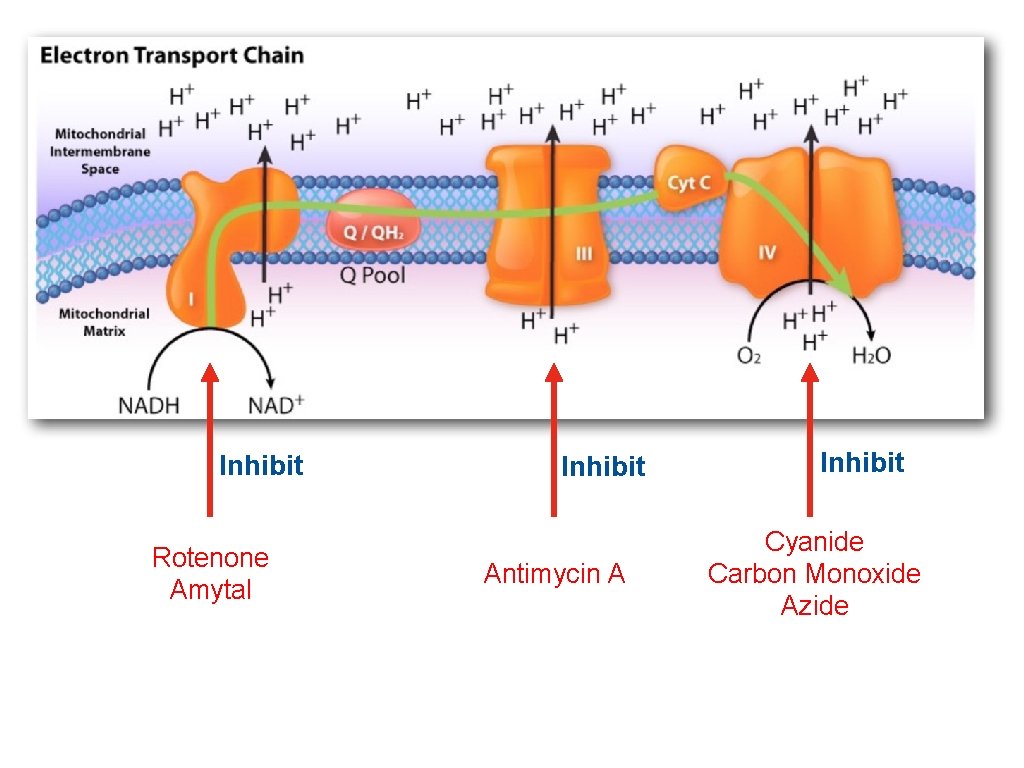
Inhibit Rotenone Amytal Inhibit Antimycin A Inhibit Cyanide Carbon Monoxide Azide
Reactive Oxygen Species
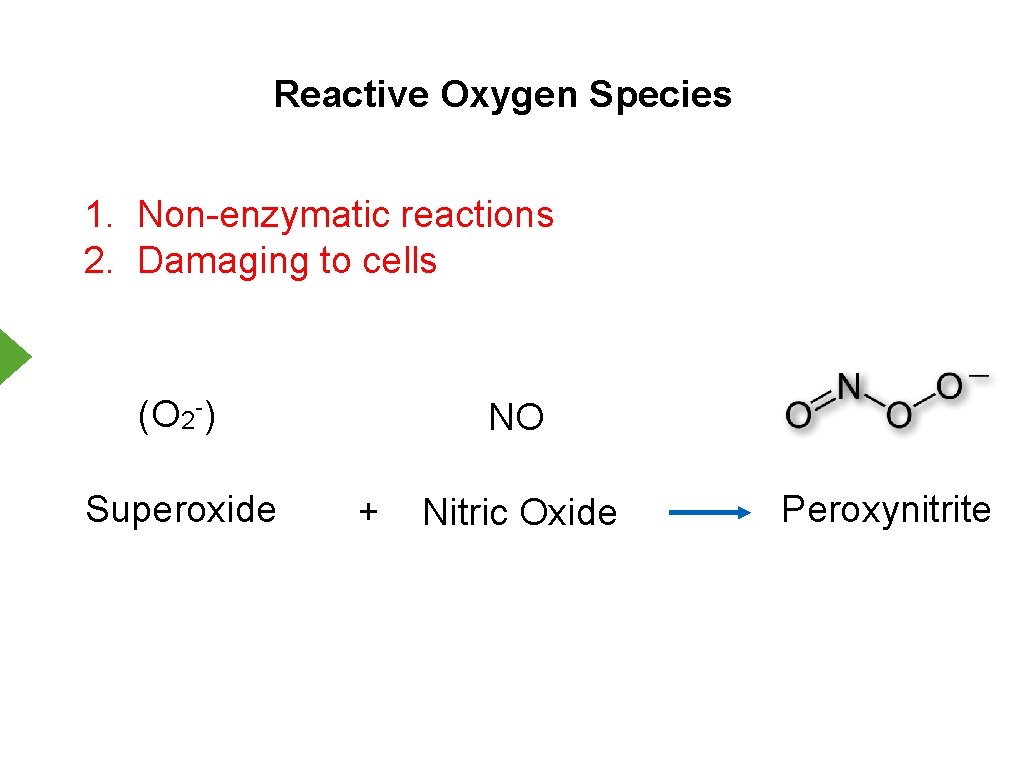
Reactive Oxygen Species 1. Non-enzymatic reactions 2. Damaging to cells (O 2 -) Superoxide NO + Nitric Oxide Peroxynitrite

Reactive Oxygen Species - Cellular Protection 1. Antioxidants a. Glutathione b. Vitamins A, C, E c. Uric acid 2. Enzymes a. Catalase b. Superoxide Dismutase
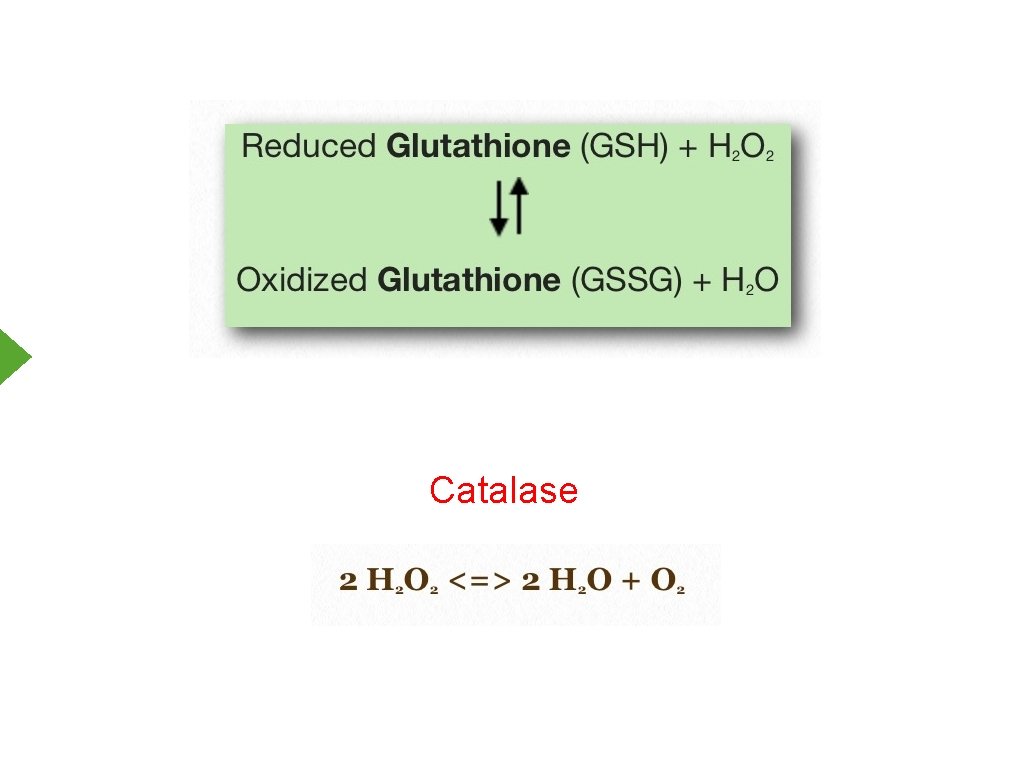
Catalase
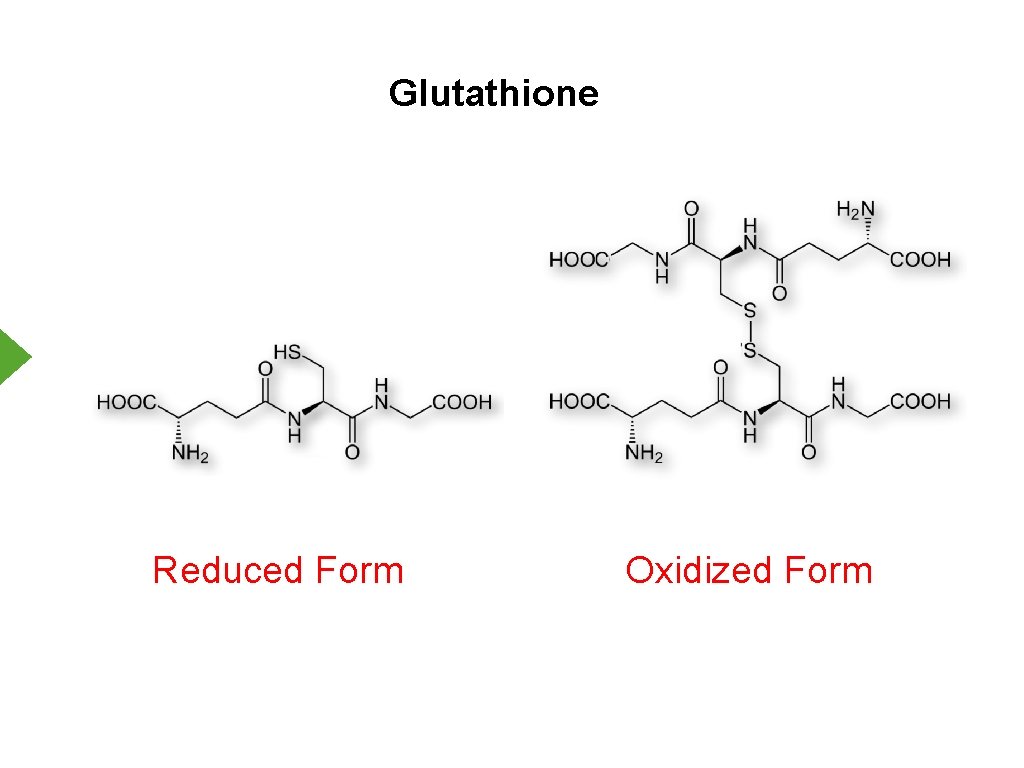
Glutathione Reduced Form Oxidized Form
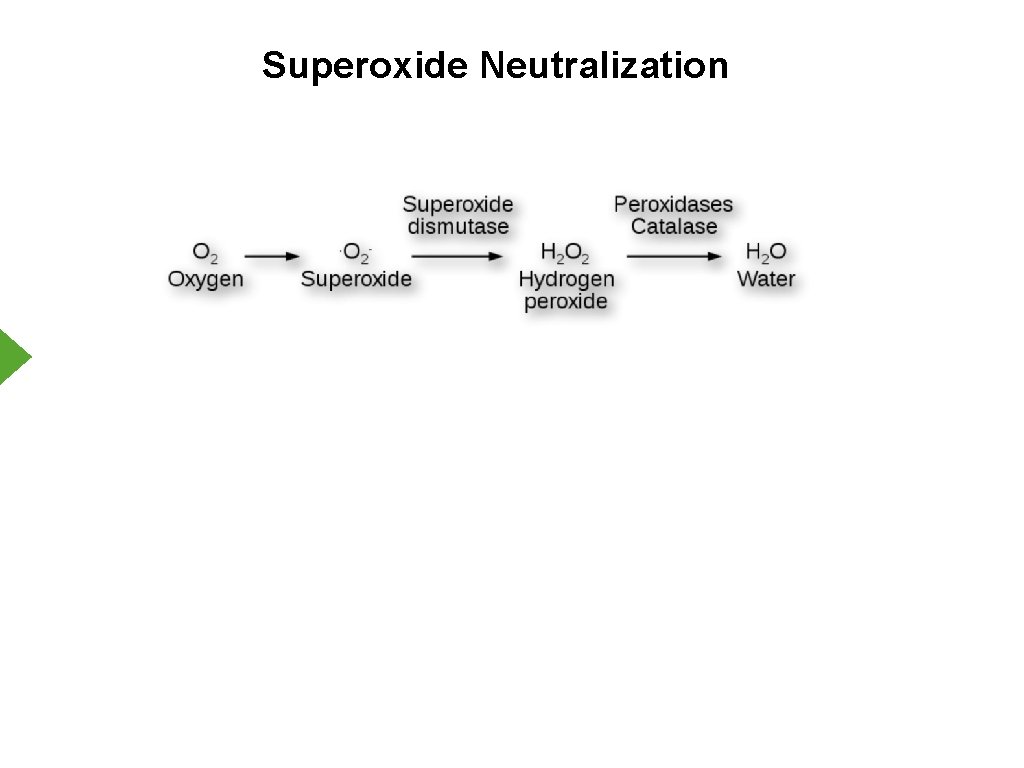
Superoxide Neutralization
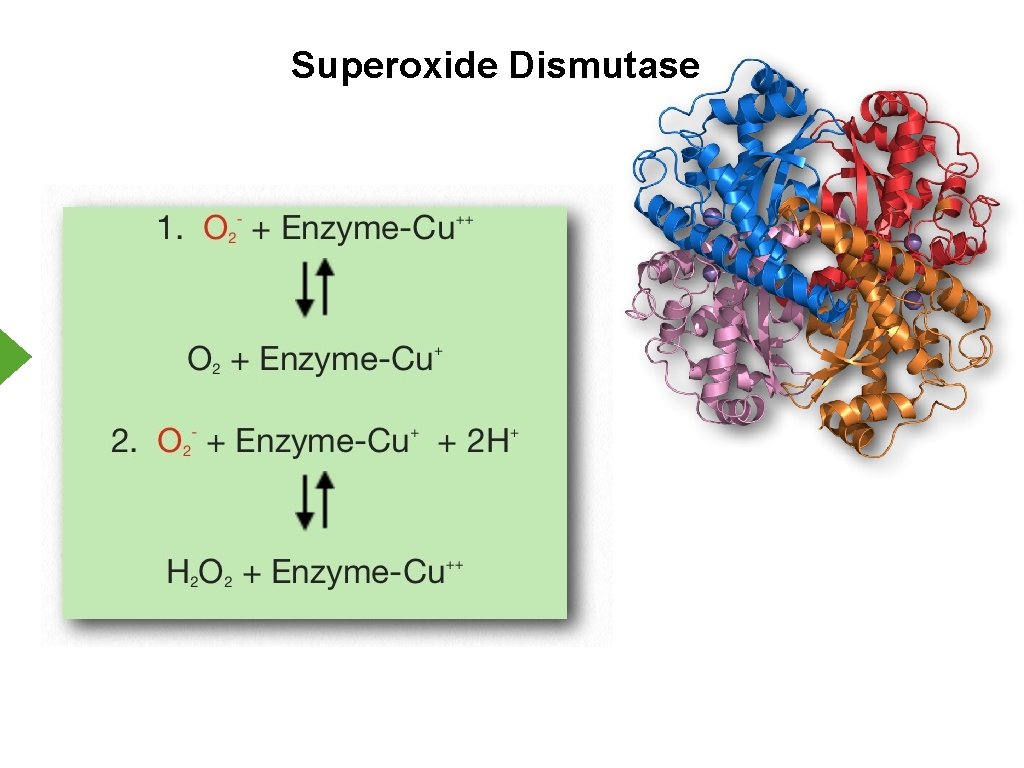
Superoxide Dismutase

Oxidative Phosphorylation Mitchell’s Chemiosmotic Process 1. Intact inner mitochondrial membrane 2. Movement of electrons “pumps” protons 3. Proton gradient drives formation of ATP
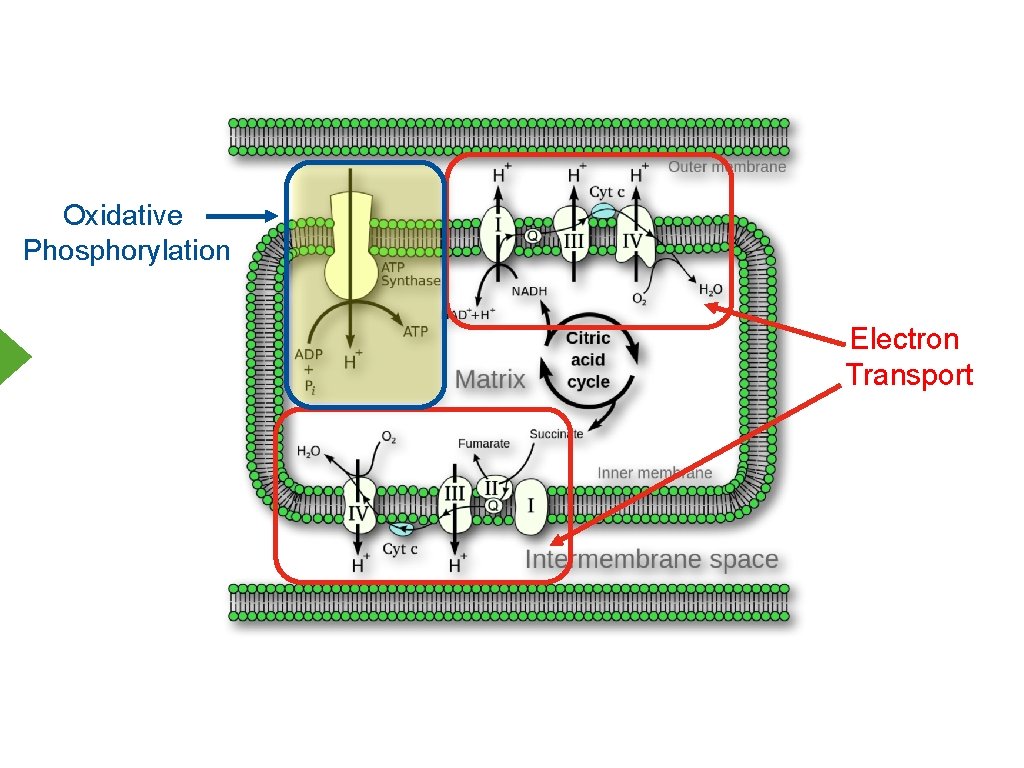
Oxidative Phosphorylation Electron Transport
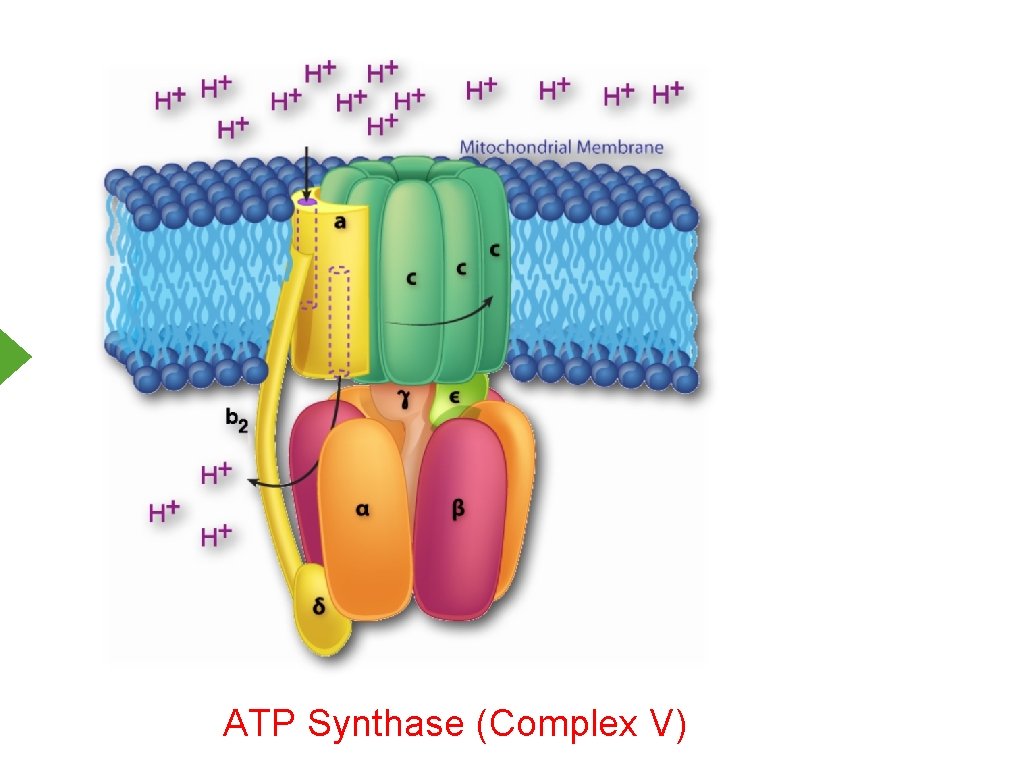
ATP Synthase (Complex V)
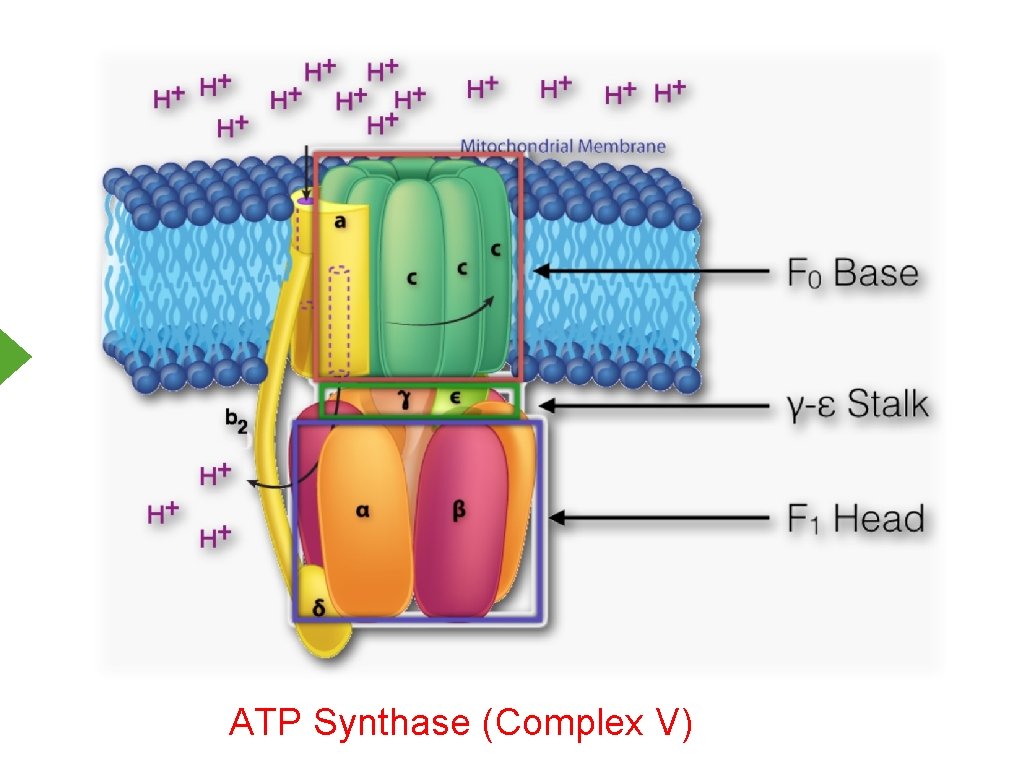
ATP Synthase (Complex V)
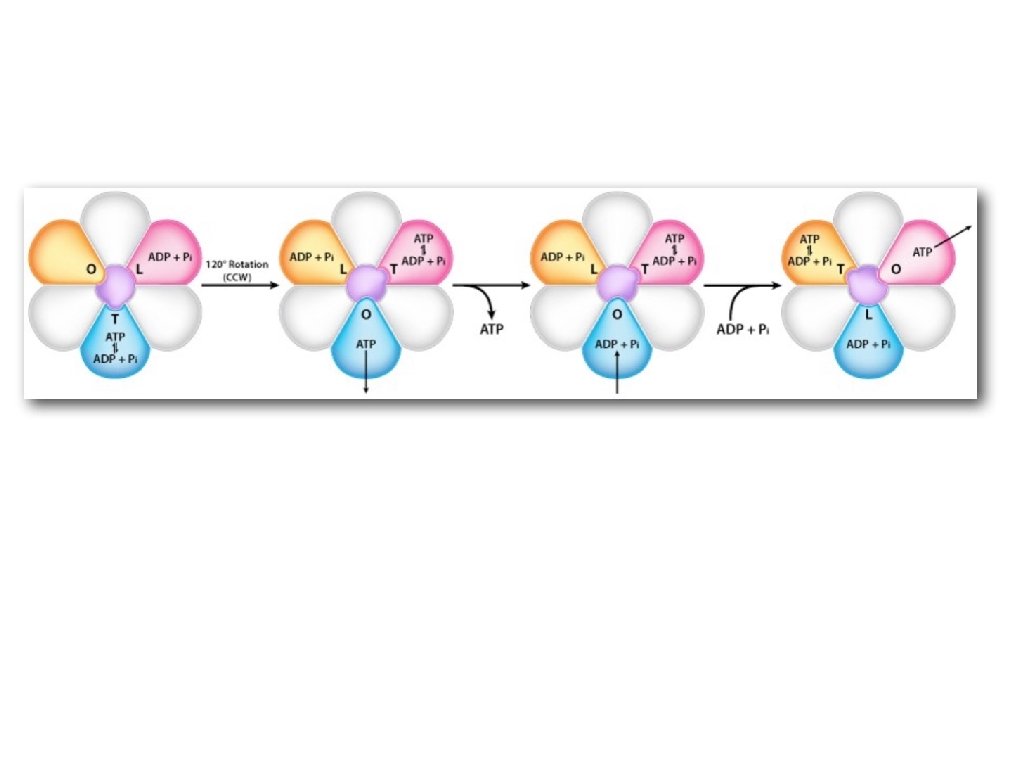


Oxidative Phosphorylation Requirements 1. Proton Gradient 2. ADP
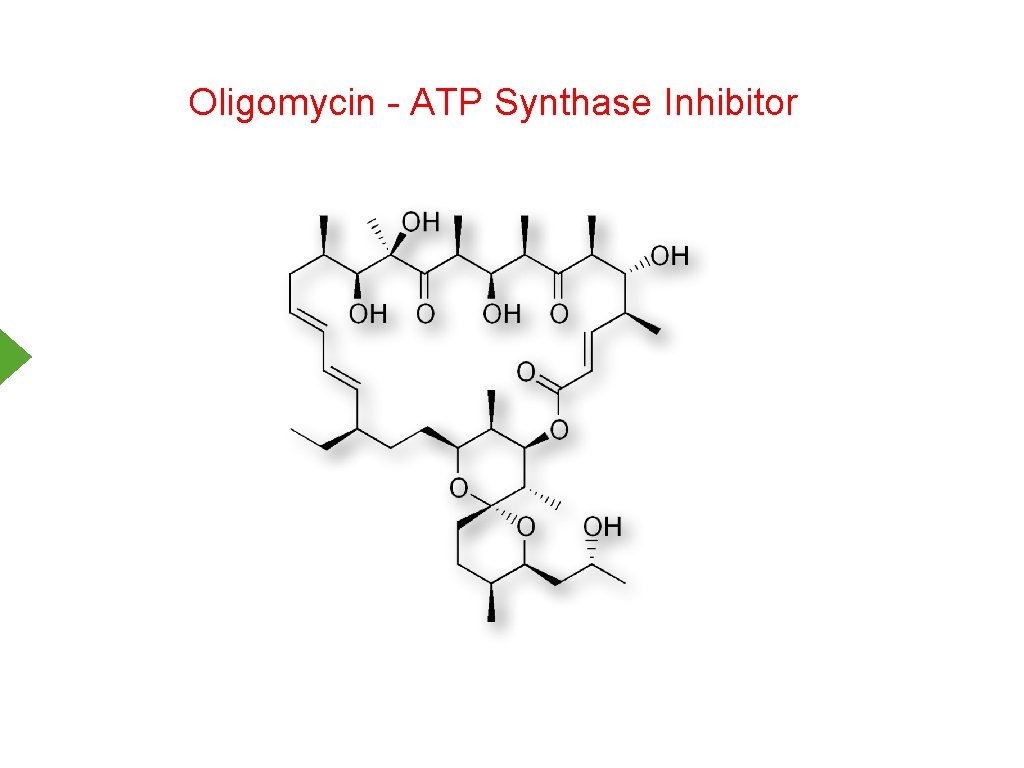
Oligomycin - ATP Synthase Inhibitor

Respiratory Control 1. 2. 3. 4. Tightly coupled vs. Uncoupled Requires intact mitochondrial inner membrane ETS and oxidative phosphorylation are interdependent Stopping either will stop the other

Respiratory Control
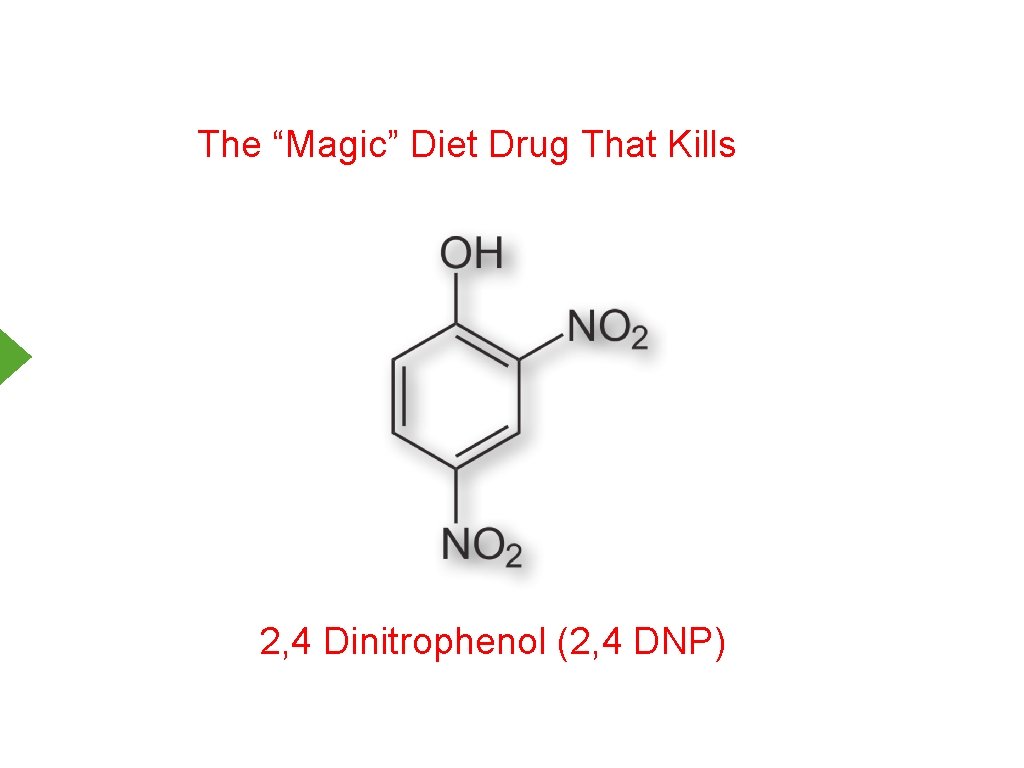
The “Magic” Diet Drug That Kills 2, 4 Dinitrophenol (2, 4 DNP)
- Slides: 46