Electron Orbits Goal of the class To understand

Electron Orbits

Goal of the class • To understand the electron shells and movement of electrons • Question of the day: What is needed for an electron to change orbitals? • Previous answer: Carbon-12 has 6 neutrons, whereas carbon-14 has 8 (both have 6 protons)

Electron Shells • As an atom gains more electrons, the electrons interact with one another – Electrons have negative charge that will repel one another – This will force electrons further away from the nucleus – Electrons will go to other regions called shells or orbitals

Inner Shells • Each shell will fill up completely before going to further shells – Takes energy to go to other shells – Each electron will spread out evenly • A full shell of electrons is stable and won’t react
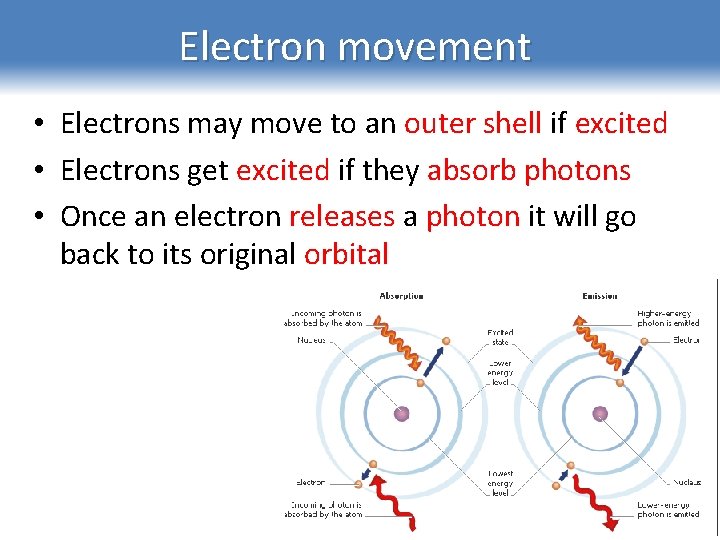
Electron movement • Electrons may move to an outer shell if excited • Electrons get excited if they absorb photons • Once an electron releases a photon it will go back to its original orbital

Valence electrons • The valence electrons are the electrons furthest from the nucleus – Most chemical reactions are interactions of the valence electrons – Valence electrons can be attracted to the protons of other atoms
Magic Numbers • The number allowed in each orbit is given by the numbers 2, 8, 8 • This corresponds to the number of atoms in each period.

Magic Numbers • In the element magnesium, the electronic configuration is given as (2, 8, 2). • How many valence electrons does magnesium have? • If a magnesium atom lost an electron, what would be the new configuration?
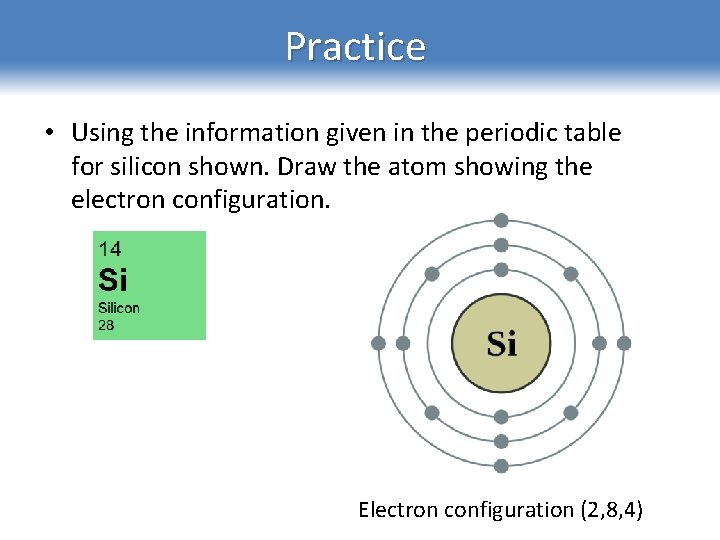
Practice • Using the information given in the periodic table for silicon shown. Draw the atom showing the electron configuration. Electron configuration (2, 8, 4)

Vocabulary • Discrete – separate, individual, noncontinuous. • Photon – a small discrete packet of light • Excite – to cause an electron to move higher than its normal state. To promote an electron to a higher level

Round Robin Game • We have now learnt a lot about the atom. • You will test your knowledge against your classmates. • Please sit in a team of 3.

Round Robin Game • Here are some sample answers: – – – – Atoms are made of protons, neutrons and electrons The atomic number is the same as the number of protons An atom is mostly empty space The charge on the electron is negative Protons are made of quarks Electrons exist in orbits All matter is made of atoms There are over 100 elements Neutrons keep the nucleus stable An electron is a fundamental particle The proton has a mass of 1 amu An atom is about 10 -10 m across Some atoms are unstable and will decay etc.

Homework • Play with http: //phet. colorado. edu/en/simulation/ build-an-atom
- Slides: 13