Electron Spin Resonance Investigations of Matrix Isolated Metal








- Slides: 13

Electron Spin Resonance Investigations of Matrix Isolated Metal Clusters at 4 K Bradford Michael Mess, John J. Banisaukas III, and Lon B. Knight, Jr. Department of Chemistry, Furman University, Greenville, SC

Introduction • Studying metal clusters is necessary to understand the transition from atomic and molecular properties to metallic bulk properties • Metal clusters also play an important role in developing new catalysts

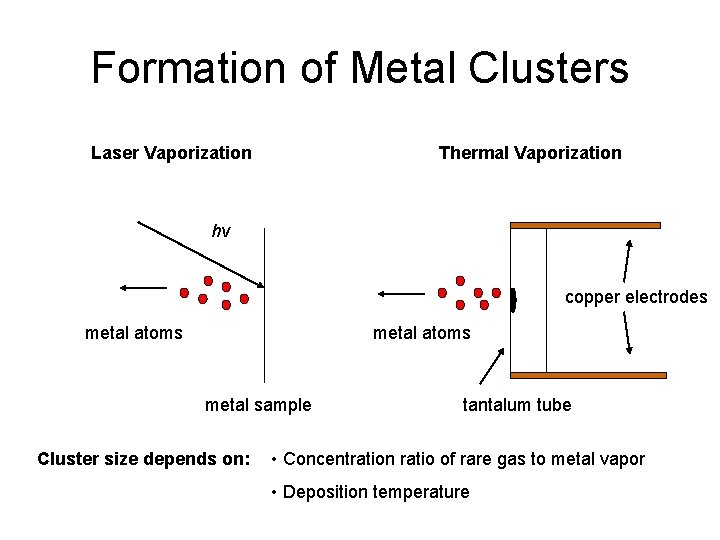
Formation of Metal Clusters Laser Vaporization Thermal Vaporization hν copper electrodes metal atoms metal sample Cluster size depends on: tantalum tube • Concentration ratio of rare gas to metal vapor • Deposition temperature

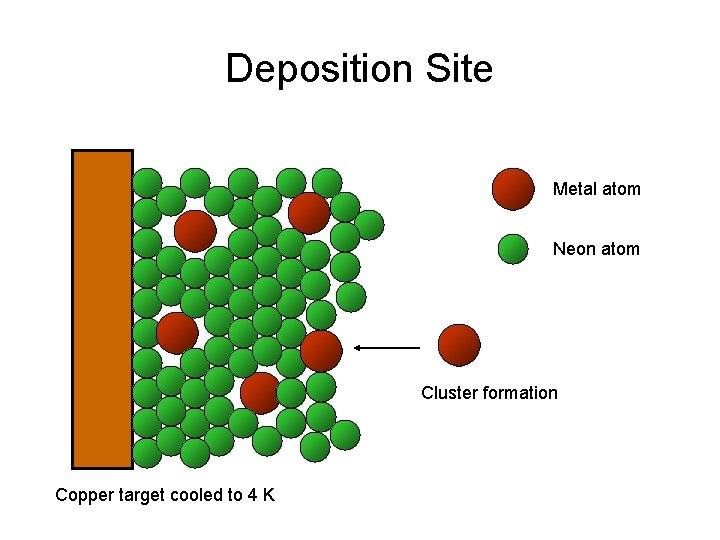
Deposition Site Metal atom Neon atom Cluster formation Copper target cooled to 4 K

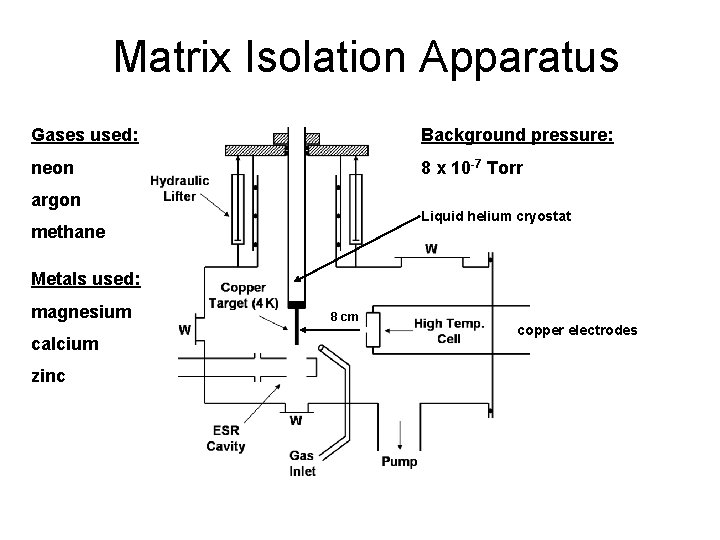
Matrix Isolation Apparatus Gases used: Background pressure: neon 8 x 10 -7 Torr argon Liquid helium cryostat methane Metals used: magnesium calcium zinc 8 cm copper electrodes

Magnesium and Calcium Experiments Conditions Deposited in: Deposit length: neon 1 hour Mg deposits argon Dilute: grayish-red methane Medium: dark red Heavy; dark gray Flow rates: Deposit temperatures: 2. 4 – 5. 0 sccm 4 – 40 K Ca deposits All: gray black Goal: Maximize high spin species

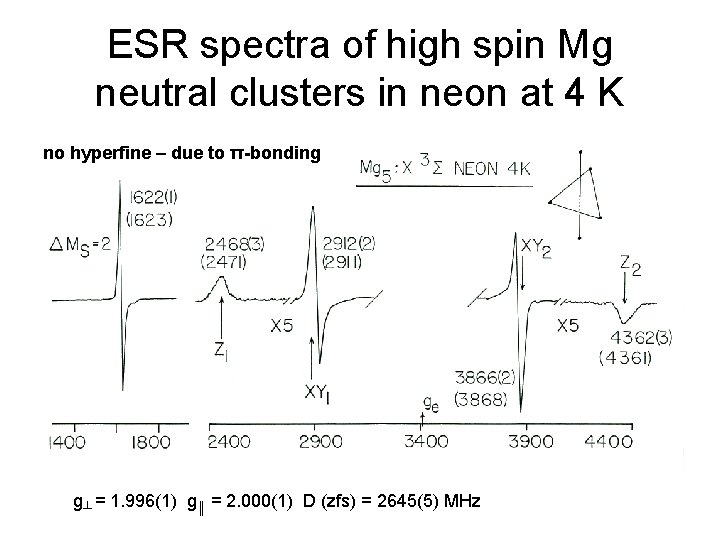
ESR spectra of high spin Mg neutral clusters in neon at 4 K no hyperfine – due to π-bonding g┴ = 1. 996(1) g║ = 2. 000(1) D (zfs) = 2645(5) MHz

ESR spectra of high spin Ca neutral clusters in neon at 4 K Signals only seen in neon

Zinc Experiments Conditions Deposited in: Deposit length: Zn deposits neon 1 hour Dilute: colorless/light orange Medium: dark orange Heavy; black Average flow rate: 4. 4 sccm Goal: control the size of clusters Deposit temperatures: X – irradiation: 4 – 40 K 30 minutes at 60 ke. V

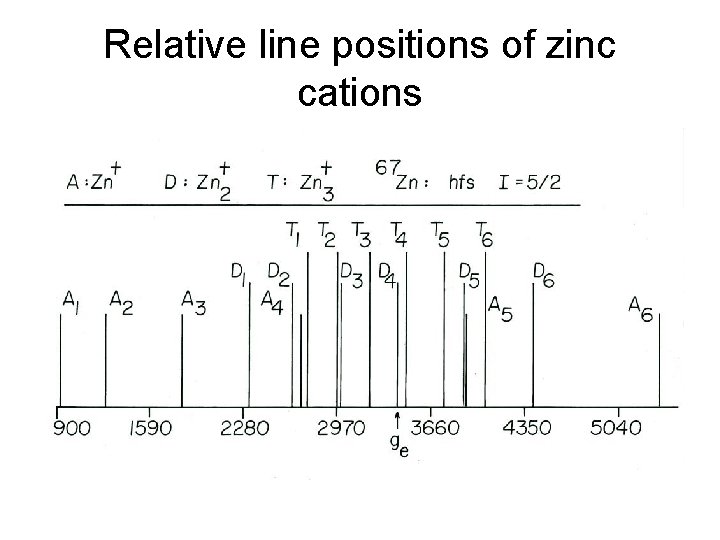
ESR spectra of zinc dimer and trimer cations in neon at 2. 6 K 67 Zn+ (4. 11 % abundance) Deposit temperature: 2. 6 K I = 5/2 Higher Zn concentration Lower Zn concentration

Magnetic parameters (MHz) of zinc monomer, dimer, and trimer, cation radicals

Relative line positions of zinc cations

Acknowledgments Thank you to: Dr. Knight TJ Grant Hendrickson