Electron Configuration Heisenberg Uncertainty Principle States that it

Electron Configuration

Heisenberg Uncertainty Principle � States that it is fundamentally impossible to know precisely both the velocity and the position of a particle at the same time › It is inaccurate to assign fixed paths for electrons like the circular orbits in Bohr’s model › The only quantity that can be known is the probability for an electron to occupy a certain region around the nucleus

Quantum Mechanical Model � Similar to Bohr’s model, it limits an electron’s energy to certain values � Unlike Bohr’s model, it does not attempt to describe the electron’s path around the nucleus Quantum mechanical model- shows the allowed energies an electron can have and how likely it is to find the electron in various locations

Quantum Mechanical Model � The Schrodinger equation predicts a threedimensional region around the nucleus where there is a high probability of finding an electron › This region is called the atomic orbital

Describing Atomic Orbitals � Principle quantum number (n): indicates the relative size and energy of atomic orbitals �As n increases, orbital becomes larger, electron may be further from nucleus, atom’s energy increases �n specifies the atom’s main/principle energy level

Describing Atomic Orbitals � Principle energy levels contain energy sublevels › The # of sublevels in the principle energy level increases as n increases
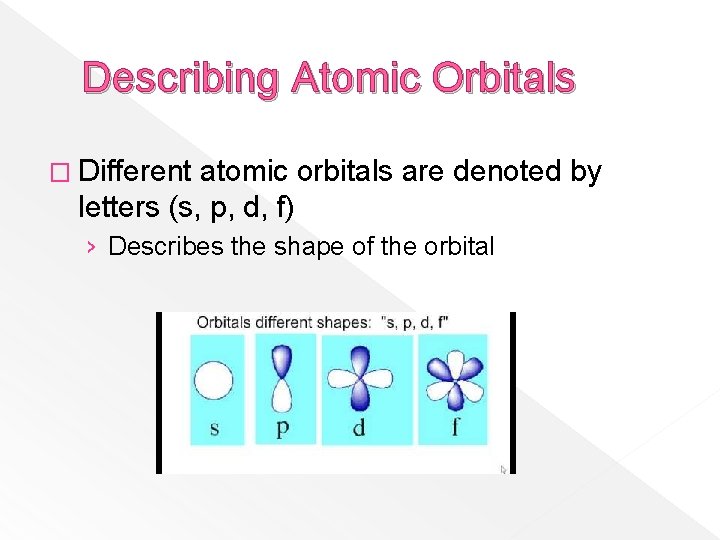
Describing Atomic Orbitals � Different atomic orbitals are denoted by letters (s, p, d, f) › Describes the shape of the orbital

Describing Atomic Orbitals � Each atomic orbital can hold 2 electrons › s orbital holds up to 2 electrons � 1 orbital (2 per orbital = 2 total) › p orbital set holds up to 6 electrons � 3 orbitals (2 x 3 = 6 total) › d orbital set holds up to 10 electrons � 5 orbitals (2 x 5 = 10 total) › f orbital set holds up to 14 electrons � 7 orbitals (2 x 7= 14 total)
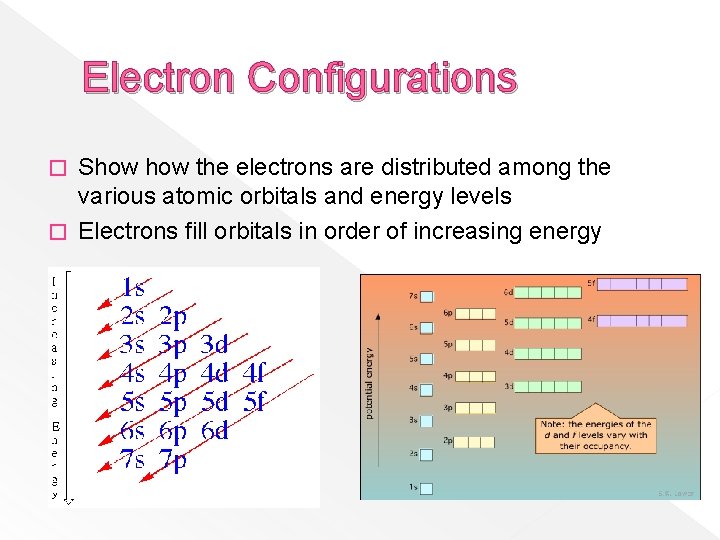
Electron Configurations Show the electrons are distributed among the various atomic orbitals and energy levels � Electrons fill orbitals in order of increasing energy �
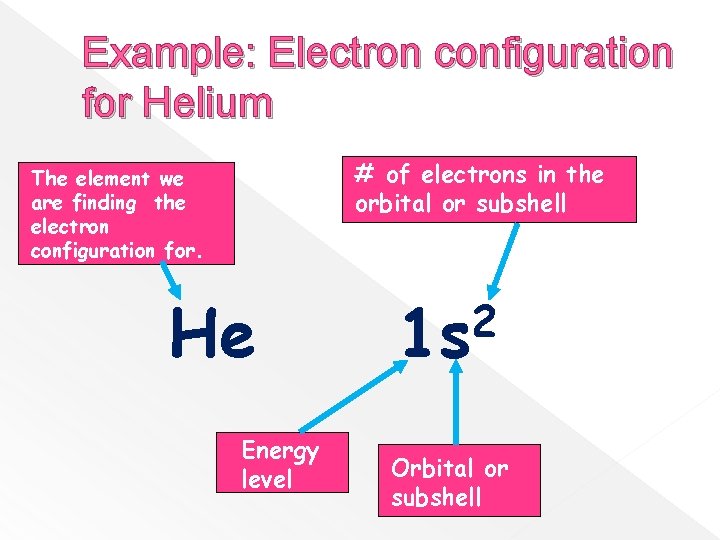
Example: Electron configuration for Helium # of electrons in the orbital or subshell The element we are finding the electron configuration for. He Energy level 2 1 s Orbital or subshell

Aufbau Principle � Electrons occupy orbitals of the lowest energy first

Aufbau Principle Example Boron: 1 s 22 p 1 � Correct › Boron: 1 s 2 s 2 p 3 s � Incorrect because did not fill the lowest energy orbital first › Boron: 1 s 2 s 2 p 3 s

Pauli Exclusion Principle �A maximum of 2 electrons can occupy an orbital at any one time and those two electrons must have opposite spins › An orbital holds two electrons that spin in opposite directions
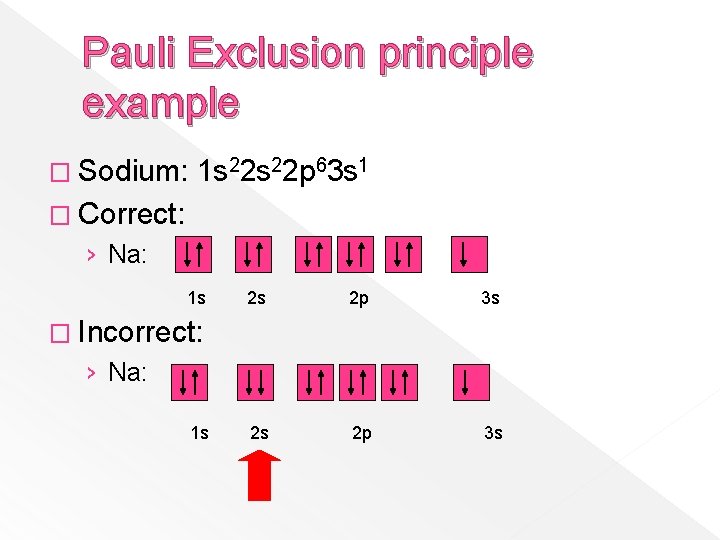
Pauli Exclusion principle example � Sodium: 1 s 22 p 63 s 1 � Correct: › Na: 1 s 2 s 2 p 3 s � Incorrect: › Na: 1 s

Hund’s Rule � When electrons fill orbitals of the same energy level, they do so in a way that maximizes the number of particles with the same spin. � Single electrons occupy available orbitals first before doubling up

Hund’s Rule in Action “Empty Seat Rule”
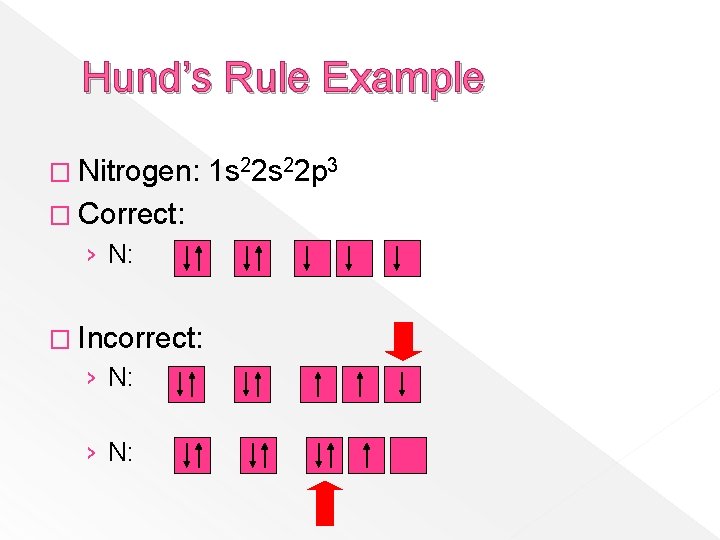
Hund’s Rule Example � Nitrogen: � Correct: › N: � Incorrect: › N: 1 s 22 p 3
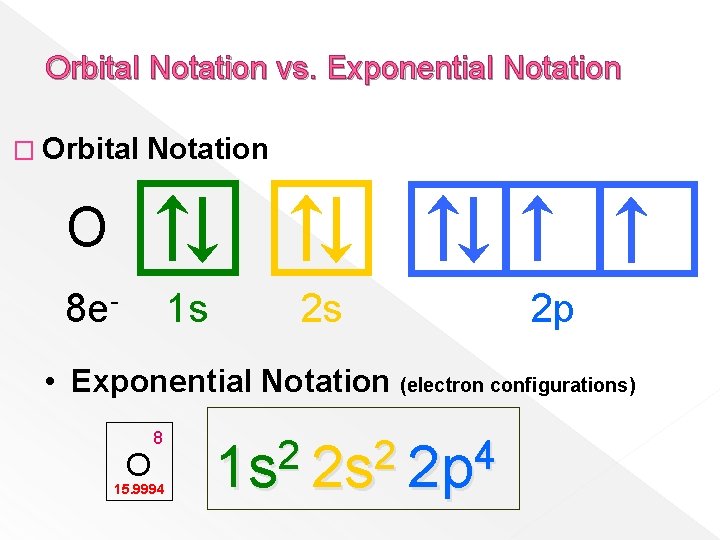
Orbital Notation vs. Exponential Notation � Orbital Notation O 8 e- 1 s 2 s 2 p • Exponential Notation (electron configurations) O 8 15. 9994 2 2 4 1 s 2 s 2 p
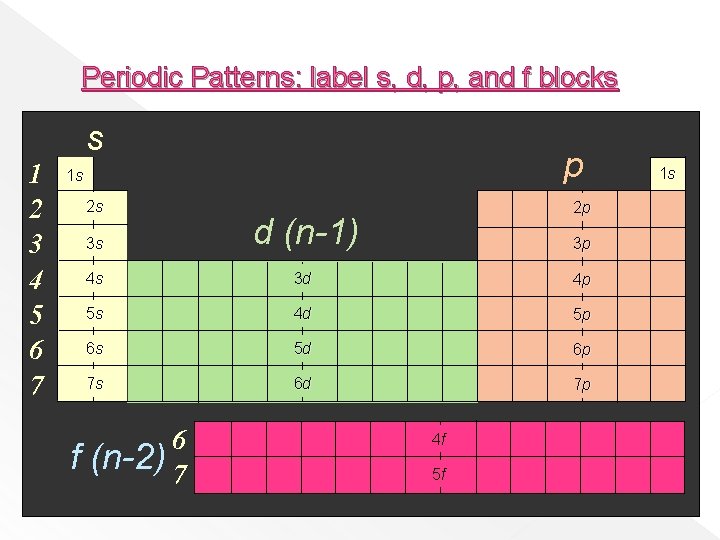
Periodic Patterns: label s, d, p, and f blocks s 1 2 3 4 5 6 7 p 1 s 2 s f 2 p 3 s d (n-1) 4 s 3 d 4 p 5 s 4 d 5 p 6 s 5 d 6 p 7 s 6 d 7 p 6 (n-2) 7 3 p 4 f 5 f 1 s
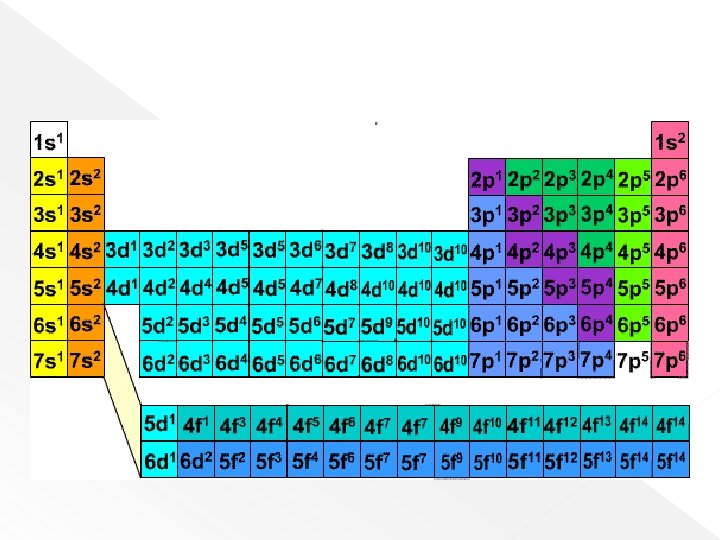
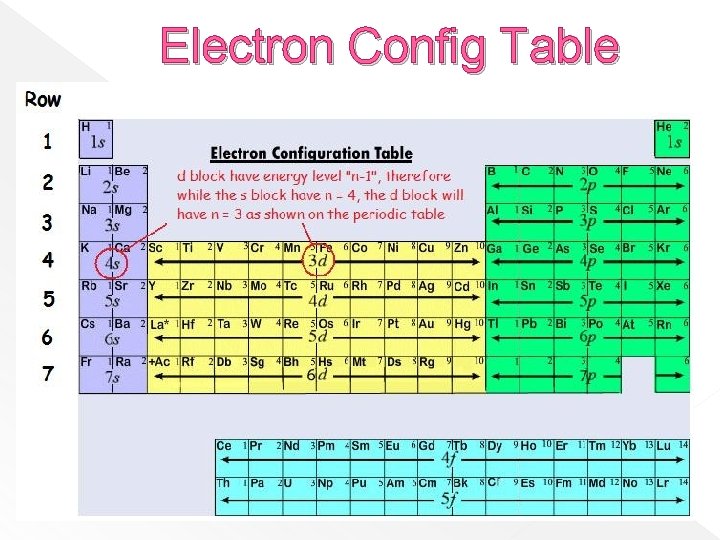
Electron Config Table

Writing shorthand electron configurations Write the symbol of the noble gas before the element in brackets 2. Then the rest of the electrons. 1. Example: � Aluminum - full configuration. � � 1 s 22 p 63 s 23 p 1 � Aluminum- short configuration: › [Ne] 3 s 23 p 1

Longhand VS Shorthand Electron Configuration � Longhand S 32. 066 Configuration 16 e 6 2 2 2 1 s 2 s 2 p 3 s Core Electrons 16 e 4 3 p Valence Electrons • Shorthand Configuration S S 2 4 [Ne] 3 s 3 p 16

Practice � Write longhand electron configurations: � Write shorthand electron configurations: 1. 2. 3. 4. 5. 6. 1. 2. 3. 4. H He Li N Cu Kr Ca Rb Cl Ag

Ions � Electrically charged atoms that have lost or gained electrons › Cation: positive ion �Lose electrons �Less electrons than protons �Metals › Anion: negative ion �Gain electrons �More electrons than protons �Nonmetals � Why do atoms lose or gain electrons?

Octet Rule � Atoms tend to gain, lose, or share electrons until they have eight valence electrons (full s and p orbitals) › This fills the valence shell and tends to give the atom the stability of the inert gasses � Ions form because the atom is trying to become more stable › They become isoelectronic with the noble gases �they have the same electron configuration
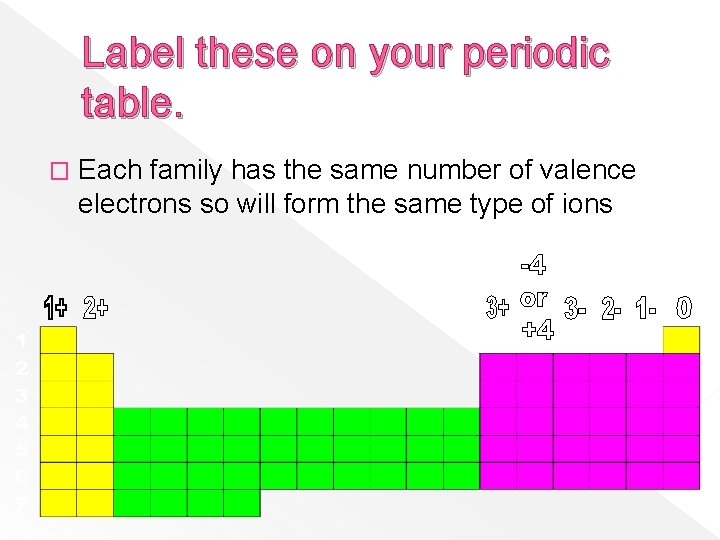
Label these on your periodic table. � Each family has the same number of valence electrons so will form the same type of ions

Ion Electron Configuration › Write the e- configuration for the closest Noble Gas �EX: Oxygen ion O 2 - Ne �Oxygen normally has 8 electrons, but if it becomes 2 - that means it gains two electrons so you must write an electron configuration with 10 electrons (8 + 2 = 10) O 2 - 10 e- [He] 2 s 2 2 p 6
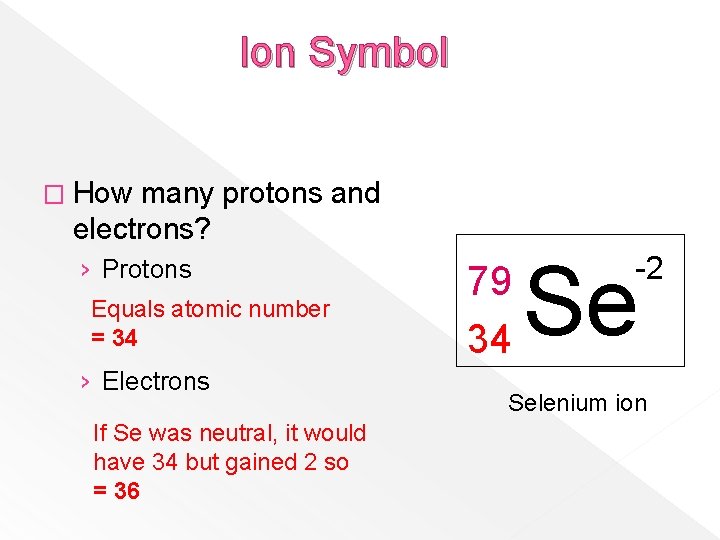
Ion Symbol � How many protons and electrons? › Protons Equals atomic number = 34 › Electrons If Se was neutral, it would have 34 but gained 2 so = 36 79 34 Se -2 Selenium ion

Ion practice › Aluminum (Al) �Electron configuration neutral Al: �How many valence electrons? �What type of ion will it form? �Ion symbol: ____ �How many total electrons in ion? �Electron configuration ion: › Sulfur (S) �Electron configuration neutral S: �How many valence electrons? �What type of ion will it form? �Ion symbol: ____ �How many total electrons in ion? �Electron configuration ion:
- Slides: 30