ELECTRON CONFIGURATION What is a distinguishing characteristic of









- Slides: 16

ELECTRON CONFIGURATION What is a distinguishing characteristic of each and every person? Fingerprints - every person has their own fingerprint DNA - every person has their own DNA signature Each element has it's own unique electron configuration They tell us where in an atom the LAST electron is located, and where each element "lives" on the periodic table.

Suppose you want to send a letter to a friend somewhere in the United States Aside from the Zip Code -- what will the post office look at first? After the state, what is the next thing the post office will need? After the city? After the street name? This is similar to an electron in an atom. The electron configuration is the electron's address in the atom.

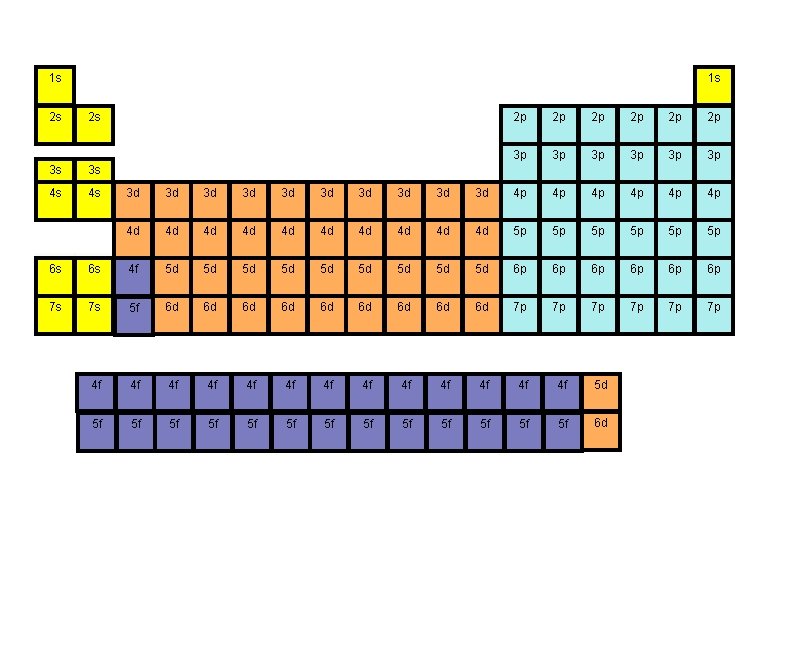
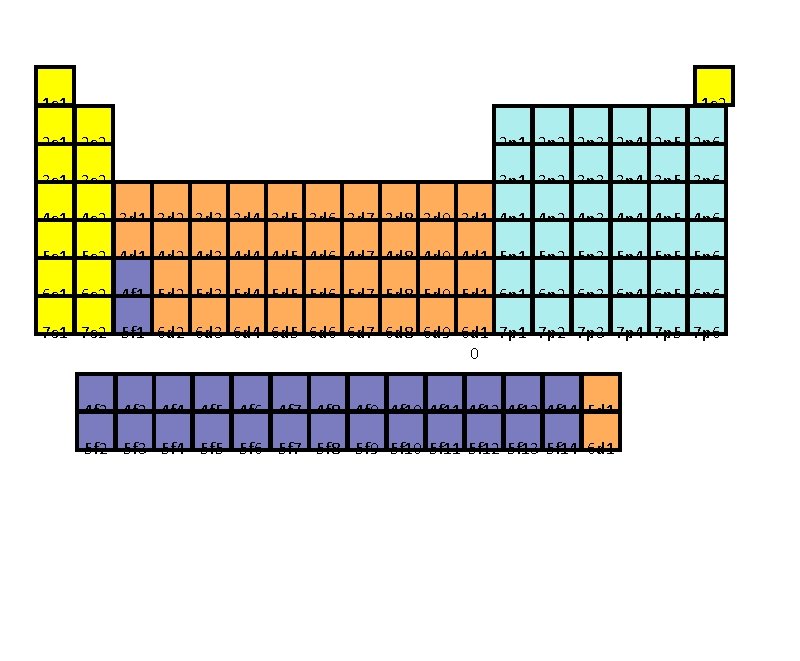
The Basics - where is the electron located? 1. The Energy Level ( it is similar to the state when you mail a letter) # 1 - 7: 7 rows on the periodic table 2. The Sublevel (it is similar to the city) s, p, d, f : these are the letters used to represent sublevels Some "states" are larger than others so they can have more "cities. " Likewise, the larger the energy level the more sublevels they can contain. Energy level 1 can only have an s sublevel Energy level 2 can only have an s & p sublevel Energy level 3 can only have an s, p & d sublevel Energy level 4 - 7 can have s, p, d & f sublevels

3. The Orbital (is similar to the street) Some "cities" have more "streets" than others s has one p has three d has five f has seven 4. The Spin (number) All orbitals can hold 2 electrons and they must be of opposite spin OR

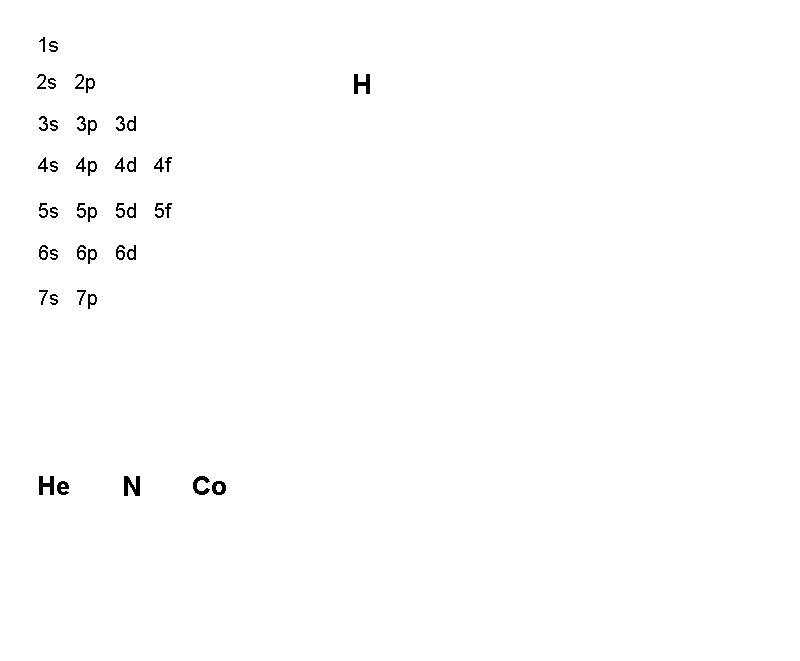
1 s H 2 s 2 p 3 s 3 p 3 d 4 s 4 p 4 d 4 f 5 s 5 p 5 d 5 f 6 s 6 p 6 d 7 s 7 p He N Co

1 s 2 s 2 p 3 s 3 p 3 d 4 s 4 p 4 d 4 f 5 s 5 p 5 d 5 f 6 s 6 p 6 d 7 s 7 p Homework


1 s 2 s 3 s 3 s 4 s 4 s 2 p 2 p 2 p 3 p 3 p 3 p 3 d 3 d 3 d 4 p 4 p 4 p 4 d 4 d 4 d 5 p 5 p 5 p 5 s 6 s 4 f 5 d 5 d 5 d 6 p 6 p 6 p 7 s 7 s 5 f 6 d 6 d 6 d 7 p 7 p 7 p 4 f 4 f 4 f 4 f 5 d 5 f 5 f 5 f 5 f 6 d

Electron Configuration 1 s 2 s 2 p Shorthand form of Orbital Fill Diagrams 3 s 3 p 3 d The only difference: 4 s 4 p 4 d 4 f Orbital Fill uses orbitals and half arrows 5 s 5 p 5 d 5 f 1 s 6 s 6 p 6 d 7 s 7 p Electron Configuration uses exponents 1 s 2 Example: Nitrogen Orbital Fill Diagram Electron Configuration 1 s 2 s 2 p 1 s 2 2 p 3

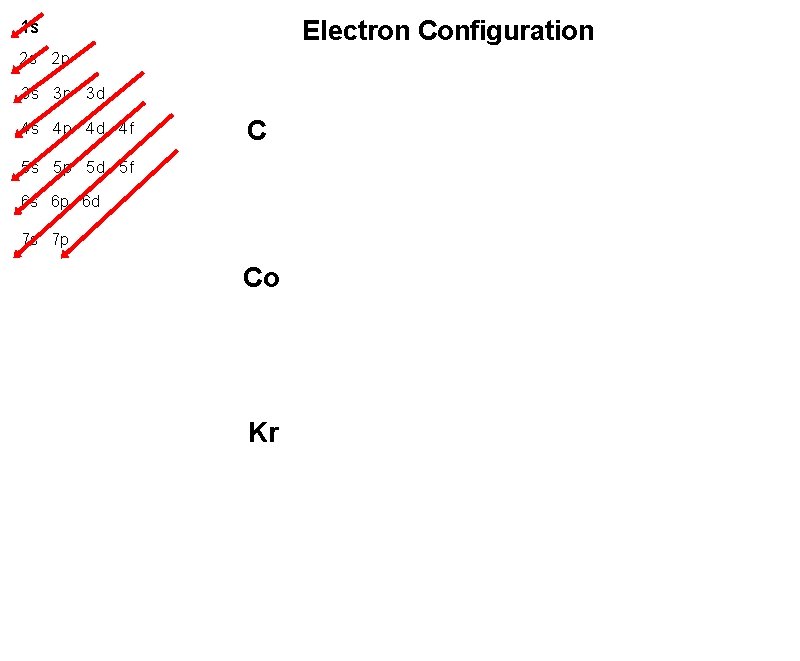
Electron Configuration 1 s 2 s 2 p 3 s 3 p 3 d 4 s 4 p 4 d 4 f C 5 s 5 p 5 d 5 f 6 s 6 p 6 d 7 s 7 p Co Kr

1 s 2 s 2 p 3 s 3 p 3 d 4 s 4 p 4 d 4 f 5 s 5 p 5 d 5 f 6 s 6 p 6 d 7 s 7 p Homework

1 s 1 1 s 2 2 s 1 2 s 2 2 p 1 2 p 2 2 p 3 2 p 4 2 p 5 2 p 6 3 s 1 3 s 2 3 p 1 3 p 2 3 p 3 3 p 4 3 p 5 3 p 6 4 s 1 4 s 2 3 d 1 3 d 2 3 d 3 3 d 4 3 d 5 3 d 6 3 d 7 3 d 8 3 d 9 3 d 1 0 5 s 1 5 s 2 4 d 1 4 d 2 4 d 3 4 d 4 4 d 5 4 d 6 4 d 7 4 d 8 4 d 9 4 d 1 0 6 s 1 6 s 2 4 f 1 5 d 2 5 d 3 5 d 4 5 d 5 5 d 6 5 d 7 5 d 8 5 d 9 5 d 1 0 7 s 1 7 s 2 5 f 1 6 d 2 6 d 3 6 d 4 6 d 5 6 d 6 6 d 7 6 d 8 6 d 9 6 d 1 0 4 p 1 4 p 2 4 p 3 4 p 4 4 p 5 4 p 6 5 p 1 5 p 2 5 p 3 5 p 4 5 p 5 5 p 6 6 p 1 6 p 2 6 p 3 6 p 4 6 p 5 6 p 6 7 p 1 7 p 2 7 p 3 7 p 4 7 p 5 7 p 6 4 f 2 4 f 3 4 f 4 4 f 5 4 f 6 4 f 7 4 f 8 4 f 9 4 f 10 4 f 11 4 f 12 4 f 13 4 f 14 5 d 1 5 f 2 5 f 3 5 f 4 5 f 5 5 f 6 5 f 7 5 f 8 5 f 9 5 f 10 5 f 11 5 f 12 5 f 13 5 f 14 6 d 1

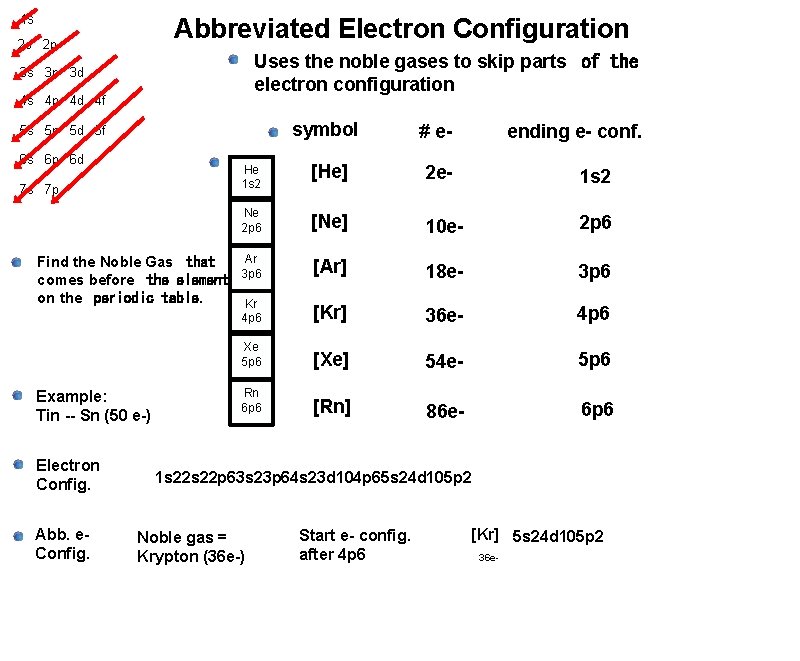
1 s Abbreviated Electron Configuration 2 s 2 p Uses the noble gases to skip parts of the electron configuration 3 s 3 p 3 d 4 s 4 p 4 d 4 f symbol # e- He 1 s 2 [He] 2 e- 1 s 2 Ne 2 p 6 [Ne] 10 e- 2 p 6 Ar 3 p 6 [Ar] 18 e- 3 p 6 Kr 4 p 6 [Kr] 36 e- 4 p 6 Xe 5 p 6 [Xe] 54 e- 5 p 6 Rn 6 p 6 [Rn] 86 e- 6 p 6 5 s 5 p 5 d 5 f 6 s 6 p 6 d 7 s 7 p Find the Noble Gas that comes before the element on the periodic table. Example: Tin -- Sn (50 e-) Electron Config. Abb. e. Config. ending e- conf. 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 2 Noble gas = Krypton (36 e-) Start e- config. after 4 p 6 [Kr] 5 s 24 d 105 p 2 36 e-

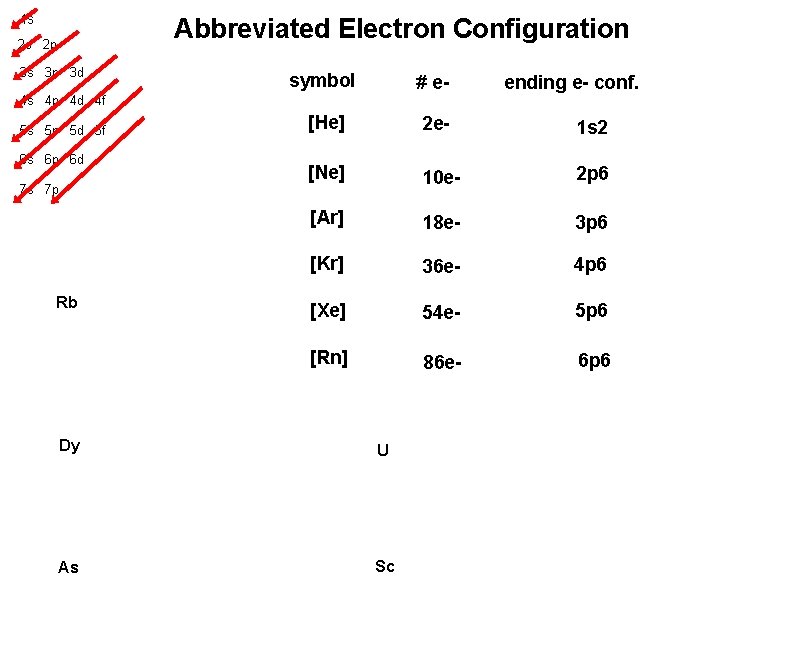
1 s Abbreviated Electron Configuration 2 s 2 p 3 s 3 p 3 d symbol # e- [He] 2 e- 1 s 2 [Ne] 10 e- 2 p 6 [Ar] 18 e- 3 p 6 [Kr] 36 e- 4 p 6 [Xe] 54 e- 5 p 6 [Rn] 86 e- 6 p 6 ending e- conf. 4 s 4 p 4 d 4 f 5 s 5 p 5 d 5 f 6 s 6 p 6 d 7 s 7 p Rb Dy U As Sc

1 s 2 s 2 p 3 s 3 p 3 d 4 s 4 p 4 d 4 f 5 s 5 p 5 d 5 f 6 s 6 p 6 d 7 s 7 p Homework
