Electron Configuration from the Periodic Table Periodic Table

Electron Configuration from the Periodic Table
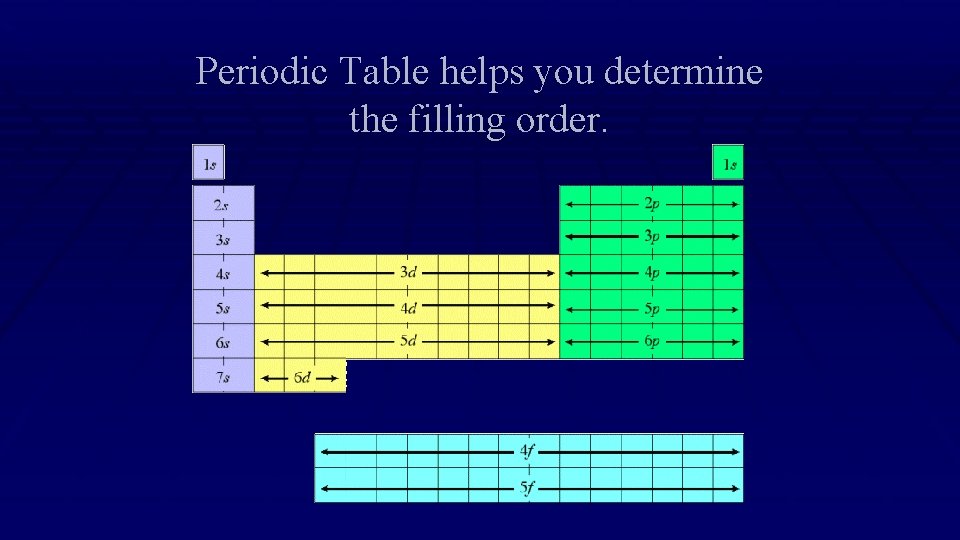
Periodic Table helps you determine the filling order.

Using The Periodic Table to Write Electron Configuration 1. Start at Hydrogen. This is 1 s 1. Then go to Helium, this is 1 s 2. 1. Each box counts as the next electron in that sublevel. 2. Once you reach the end of the sublevel, write this down on your paper. 3. Continue to follow the periodic table, writing down the configuration as you go. 4. Stop when you get to the element you are writing the electron configuration for.

Write the Electron Configurations 1. Si 2. Ar 3. Ca

Brain Break: Interval Countdown § § § § § 10 Air Squats 9 Bicep book/backpack curls 8 imaginary jump rope 7 overhead book/backpack extensions 6 star jumping jacks 5 alternating forward lunges (both legs=1 rep) 4 deep breaths 3 toe touches 2 shoulder rolls 1 deep sigh

Finding Valence Electrons from the Electron Configuration § The valence electrons are the electrons in the outermost energy level. 1. Find the highest energy level (The numbers) 2. Count the electrons in all sublevels (the letters) of this energy level. Example: 1 s 22 p 63 s 23 p 64 s 23 d 104 p 2 2 + 2 = 4 valence electrons

Example § How many valence electrons do the following elements have? § 1 s 22 p 63 s 23 p 64 s 23 d 9 § 1 s 22 p 2 § 1 s 22 p 63 s 23 p 64 s 23 d 104 p 4 § 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 4

Example: § How many core electrons do the following elements have? § 1 s 22 p 63 s 23 p 1 § 1 s 22 p 63 s 23 p 64 s 23 d 1 § 1 s 22 p 4 § 1 s 22 p 63 s 23 p 5

Example § What type of orbital are the valence electrons in for the following elements: § Na § Cu §O § Ce
- Slides: 9