Electron Transport and Oxidative Phosphorylation Andy Howard Biochemistry

Electron Transport and Oxidative Phosphorylation Andy Howard Biochemistry Lectures 1 November 2010 Ox. Phos + Electron Transport 11/01/2010

Electron Transport n This shows how we can really make ATP from all those reducing equivalents that we amassed during glycolysis and the TCA cycle 11/01/2010 Ox. Phos + Electron Transport 2
What we’ll discuss n Oxidative Phosphorylation n n Generalizations about oxidation-reduction reactions Electron Transport: Complexes I-IV n The proton pump n n Driving protons across the membrane ATP synthase Bookkeeping Control 11/01/2010 Ox. Phos + Electron Transport 3
i. Clicker quiz, question 1 Why does it matter that the G 6 PDH gene is located on the X chromosome? (a) males don’t possess the gene (b) females don’t possess the gene (c) only one copy available per cell (d) no DNA-repair mechanisms available for X-Chromosome genes n 11/01/2010 Ox. Phos + Electron Transport 4

i. Clicker question 2 A reaction that replenishes an intermediate is said to be n (a) anabolic n (b) catabolic n (c) anapleurotic n (d) amphibolic n (e) none of the above. 11/01/2010 Ox. Phos + Electron Transport 5
TCA Cycle and Evolution n n The entire pathway didn’t evolve together Some reactions much older than others Some ran backward in early implementations Several enzymes adapted from amino acid degradation Youngest enzyme: -ketoglutarate dehydrogenase 11/01/2010 Ox. Phos + Electron Transport 6

Aerobes and anaerobes n n n Because of close coupling between TCA cycle and oxidative phosphorylation, the complete TCA cycle is an aerobic phenomenon Anaerobes do have most of these enzymes, but the sequence of reactions is different Oxygen is actually toxic to many anaerobes 11/01/2010 Ox. Phos + Electron Transport 7
Overall role of electron transport n n n Last 3 lectures: we discussed carbohydrate metabolism and the TCA cycle, each of which produced some highenergy phosphate energy directly. In both of those systems much of the energy generated took the form of reduced cofactors--NADH in both systems, and FADH 2 (or QH) in the Krebs cycle. Now we’ll see what happens to those! 11/01/2010 Ox. Phos + Electron Transport 8
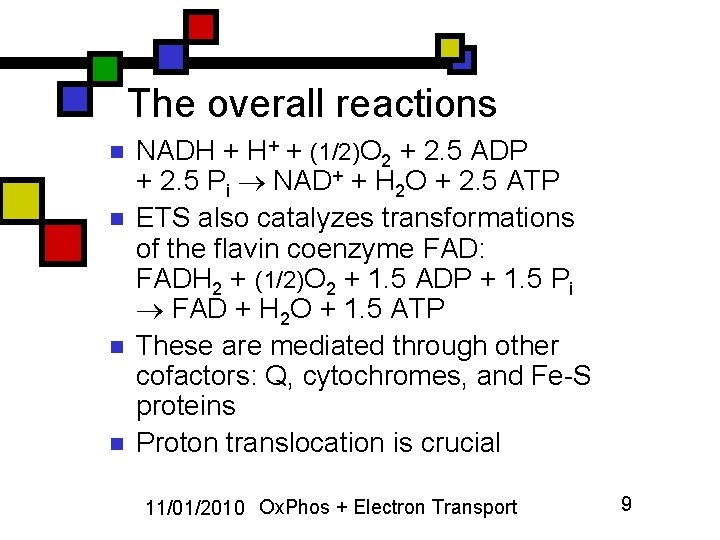
The overall reactions n n NADH + H+ + (1/2)O 2 + 2. 5 ADP + 2. 5 Pi NAD+ + H 2 O + 2. 5 ATP ETS also catalyzes transformations of the flavin coenzyme FAD: FADH 2 + (1/2)O 2 + 1. 5 ADP + 1. 5 Pi FAD + H 2 O + 1. 5 ATP These are mediated through other cofactors: Q, cytochromes, and Fe-S proteins Proton translocation is crucial 11/01/2010 Ox. Phos + Electron Transport 9
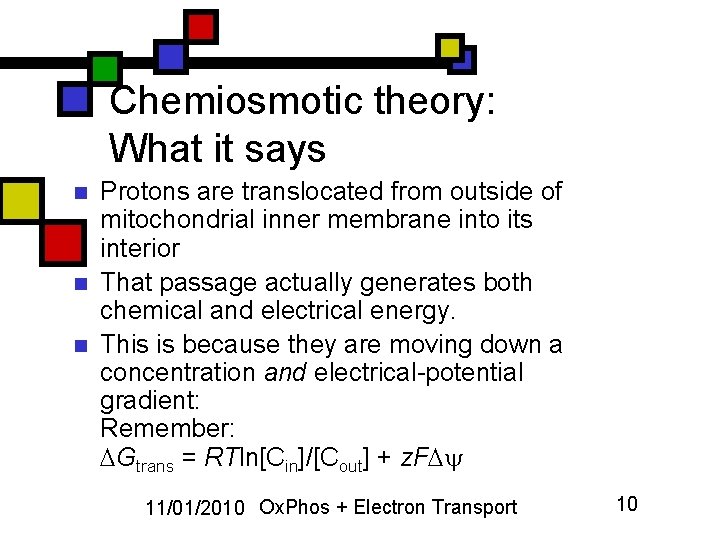
Chemiosmotic theory: What it says n n n Protons are translocated from outside of mitochondrial inner membrane into its interior That passage actually generates both chemical and electrical energy. This is because they are moving down a concentration and electrical-potential gradient: Remember: Gtrans = RTln[Cin]/[Cout] + z. F 11/01/2010 Ox. Phos + Electron Transport 10

How it works n n This energy is used to drive the synthesis of ATP from ADP and Pi within an enzyme called ATP synthase, which is (big surprise!) anchored on the inside of the inner mitochondrial membrane. The structure of two components of this enzyme system were determined in 1999 by Andrew Leslie and others. 11/01/2010 Ox. Phos + Electron Transport 11

Oxidation state and energy n n We typically measure oxidation states in volts. We can relate the energy associated with an oxidation-reduction reaction— the so-called change in redox potential— with the change in the oxidation state of the molecules involved in the reaction. 11/01/2010 Ox. Phos + Electron Transport 12
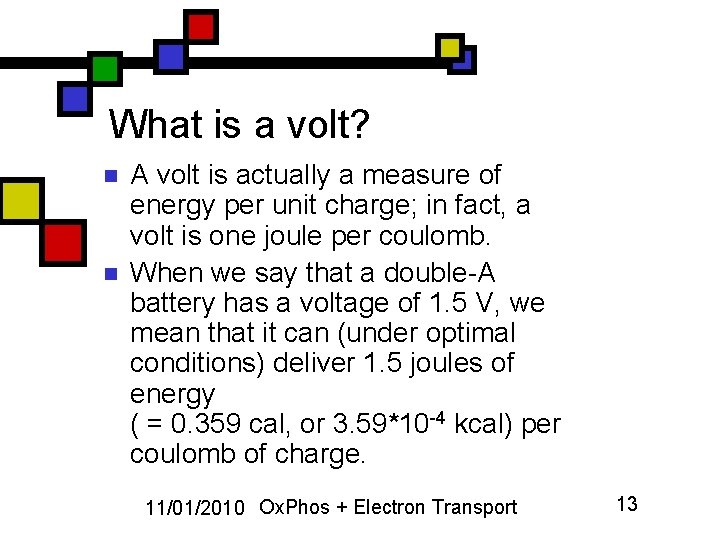
What is a volt? n n A volt is actually a measure of energy per unit charge; in fact, a volt is one joule per coulomb. When we say that a double-A battery has a voltage of 1. 5 V, we mean that it can (under optimal conditions) deliver 1. 5 joules of energy ( = 0. 359 cal, or 3. 59*10 -4 kcal) per coulomb of charge. 11/01/2010 Ox. Phos + Electron Transport 13
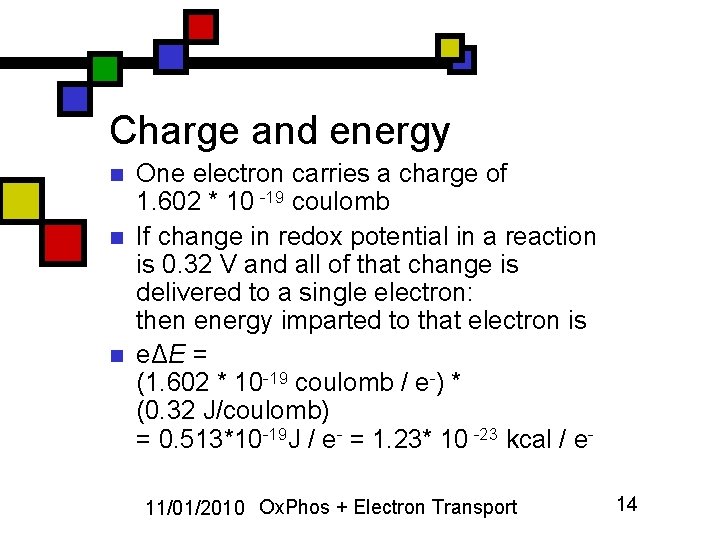
Charge and energy n n n One electron carries a charge of 1. 602 * 10 -19 coulomb If change in redox potential in a reaction is 0. 32 V and all of that change is delivered to a single electron: then energy imparted to that electron is eΔE = (1. 602 * 10 -19 coulomb / e-) * (0. 32 J/coulomb) = 0. 513*10 -19 J / e- = 1. 23* 10 -23 kcal / e 11/01/2010 Ox. Phos + Electron Transport 14

… in biochemical units … n n That doesn't sound like much, but if we look at that on a per mole basis, 0. 32 V applied to a single electron is (1. 23 * 10 -23 kcal/e-) * 6. 022 * 1023 e -/mole = 30. 87 k. J/mol = 7. 38 kcal/mol which is a reasonable amount of energy on the scale we're accustomed to examining (~1 ATP hydrolysis). 11/01/2010 Ox. Phos + Electron Transport 15
So what can we get? n n There is enough energy bound up in the reduced state of NAD relative to the oxidized state to drive the net creation of 2. 5 molecules of ATP from ADP and phosphate, as indicated in the equations shown above. Since there are NADH molecules created in several steps in glycolysis and the Krebs cycle, there numerous net ATP molecules that arise from the overall process. 11/01/2010 Ox. Phos + Electron Transport 16
Results from TCA cycle n n n 3 NADH produce 7. 5 ATP 1 FADH 2 produces 1. 5 ATP 1 substrate-level phosphorylation (from succinyl Co. A hydrolysis) Total: 10 ATP per round, if we don’t get interrupted! Since we formally shove 2 molecules of acetyl Co. A into the system per molecule of glucose input into glycolysis, we get 20 ATPs out of the TCA cycle per glucose. 11/01/2010 Ox. Phos + Electron Transport 17
ETS: The big picture n n n 5 membrane-associated, multi-enzyme complexes in mitochondrial inner membrane Complexes I-IV associated with electron transport and proton translocation: Complexes I, III, IV move protons Complex V uses that proton gradient to produce ATP from ADP and Pi 11/01/2010 Ox. Phos + Electron Transport 18

Complexes I-IV n There are several multi-enzyme complexes involved in converting the reductive energy in NADH to its final products. # Name I NADH-Ubiquinone oxidoreductase II Succinate-ubiquinone oxidoreductase III Ubiquinol-cytochrome c oxidoreductase IV Cytochrome c oxidase 11/01/2010 Ox. Phos + Electron Transport 19
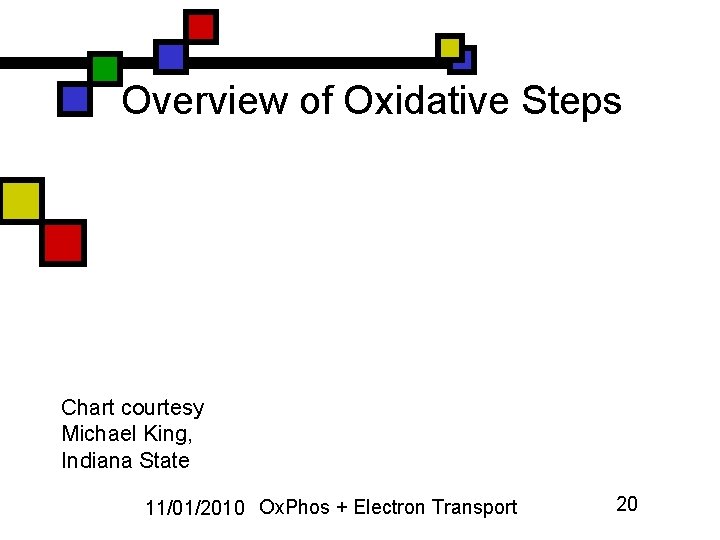
Overview of Oxidative Steps Chart courtesy Michael King, Indiana State 11/01/2010 Ox. Phos + Electron Transport 20

Reduced cofactors to ATP n We will discuss how the energy latent in these reduced cofactors can be turned into energy in the form of highenergy phosphate bonds in nucleoside triphosphates--the standard currency of energy. 11/01/2010 Ox. Phos + Electron Transport 21

What the ETS does n n The electron transport system (ETS) is responsible for these transformations. Like the Krebs cycle or glycolysis, the electron transport chain is a series of chemical transformations facilitated by proteins. 11/01/2010 Ox. Phos + Electron Transport 22

Roles of these systems n n n Some of these proteins are enzymes in the conventional sense others are not—they're electron transport proteins only: so they can only be regarded as enzymes if we allow that the entire ETS is a large, multi-polypeptide transformation system--a multi-component enzyme, analogous to the pyruvate decarboxylase complex or the fatty acid synthase complex 11/01/2010 Ox. Phos + Electron Transport 23
Flavin cofactors n n Participants in ETS Sometimes depicted as starting points, but it’s probably better to think of them as intermediaries 11/01/2010 Ox. Phos + Electron Transport 24

Complex I n n n NADH: Ubiquinone oxidoreductase Embedded in inner mitochondrial membrane Passes electrons from NADH to ubiquinone: 11/01/2010 Ox. Phos + Electron Transport 25

Properties of ubiquinone n n The quinone moiety is where the oxidation-reduction takes place (1 e- or 2 e- at a time) The hydrophobic tail (15 C = 3 isoprene units) anchors this cofactor to the mitochondrial membrane 11/01/2010 Ox. Phos + Electron Transport 26

Protons in Complex I n n Complex I picks up a pair of protons from the matrix and passes them up through to the other side of the membrane Energy for this translocation (against the concentration gradient) supplied by oxidation of NADH Diagram courtesy of Rice University 11/01/2010 Ox. Phos + Electron Transport 27
Complex II n n n Succinate-ubiquinone oxidoreductase We’ve looked at this already as succinate dehydrogenase Succinate + Q Fumarate + QH 2 No protons translocated in this step: Reaction is close to isoergic FAD and Fe-S proteins involved 11/01/2010 Ox. Phos + Electron Transport PDB 2 FBW 246 k. Da dimer of heterotetamers Chicken 28
Complex III n n n Ubiquinol: cytochrome c oxidoreductase Transfers electrons from reduced ubiquinol to iron atoms in cytochrome c One electron per cytochrome c Enzyme contains three main subunits: cytochrome c 1, cytochrome b, and an iron-sulfur (“Rieske”) protein Net reactions: n n QH 2 + cyto c–Fe 3+ QH • + cyto c–Fe 2++ H+ QH • + cyto c–Fe 3+ Q + cyto c–Fe 2+ + H+ 11/01/2010 Ox. Phos + Electron Transport 29
Complex III structures n Cytochrome bc 1 complex n Rieske Fe-S protein 11/01/2010 Ox. Phos + Electron Transport 30
Cytochrome-dependent steps n n Cytochromes are, in general, proteins involved in electron transport. Cytochrome c is a mobile, soluble compound Others are generally membraneassociated What they have in common: covalently bound heme 11/01/2010 Ox. Phos + Electron Transport 31

Cytochromes n n n The name derives from the fact that many of them are colored (–chrome) and found in cells (cyto–). In particular, cytochrome c, which is a significant intermediary in the ETS, is a water-soluble, relatively small, heme-containing protein Cytochrome c received substantial attention in the early years of biochemistry both because of its inherent importance and because it's easy to study. 11/01/2010 Ox. Phos + Electron Transport 32
Where do the individual names come from? n n n Early analyses of cellular or mitochondrial extracts showed several peaks in the absorption spectra Cytochromes were named as those spectral peaks became associated with specific spectral peaks Sometimes two different species (e. g. cytochrome c and cytochrome c 1) have very similar spectra 11/01/2010 Ox. Phos + Electron Transport 33

Cytochrome c and evolution Cytochrome c is highly conserved n rate of mutation across the billions of years of evolution is remarkably slow, as compared to other proteins. n This is generally a sign that its function is sufficiently irreplacable that even a modest modification in the protein renders the cell unviable. 11/01/2010 Ox. Phos + Electron Transport 34
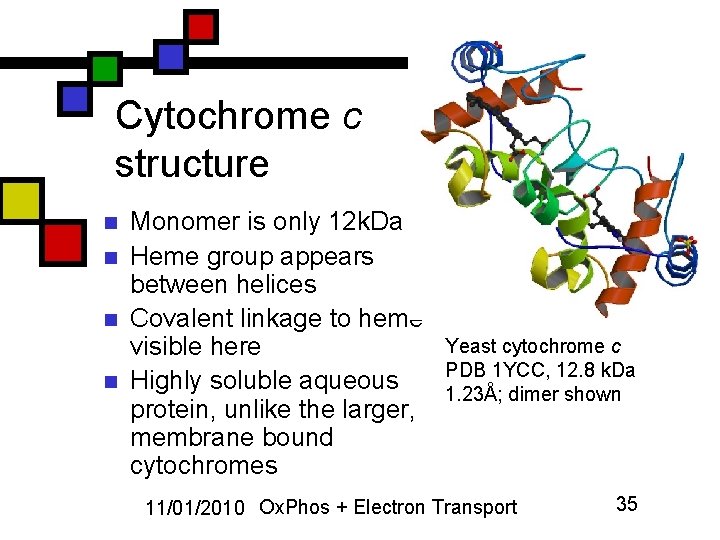
Cytochrome c structure n n Monomer is only 12 k. Da Heme group appears between helices Covalent linkage to heme visible here Highly soluble aqueous protein, unlike the larger, membrane bound cytochromes Yeast cytochrome c PDB 1 YCC, 12. 8 k. Da 1. 23Å; dimer shown 11/01/2010 Ox. Phos + Electron Transport 35

Cytochrome c and apoptosis n n Cytochrome c plays a role in apoptosis: When a cell receives (and pays attention to) an external apoptotic signal, typically via IP 3 signaling pathway, Ca 2+ is released from the ER One of the first responses is release of cytochrome c from the mitochondrion into the cytosol This triggers lytic events that eventually lead to shrinkage and absorption of the cell fragment 11/01/2010 Ox. Phos + Electron Transport 36

Complex 3: schematic Courtesy U. Texas 11/01/2010 Ox. Phos + Electron Transport 37
Complex IV n n n Cytochrome c oxidase Transfers electrons from (soluble) cytochrome c to molecular oxygen: Product is water 2 Cyto c–Fe 2+ + (1/2)O 2 + 2 H+ 2 Cyto c–Fe 3+ + H 2 O 11/01/2010 Ox. Phos + Electron Transport 38

Cytochrome oxidase n n 2 functional units; Up to 13 subunits containing membrane-spanning helices 4 protons produced per oxidation of two molecules of QH 2 at the Q 0 site See fig. 14. 13 11/01/2010 Ox. Phos + Electron Transport 39
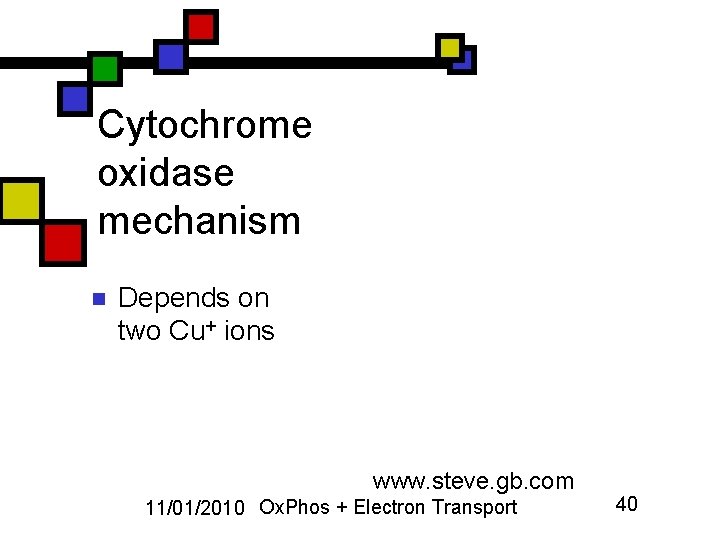
Cytochrome oxidase mechanism n Depends on two Cu+ ions www. steve. gb. com 11/01/2010 Ox. Phos + Electron Transport 40

Mitochondrial sequestration n Mitochondrion is a fairly complex organelle, found in all eukaryotes. Some simple algae have one mitochondrion per cell, whereas some protozoa have a half-million per cell. A mammalian liver cell contains about 5000 mitochondria. These organelles resemble bacteria in size and complexity. 11/01/2010 Ox. Phos + Electron Transport 41

Mitochondrial genetics n n Vertebrate mitochondrion has its own chromosome, but it does not code for many proteins Human mitochondrion codes for 17 proteins, plus two dozen specialized t. RNAs and (presumably) some control elements. 11/01/2010 Ox. Phos + Electron Transport 42

Where is the rest of the mitochondrion’s function specified? n n Those 17 proteins are ~2% of the functioning proteins in the mitochondrion Most of the ~1000 proteins that function in the mitochondrion are coded for in the host genome and are translocated, sometimes with some amount of proteolytic processing, from the ribosomal protein-synthesis mechanisms of the cytosol into the interior of the mitochondrion. 11/01/2010 Ox. Phos + Electron Transport 43

Mitochondrial structure n n n The mitochondrion has a smooth outer membrane and a highly convoluted inner membrane Intermembrane space between them Outer membrane is permeable to small molecules, so functionally the intermembrane space is equivalent to the cytosol. 11/01/2010 Ox. Phos + Electron Transport 44

Mitochondrial localization n In eukaryotes, ETS and Krebs reactions take place in the mitochondrion. Many reactions occur on the inner membrane in its folded surfaces called cristae. Localization to the membrane provides for orderly passage of substrates or electrons from one protein to the next, helping to defeat old man entropy. 11/01/2010 Ox. Phos + Electron Transport 45
Mitochondrial matrix enzymes Some proteins do function in the matrix, the aqueous compartment of the mitochondrion interior to the inner membrane. n Pyruvate dehydrogenase complex n TCA-cycle enzymes (except succinate dehydrogenase, which is embedded in the inner membrane) n Some enzymes involved in fatty acid oxidation. 11/01/2010 Ox. Phos + Electron Transport 46

What’s the matrix like? n n This matrix is has such a high overall protein concentration that it is not really an aqueous medium; it's a gel. Think of reactions that occur in the mitochondrial matrix as occuring in Karo syrup (except the syrup is made of protein + H 2 O, not sugar + H 2 O) 11/01/2010 Ox. Phos + Electron Transport 47

Regulation of ETS n An element of regulation of electron transport is based on the availability of adenosine diphosphate, the substrate for the oxygen-mediated respiration steps. Mitochondria consume oxygen rapidly when ADP is available. 11/01/2010 Ox. Phos + Electron Transport 48
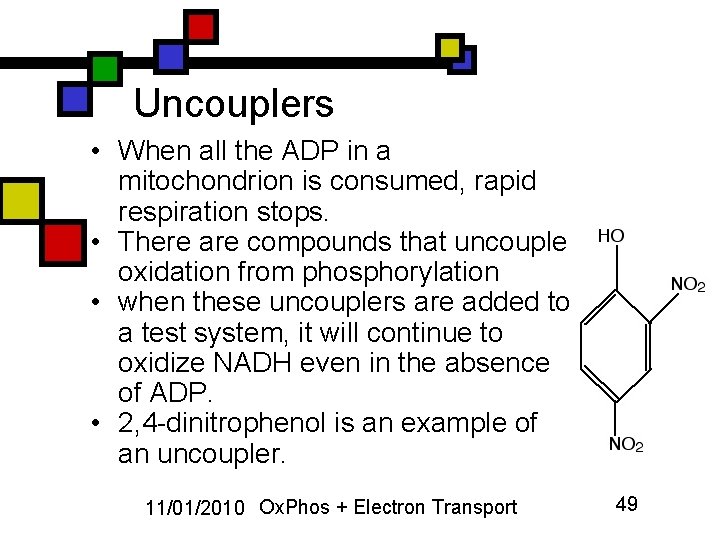
Uncouplers • When all the ADP in a mitochondrion is consumed, rapid respiration stops. • There are compounds that uncouple oxidation from phosphorylation • when these uncouplers are added to a test system, it will continue to oxidize NADH even in the absence of ADP. • 2, 4 -dinitrophenol is an example of an uncoupler. 11/01/2010 Ox. Phos + Electron Transport 49
ATP synthase n n n Crucial example of a molecular motor, i. e. , a machine that translates between chemical energy and mechanical work Ultimately its job is to pull protons across the membrane, using the energy associated with that favorable translocation to drive the synthesis of ATP from ADP and Pi The motor is what rotates the protein through various positions to enable its reactions 11/01/2010 Ox. Phos + Electron Transport 50
Chemiosmotic theory: history n n How are these reactions actually employed to drive ATP synthesis? This was hotly debated for many years. Problem was finally solved in 1960's: chemiosmotic theory, which links ATP synthesis to proton translocation across the membrane Oxidations in complexes I, III, and IV is what drove the protons against a concentration gradient in the first place 11/01/2010 Ox. Phos + Electron Transport 51

The developers n n Peter Mitchell, Paul Boyer, Ephraim Racker, and others. Mitchell received the Chemistry Nobel Prize in 1978; Boyer received his nineteen years later, to the immense satisfaction of those who felt he had been passed over the first time around. 11/01/2010 Ox. Phos + Electron Transport 52

Chemiosmotic hypothesis n n Recognizes that protons have been pushed across the membrane against the concentration gradient Flow back across generates energy, which is harnessed to perform rotations of the ATP synthase molecule, driving synthesis of ATP 11/01/2010 Ox. Phos + Electron Transport 53

Structural realities: F 0 n n F 0 component is transmembrane helical domain: Grabs protons, channels them inward F 0 rotates 120º with each translocation 11/01/2010 Ox. Phos + Electron Transport 54
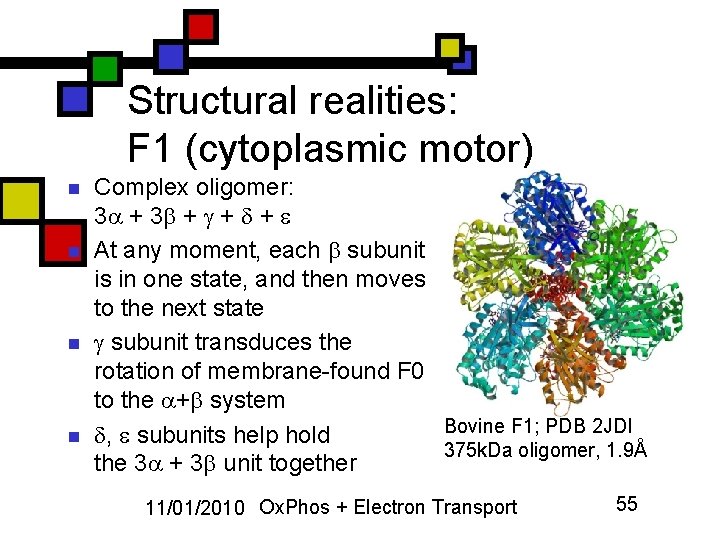
Structural realities: F 1 (cytoplasmic motor) n n Complex oligomer: 3 + + + At any moment, each subunit is in one state, and then moves to the next state subunit transduces the rotation of membrane-found F 0 to the + system Bovine F 1; PDB 2 JDI , subunits help hold 375 k. Da oligomer, 1. 9Å the 3 + 3 unit together 11/01/2010 Ox. Phos + Electron Transport 55

ATP Synthase: Details n n ADP and Pi bound to 1 subunit (open site) Fully-formed but bound ATP attached at second subunit (the loose site) ATP ready for release located in third subunit (the tight site) After one event, the entire complex rotates 120º; process continues Diagram courtesy of Gonzaga U. 11/01/2010 Ox. Phos + Electron Transport 56
ATP Synthase mechanism n n n Wikipedia provides up-to-date info Careful explanation in section 14. 9; attend to it! 130 revolutions per second observed in model studies with actin attached to the gamma (rotating) subunit 11/01/2010 Ox. Phos + Electron Transport 57
i. Clicker question #3 Which of these proteins would you not expect to contain membrane-spanning helices? n n n (a) cytochrome bc 1 complex (b) complex IV (c) cytochrome c (d) complex V, F 0 component (e) all of those proteins will have membranespanning helices 11/01/2010 Ox. Phos + Electron Transport 58
i. Clicker question #4 Suppose a cell has a non-functional subunit in its ATP synthase. This cell will be able to n n n (a) Oxidize NADH + H+ to NAD (b) Transport protons against a gradient across the inner mitochondrial membrane (c) Harness the energy from proton translocation to phosphorylate ADP (d) (a) and (b) only (e) (a), (b), and (c). 11/01/2010 Ox. Phos + Electron Transport 59
- Slides: 59