ELECTRON CONFIGURATION ATOMIC MODELS Bohr Model Defined quantized

ELECTRON CONFIGURATION
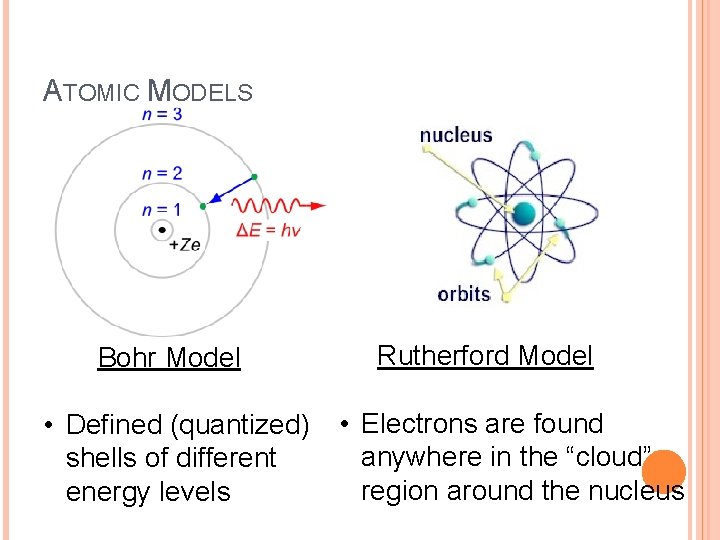
ATOMIC MODELS Bohr Model • Defined (quantized) shells of different energy levels Rutherford Model • Electrons are found anywhere in the “cloud” region around the nucleus
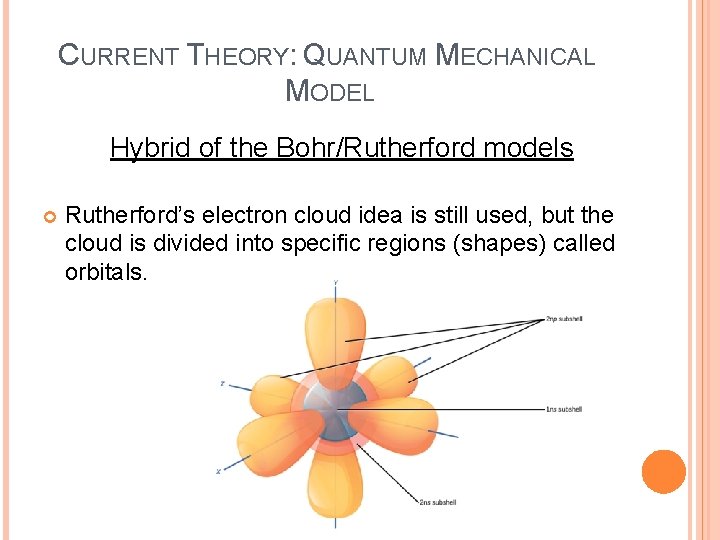
CURRENT THEORY: QUANTUM MECHANICAL MODEL Hybrid of the Bohr/Rutherford models Rutherford’s electron cloud idea is still used, but the cloud is divided into specific regions (shapes) called orbitals.
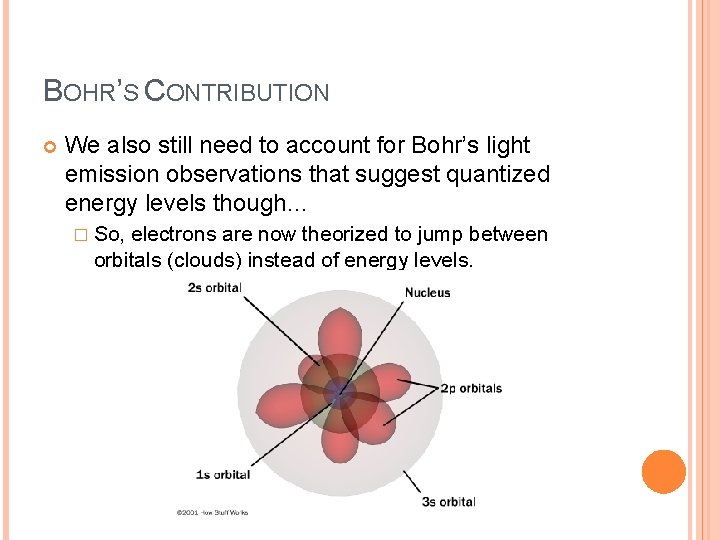
BOHR’S CONTRIBUTION We also still need to account for Bohr’s light emission observations that suggest quantized energy levels though… � So, electrons are now theorized to jump between orbitals (clouds) instead of energy levels.
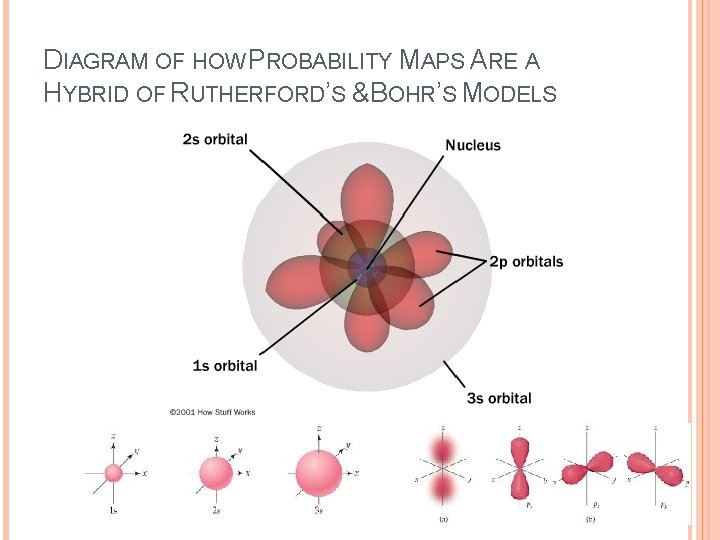
DIAGRAM OF HOW PROBABILITY MAPS ARE A HYBRID OF RUTHERFORD’S &BOHR’S MODELS

HOW DID WE MAP THE ELECTRON CLOUD SHAPES? The clouds represent probability distribution maps for electron positioning � We can specify the energy of an electron (emission), but not a well-defined location at any given instant (no uniform circular paths). � Instead, the electron’s position is described in terms of an orbital, a probability distribution map showing where the electron is likely to be found at any given moment.
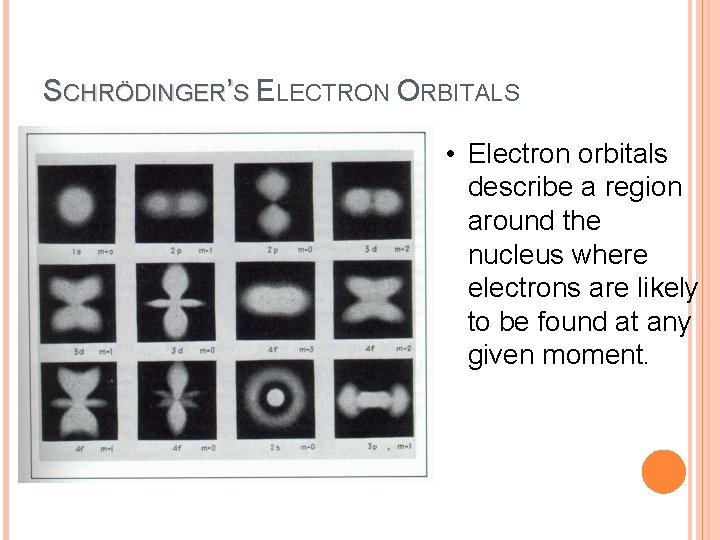
SCHRÖDINGER’S ELECTRON ORBITALS • Electron orbitals describe a region around the nucleus where electrons are likely to be found at any given moment.
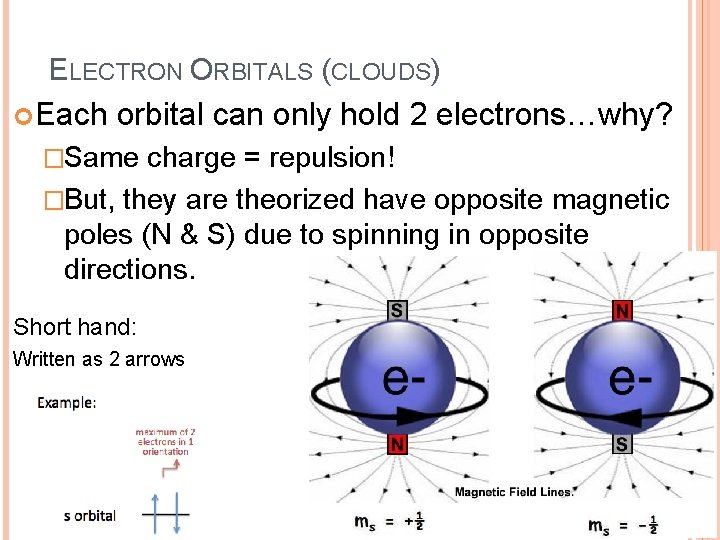
ELECTRON ORBITALS (CLOUDS) Each orbital can only hold 2 electrons…why? �Same charge = repulsion! �But, they are theorized have opposite magnetic poles (N & S) due to spinning in opposite directions. Short hand: Written as 2 arrows
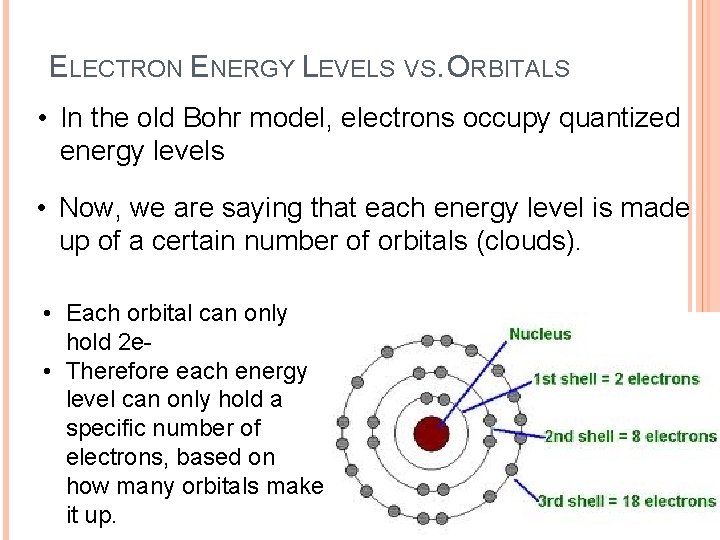
ELECTRON ENERGY LEVELS VS. ORBITALS • In the old Bohr model, electrons occupy quantized energy levels • Now, we are saying that each energy level is made up of a certain number of orbitals (clouds). • Each orbital can only hold 2 e • Therefore each energy level can only hold a specific number of electrons, based on how many orbitals make it up.

ELECTRON CONFIGURATION Within each energy level, there are specific orbitals that electrons occupy. Electron orbitals are categorized based on the energy level (shell 1, 2, 3, etc) and sublevel shape (s, p, d, f) • S-sublevels can hold 2 electrons (made of 1 orbital) • P-sublevels can hold 6 electrons (made of 3 orbitals) • D-sublevels can hold 10 electrons (made of 5 orbitals) • F-sublevels can hold 14 electrons (made of 7 orbitals)
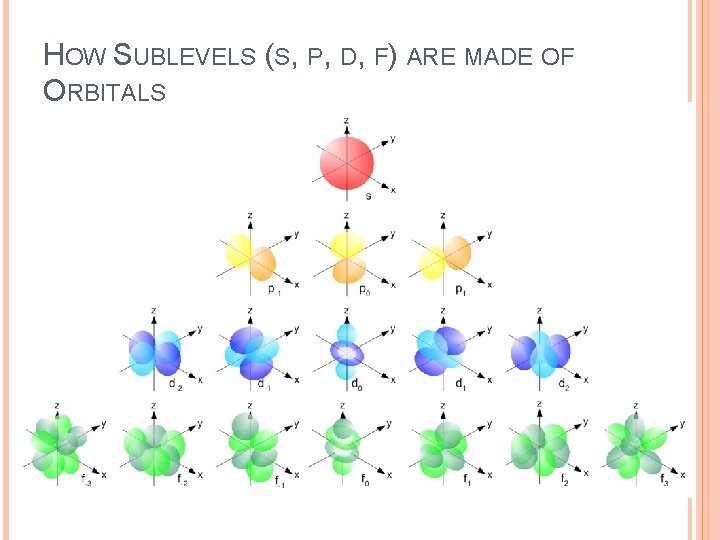
HOW SUBLEVELS (S, P, D, F) ARE MADE OF ORBITALS

HIERARCHY OF ENERGY LEVELS, SUBLEVELS, AND ORBITALS
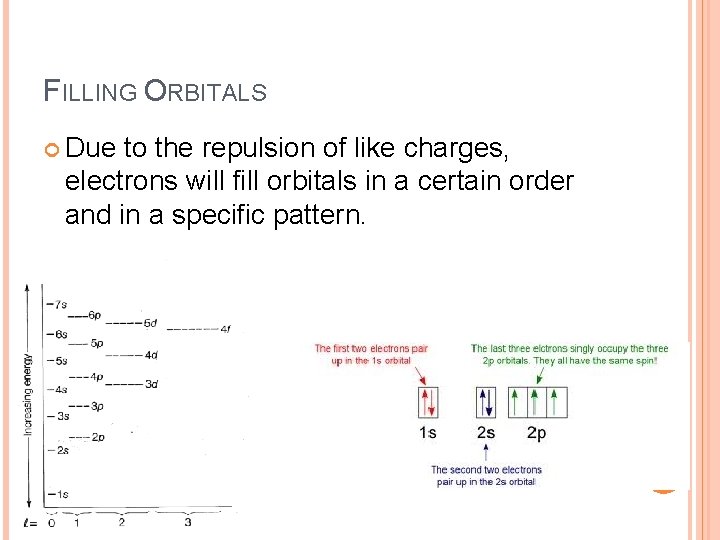
FILLING ORBITALS Due to the repulsion of like charges, electrons will fill orbitals in a certain order and in a specific pattern.
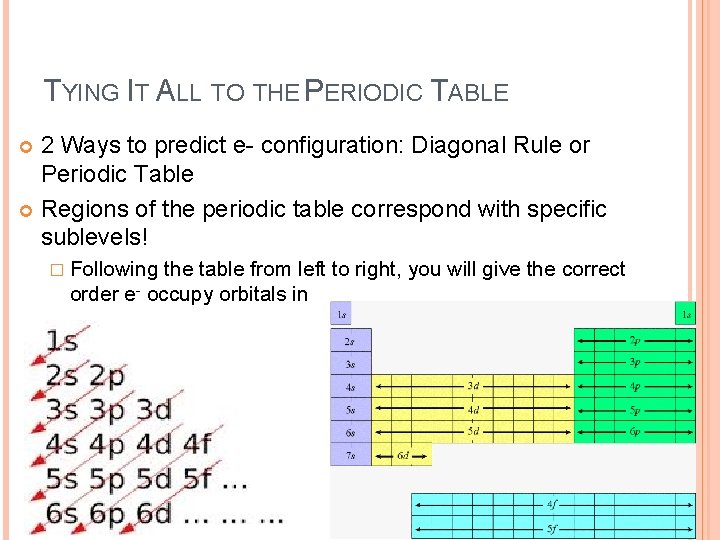
TYING IT ALL TO THE PERIODIC TABLE 2 Ways to predict e- configuration: Diagonal Rule or Periodic Table Regions of the periodic table correspond with specific sublevels! � Following the table from left to right, you will give the correct order e- occupy orbitals in

ELECTRON CONFIGURATION EXAMPLES Nitrogen (7 electrons) � 2 in 1 s sublevel (1 orbital) � 2 in 2 s sublevel (1 orbital) � 3 in 2 p sublevel (3 orbitals) Electron Configuration = 1 s 2 2 p 3 Coefficient = Energy Level orbitals are found in. Letter = Shape of the orbitals Exponent = Number of electrons occupying the orbitals

TRY IT! Titanium (Ti) Sulfur (S) Tantalum (Ta)
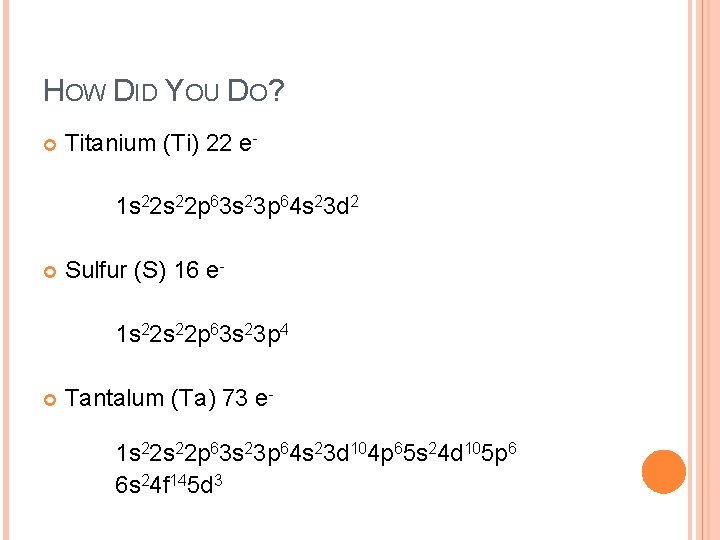
HOW DID YOU DO? Titanium (Ti) 22 e 1 s 22 p 63 s 23 p 64 s 23 d 2 Sulfur (S) 16 e 1 s 22 p 63 s 23 p 4 Tantalum (Ta) 73 e 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 6 6 s 24 f 145 d 3
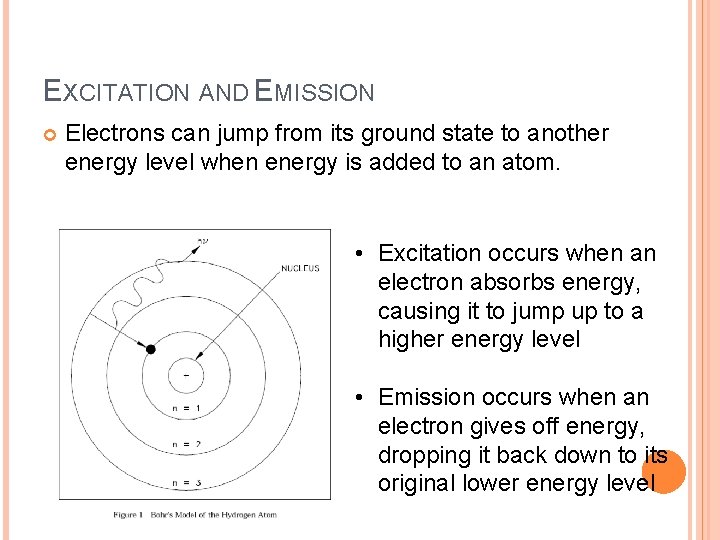
EXCITATION AND EMISSION Electrons can jump from its ground state to another energy level when energy is added to an atom. • Excitation occurs when an electron absorbs energy, causing it to jump up to a higher energy level • Emission occurs when an electron gives off energy, dropping it back down to its original lower energy level
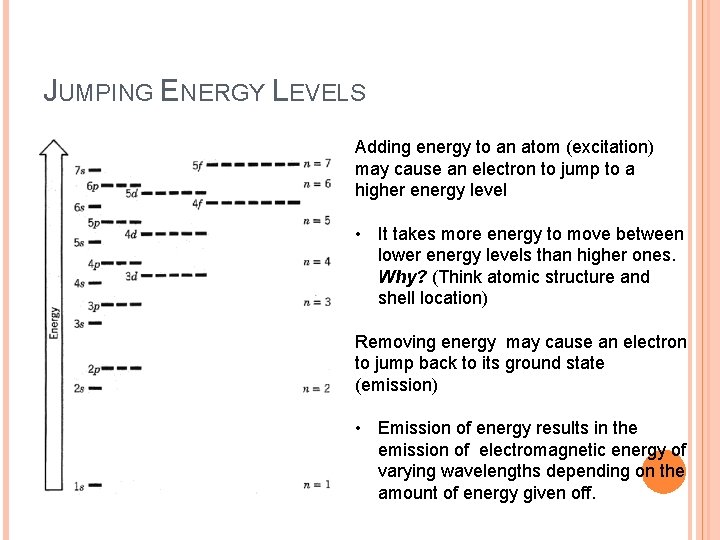
JUMPING ENERGY LEVELS Adding energy to an atom (excitation) may cause an electron to jump to a higher energy level • It takes more energy to move between lower energy levels than higher ones. Why? (Think atomic structure and shell location) Removing energy may cause an electron to jump back to its ground state (emission) • Emission of energy results in the emission of electromagnetic energy of varying wavelengths depending on the amount of energy given off.

VALENCE VS CORE ELECTRONS Valence electrons are those found in the outermost energy level. Core electrons are the electrons occupying the orbitals of inner energy levels. Using Bohr model: how many core/valence electrons for Cl?
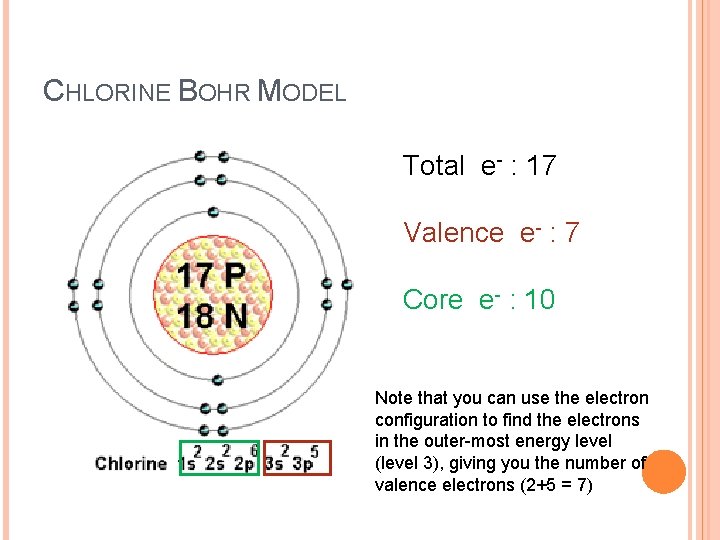
CHLORINE BOHR MODEL Total e- : 17 Valence e- : 7 Core e- : 10 Note that you can use the electron configuration to find the electrons in the outer-most energy level (level 3), giving you the number of valence electrons (2+5 = 7)
- Slides: 21