Electron energy levels have sublevels of different shapes
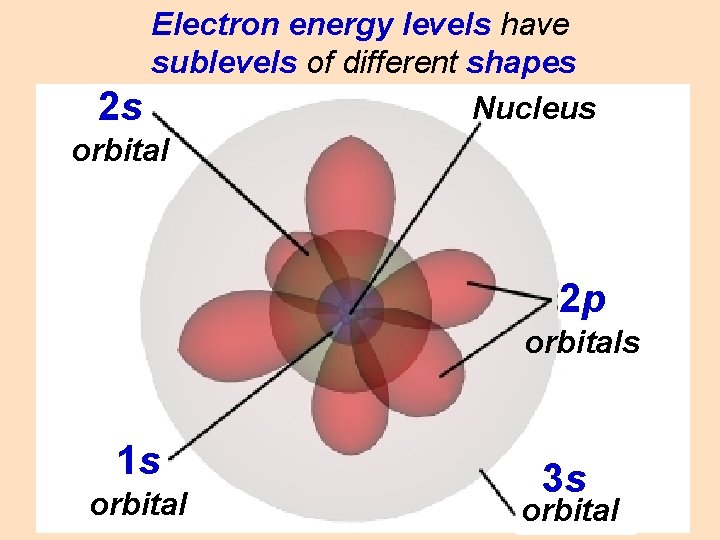
Electron energy levels have sublevels of different shapes Nucleus 2 s orbital 2 p orbitals 1 s orbital 3 s orbital
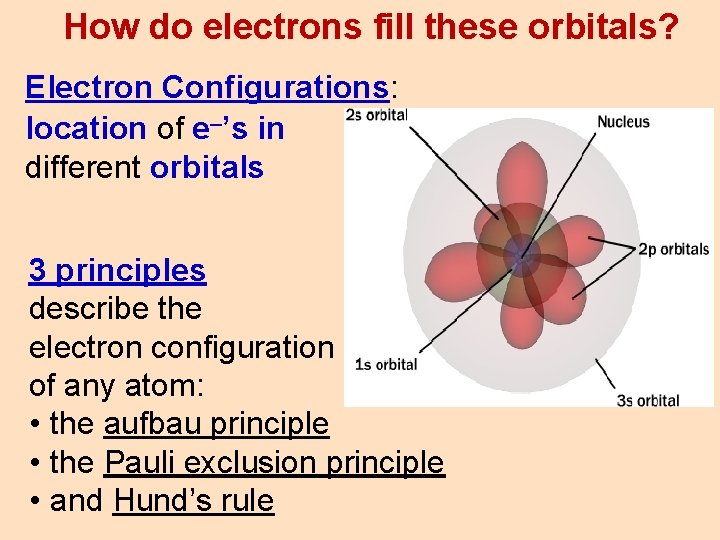
How do electrons fill these orbitals? Electron Configurations: location of e–’s in different orbitals 3 principles describe the electron configuration of any atom: • the aufbau principle • the Pauli exclusion principle • and Hund’s rule
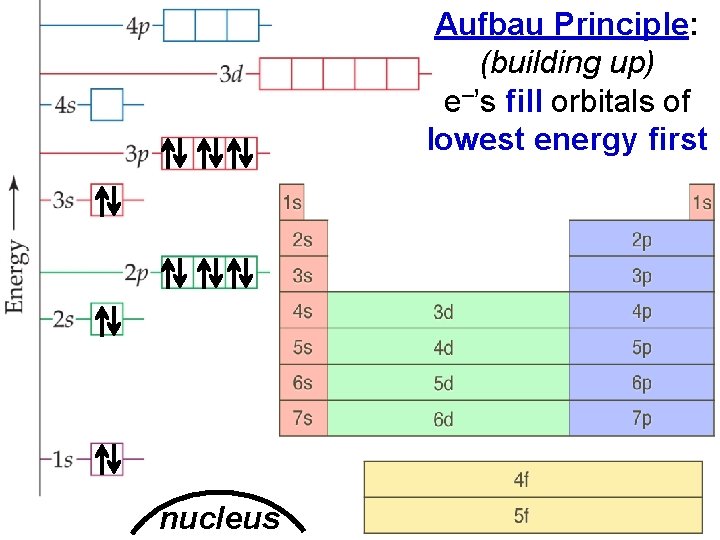
Electron Arrangement in Atoms nucleus > Aufbau Principle: (building up) e–’s fill orbitals of lowest energy first Slide 3 of 20 © Copyright Pearson Prentice Hall End Show
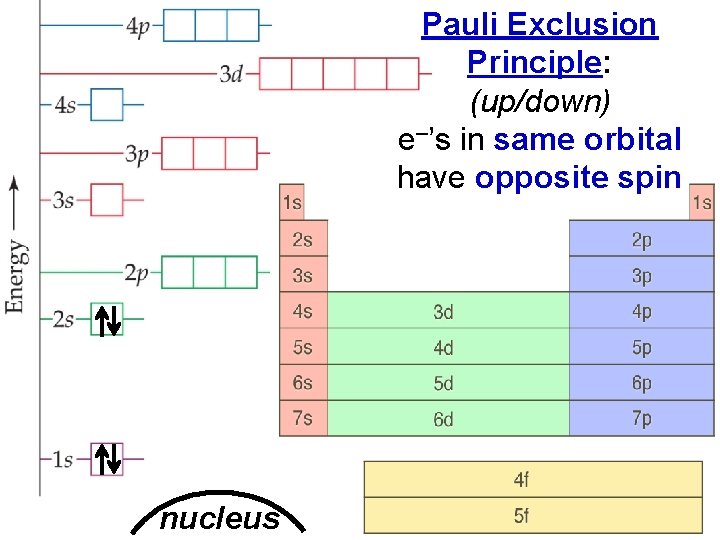
Electron Arrangement in Atoms nucleus > Pauli Exclusion Principle: (up/down) e–’s in same orbital have opposite spin Slide 4 of 20 © Copyright Pearson Prentice Hall End Show
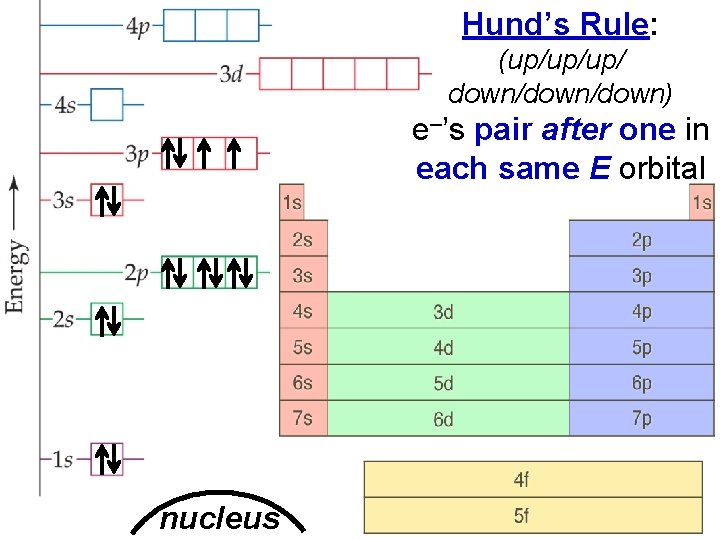
Electron Arrangement in Atoms Hund’s Rule: > (up/up/up/ down/down) e–’s pair after one in each same E orbital nucleus Slide 5 of 20 © Copyright Pearson Prentice Hall End Show
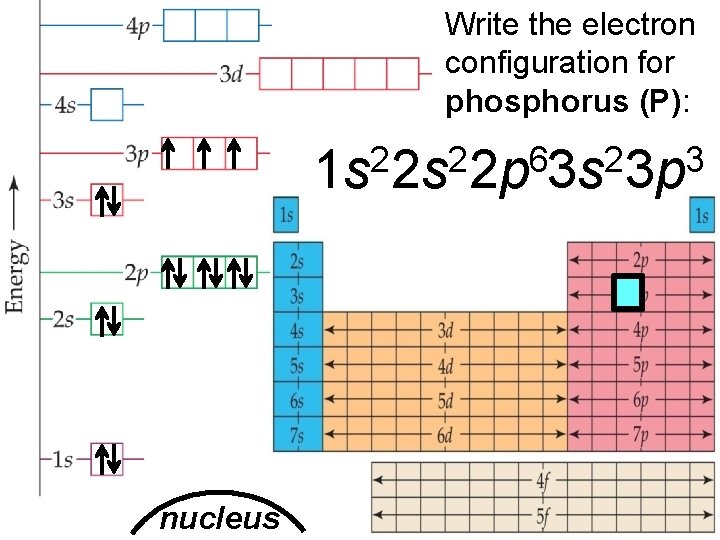
Write the electron configuration for phosphorus (P): 2 2 6 2 3 1 s 2 s 2 p 3 s 3 p nucleus Slide 6 of 20 © Copyright Pearson Prentice Hall End Show
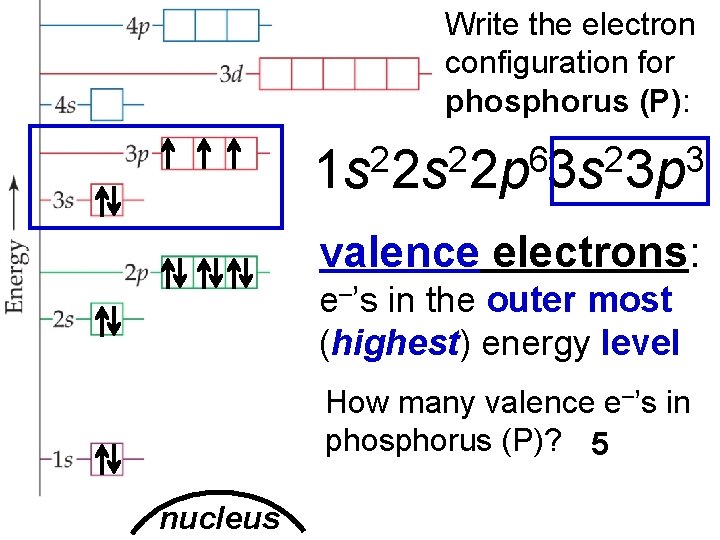
Write the electron configuration for phosphorus (P): 2 2 6 2 3 1 s 2 s 2 p 3 s 3 p valence electrons: e–’s in the outer most (highest) energy level How many valence e–’s in phosphorus (P)? 5 nucleus Slide 7 of 20 © Copyright Pearson Prentice Hall End Show
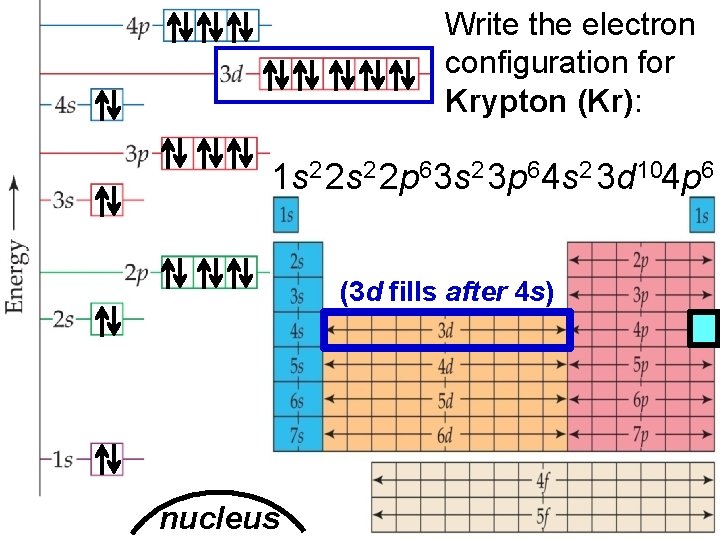
Electron Arrangement in Atoms > Write the electron configuration for Krypton (Kr): 1 s 2 2 p 63 s 2 3 p 64 s 2 3 d 104 p 6 (3 d fills after 4 s) nucleus Slide 8 of 20 © Copyright Pearson Prentice Hall End Show
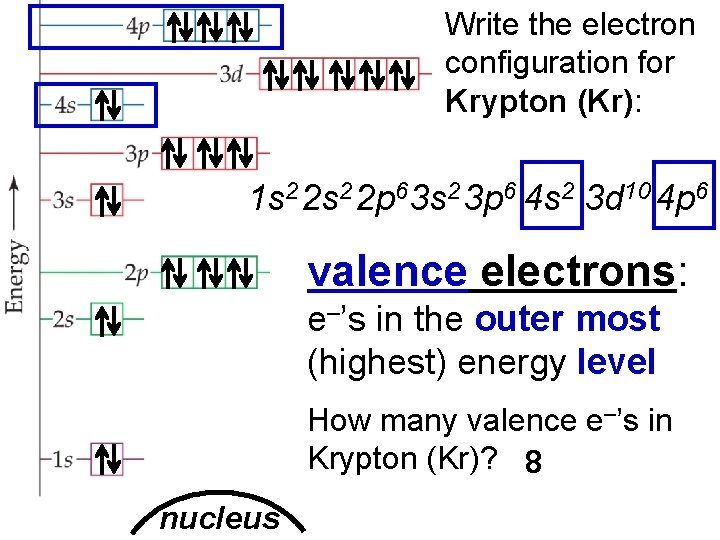
Electron Arrangement in Atoms > Write the electron configuration for Krypton (Kr): 1 s 2 2 p 63 s 2 3 p 6 4 s 2 3 d 10 4 p 6 valence electrons: e–’s in the outer most (highest) energy level How many valence e–’s in Krypton (Kr)? 8 nucleus © Copyright Pearson Prentice Hall Slide 9 of 20 End Show
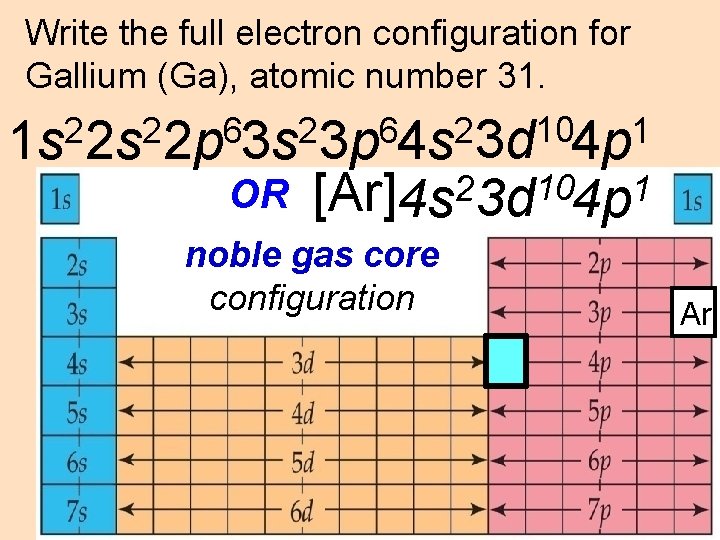
Write the full electron configuration for Gallium (Ga), atomic number 31. 10 2 2 6 2 1 1 s 2 s 2 p 3 s 3 p 4 s 3 d 4 p OR [Ar]4 s 23 d 104 p 1 noble gas core configuration Ar

valence e–’s: e–’s in highest energy level Lewis dot diagram: valence e–’s as dots Phosphorus (P): Bromine (Br): 1 s 2 2 p 6 3 s 2 3 p 3 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 5 [Ne]3 s 2 3 p 3 [Ar]4 s 2 3 d 10 4 p 5 # valence e–’s: ___ 7 Group # : ___ 5 A Group # : ___ 7 A Lewis dot diagram: P Lewis dot diagram: Br
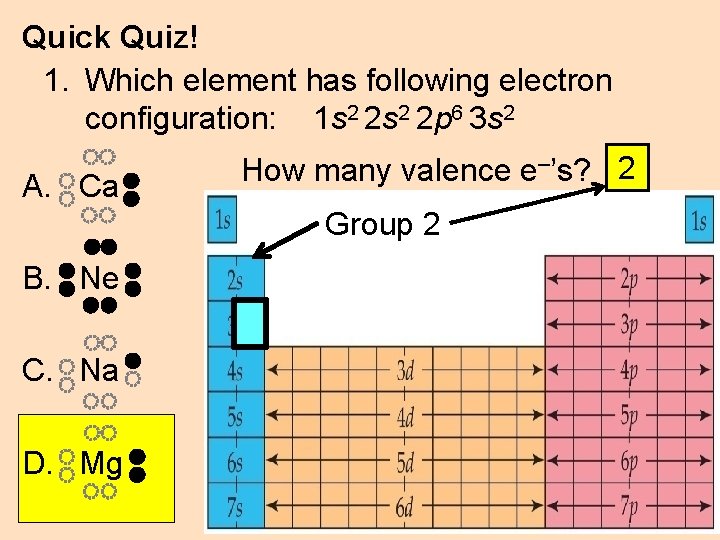
Quick Quiz! 1. Which element has following electron configuration: 1 s 2 2 p 6 3 s 2 A. Ca How many valence e–’s? 2 Group 2 B. Ne C. Na D. Mg
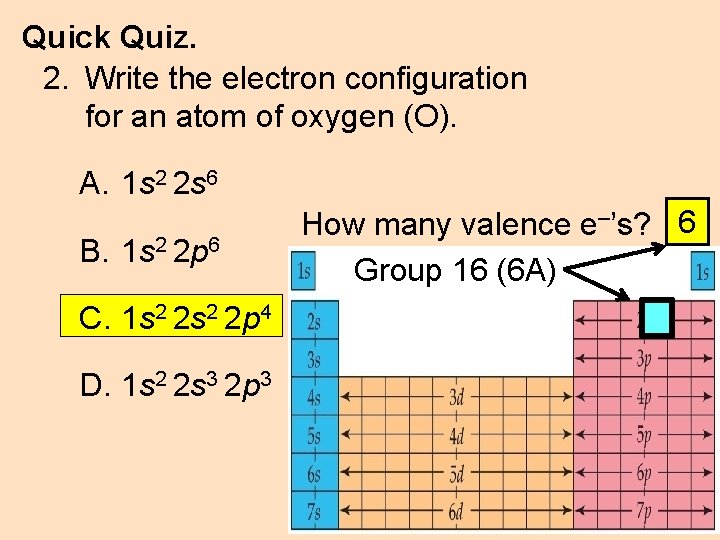
Quick Quiz. 2. Write the electron configuration for an atom of oxygen (O). A. 1 s 2 2 s 6 B. 1 s 2 2 p 6 C. 1 s 2 2 p 4 D. 1 s 2 2 s 3 2 p 3 How many valence e–’s? 6 Group 16 (6 A)
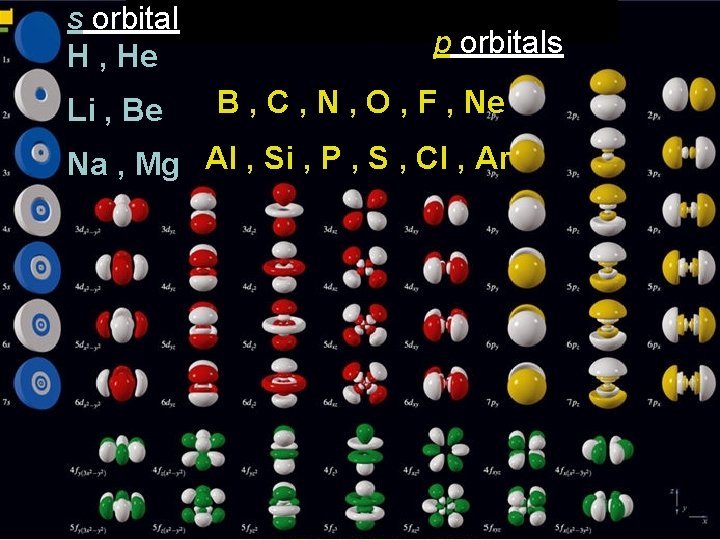
s orbital H , He Li , Be p orbitals B , C , N , O , F , Ne Na , Mg Al , Si , P , S , Cl , Ar
- Slides: 14