Electron Orbitals Cartoon courtesy of labinitio com Quantum
Electron Orbitals Cartoon courtesy of lab-initio. com

Quantum Mechanical Model of the Atom The model says that the nucleus is surrounded by a space in which electrons orbit- the electron cloud.

The model says that the electron cloud is divided the following parts: 1) The electron cloud is divided into energy levels (n) numbered 1, 2, 3, 4, … to infinity(∞) 2) Each energy level is divided into sublevels. (s, p, d, f) 3) Each sublevel is divided into orbitals. Regardless of which energy level it resides, sublevel s has 1 orbital. Sublevel p has 3 orbitals. Sublevel d has 5 orbitals. Sublevel f has 7 orbitals. (Note: the pattern continues to go up by odd numbers. ) 4) Each orbital can hold a maximum of 2 electrons with opposite spins.

1) Electron Energy Levels • “n” is also known as the Principle Quantum number • Formula to find the total number of electrons that can fit in an energy level: 2 n 2 n 1 is closest to the nucleus.

• The electron cloud is divided into energy levels (n) numbered 1, 2, 3, 4, … to infinity(∞) • n 1 is closest to the nucleus. • The farther out from the nucleus the more energy the level has.

2) Each energy level is divided into sublevels. -Level 1 has 1 sublevel (s). -Level 2 has 2 sublevels (s, p). - Level 3 has 3 sublevels (s, p, d). -Level 4 has 4 sublevels (s, p, d, f). Sublevels continue to be labeled with letters. After s, p, d, f they just go through the rest of the alphabet (g, h, i, etc. ).
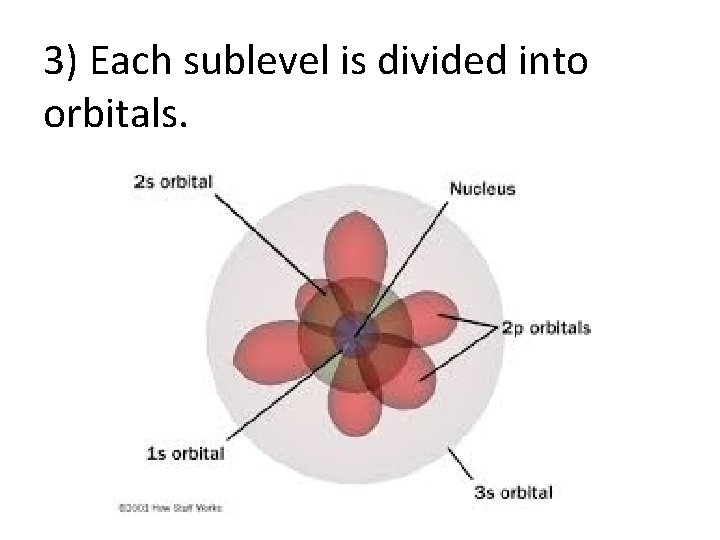
3) Each sublevel is divided into orbitals.

What is an Electron Orbital? An orbital is a region within an energy level where there is a probability of finding an electron. Orbital shapes are defined as the surface that contains 90% of the total electron probability.

Scientists can not draw the path the electrons take in the orbital because… To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. You can't do this for electrons.

Heisenberg Uncertainty Principle “One cannot simultaneously determine both the position and momentum of an electron. ” You can find out where the electron is, but not where it is going. Werner Heisenberg OR… You can find out where the electron is going, but not where it is!
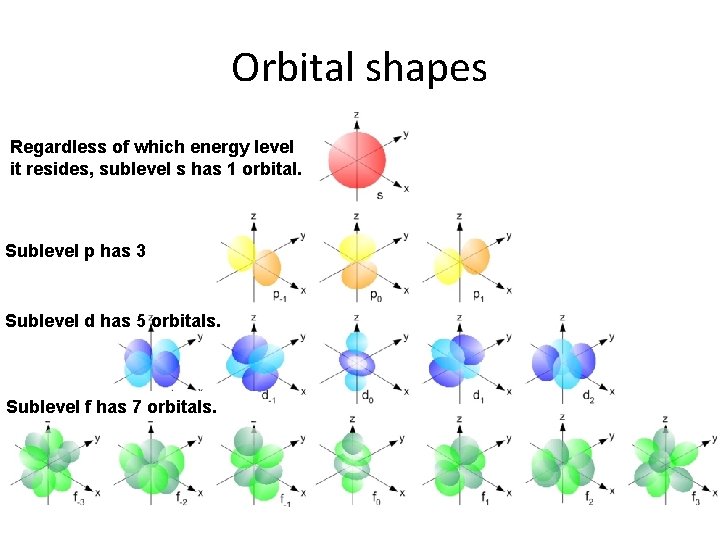
Orbital shapes Regardless of which energy level it resides, sublevel s has 1 orbital. Sublevel p has 3 orbitals. Sublevel d has 5 orbitals. Sublevel f has 7 orbitals.
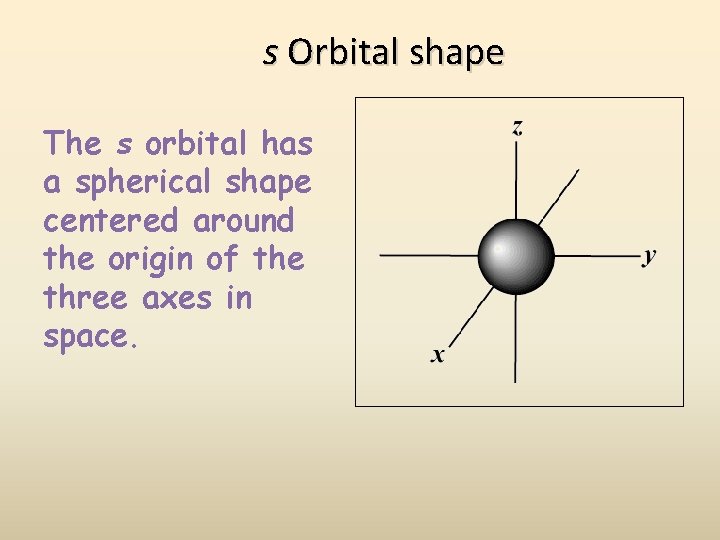
s Orbital shape The s orbital has a spherical shape centered around the origin of the three axes in space.
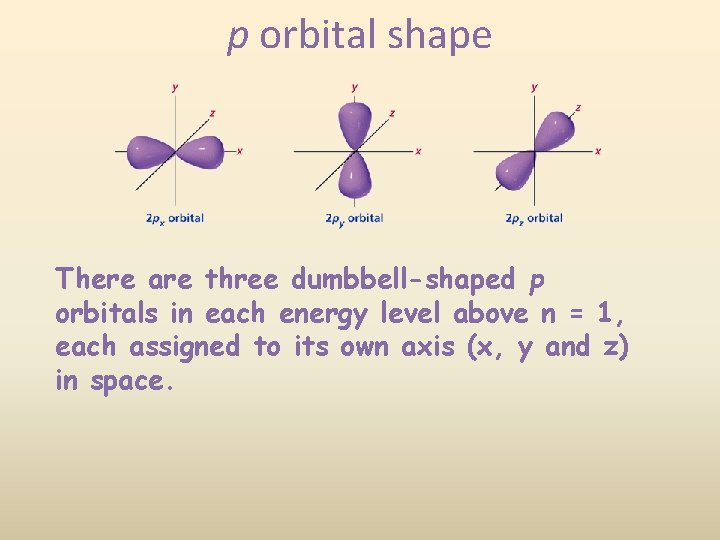
p orbital shape There are three dumbbell-shaped p orbitals in each energy level above n = 1, each assigned to its own axis (x, y and z) in space.

d orbital shapes Things get a bit more complicated with the five d orbitals that are found in the d sublevels beginning with n = 3. To remember the shapes, think of “double dumbbells” …and a “dumbbell with a donut”!
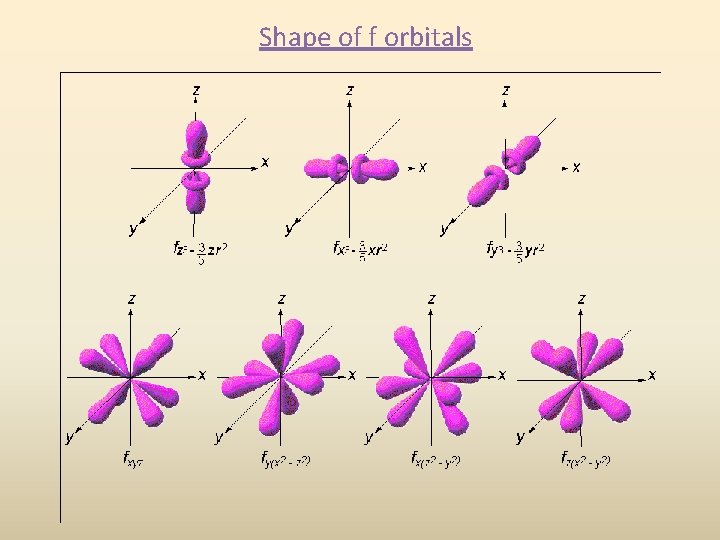
Shape of f orbitals

Electron Spin 4) Each orbital can hold a maximum of 2 electrons with opposite spins. Electron spin describes the behavior (direction of spin) of an electron within a magnetic field. Possibilities for electron spin: Forward, backward

Pauli Exclusion Principle Two electrons occupying the same orbital must have opposite spins Wolfgang Pauli

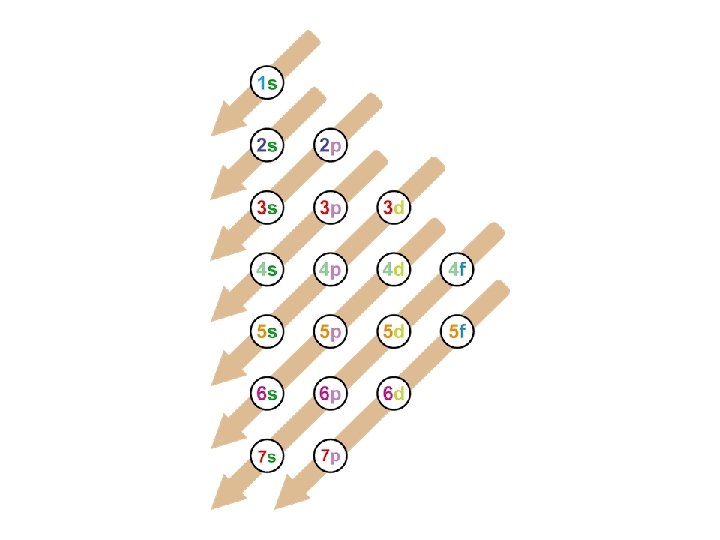
Principles of electron configurations: • Aufbau Principle: Electrons enter the orbital of the lowest energy level first. • Hund’s Rule: Electrons enter the orbital of equal energy and then pair up. • Pauli Exclusion Principle: Two electrons occupying the same orbital must have opposite spins • Let’s Practice!!!
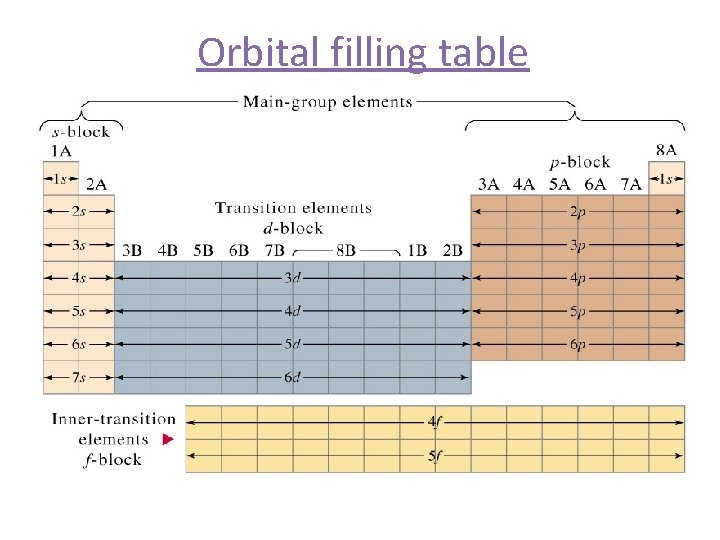
Orbital filling table

Electron configuration of the elements of the first three series
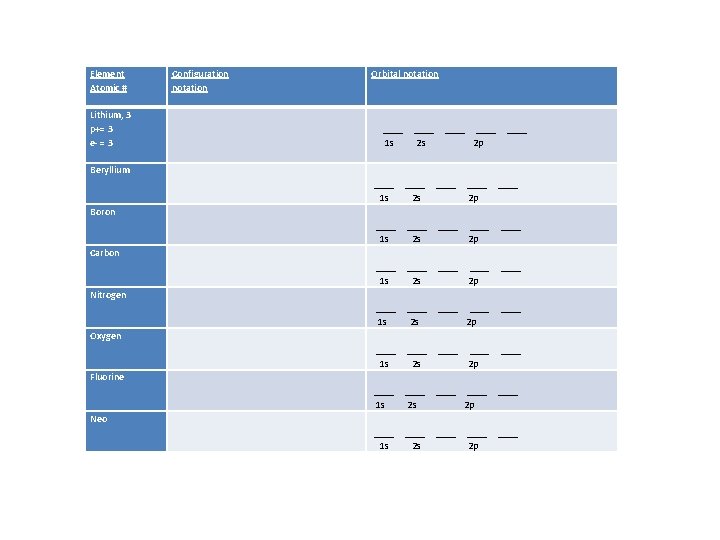
Element Atomic # Lithium, 3 p+= 3 e- = 3 Configuration notation Orbital notation ____ 1 s ____ 2 s ____ 2 p ____ Beryllium ____ 1 s ____ 2 s ____ ____ 2 p ____ ____ 1 s ____ 2 s ____ 2 p ____ 1 s ____ 2 s ____ 2 p ____ Boron Carbon Nitrogen Oxygen Fluorine Neo

Periodic Trends Blocks can be grouped according to the sublevels s block – Group 1 A ands 2 A p block – Group 3 A through 8 A d block – the transition metals/elements f block – the Inner Transition Metals

Practice • Then a few more things….


Examples Row number = period = energy level Hydrogen- row 1= period 1, block 1 s H-1 s 1 Beryllium – row 2 =period 2, block 2 s Be- 1 s 2 2 s 2 Carbon – row 2 =period 2, block 2 p C- 1 s 2 2 p 2


The Exceptions! 1. Transition elements (d sublevel)The energy level is 1 less than the row/period # Ex: Sc- period 4 d but it will end in 3 d! Sc- 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 1 2. Inner transition metals (f sublevel)The energy level is 2 less than the row /period # Ex: Dy- period 6 f but it will end in 4 f! Dy- 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 P 6 5 s 2 4 d 10 5 p 6 6 s 2 4 f 9
- Slides: 28