Electron Configurations SECTION 5 3 Electron Configurations Atom












![Noble Gas Notation 2 14 9 [Xe]6 s 4 f 5 d Check: (Xe’s Noble Gas Notation 2 14 9 [Xe]6 s 4 f 5 d Check: (Xe’s](https://slidetodoc.com/presentation_image_h2/2b6e8f5bdbf6130f0dd3129efdbdb082/image-15.jpg)









- Slides: 26

Electron Configurations SECTION 5. 3

Electron Configurations Atom is in the ground state 3 rules or laws �Aufbau principle �Pauli Exclusion principle �Hund’s Law

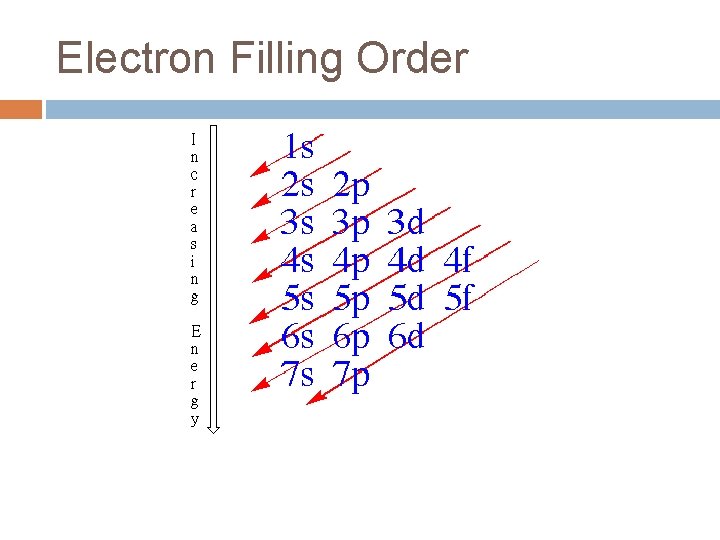
Aufbau Principle Electrons occupy lowest energy orbital available �All orbitals of a sublevel are equal energy �Sublevels have different energies s < p < d <f �Orbitals within one principal energy level can overlap orbitals of another

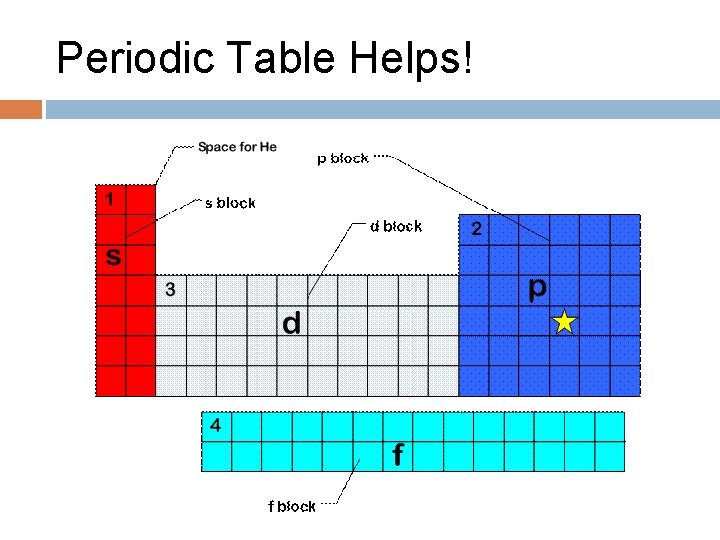
Electron Filling Order Which orbital will first, 4 s or 3 d?

Periodic Table Helps!

Pauli Exclusion Principle �Maximum of 2 electrons in an orbital, but only if they have opposite spin.

Hund’s rule �Electrons with same spin must occupy each equal energy orbital before additional electrons with opposite spins can occupy the same orbital.

Orbital Filling Order 1 s 2 s 2 p 3 s 3 p 4 s 3 d 4 p 5 s 4 d 5 p 6 s 4 f 5 d 6 p 7 s 5 f 6 d 7 p

Electron Configurations Three methods 1. Electron Configuration Notation 2. Noble Gas Notation 3. Orbital Diagrams

Electron Configuration Notation Ex: N �# of electrons? Start with the lowest energy, fill to capacity, go to next lowest energy, etc. Stop when you run out of electrons. �Superscripts = total electrons

Electron Configuration Notation You try … Zinc

Electron Configuration Notation 2 2 6 2 10 1 s 2 s 2 p 3 s 3 p 4 s 3 d

Assignment Write the electron configuration notation for the following elements: �Boron (B), Neon (Ne), Sulfur (S), Magnesium (Mg), Vanadium (V), Silver (Ag)

Noble Gas Notation Shorthand �Find the noble gas closest to the element (without going over). �Ex: For Gold (79): Xenon (54) �We write [Xe] and start counting from there �The first orbital after xenon is 6 s
![Noble Gas Notation 2 14 9 Xe6 s 4 f 5 d Check Xes Noble Gas Notation 2 14 9 [Xe]6 s 4 f 5 d Check: (Xe’s](https://slidetodoc.com/presentation_image_h2/2b6e8f5bdbf6130f0dd3129efdbdb082/image-15.jpg)
Noble Gas Notation 2 14 9 [Xe]6 s 4 f 5 d Check: (Xe’s atomic number) 54 + 2 + 14 + 9 = 79 (gold’s Atomic number)!

You Try… Silver

Assignment P. 147 #79

Orbital Diagrams Draw a line for each orbital Designate each electron with an arrow = orbital with 1 electron = orbital with 2 electrons

Orbital Diagrams Nitrogen: 1 s 22 p 3 � Write notation of N � Then we fill them with electrons using the Aufbau principle, Pauli exclusion principle, and Hund’s law

Orbital Diagrams You try another example: Cu

Orbital Diagrams Answer

Assignment Draw orbital diagrams for beryllium, aluminum, nitrogen, and sodium, manganese, germanium, and europium.

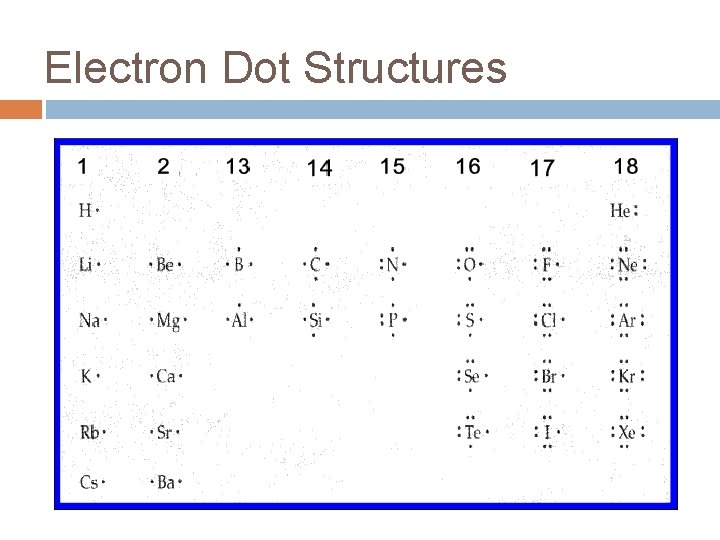
Electron Dot Structures Valence electrons �Electrons in outermost energy level. �Responsible for chemical properties

Electron Dot Structures Valence electrons only Place ‘dots’ around element symbol 4 sides of element = orbitals Fill these orbitals one at a time (Hund’s)

Electron Dot Structures

Assignment P. 141 #23(yellow box), 26, 28 P. 147 #81