Electron configuration and electron shells Numbers of the

Electron configuration and electron shells

Numbers of the periodic table Remembering these will help you draw electron configurations Atomic Number – Number of protons in the nucleus Mass Number- the number of protons and neutrons in the nucleus Atomic mass – The average mass of an atom of a particular element
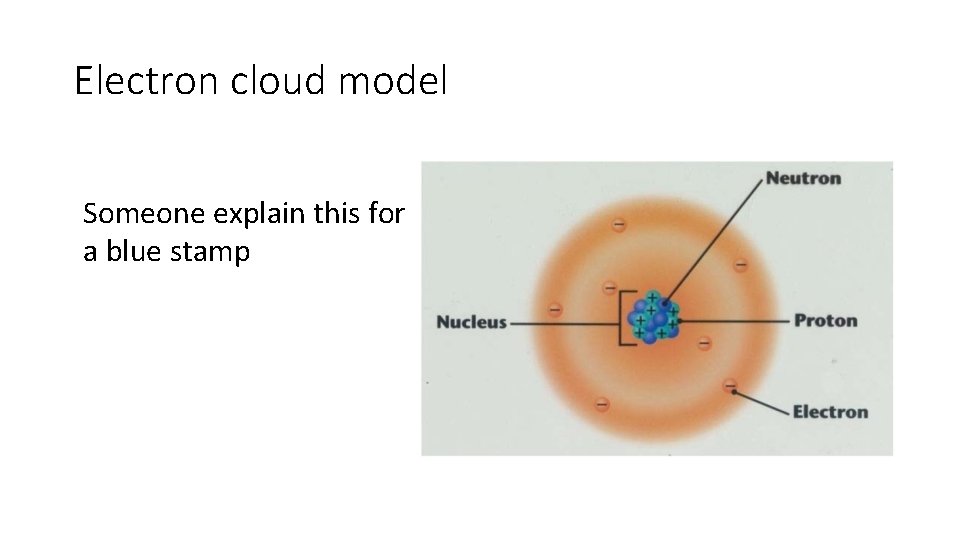
Electron cloud model Someone explain this for a blue stamp
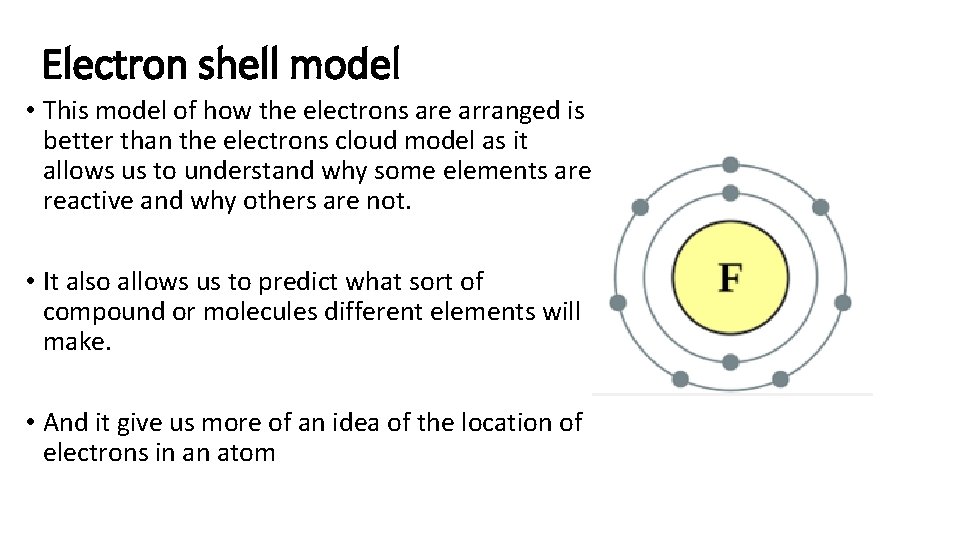
Electron shell model • This model of how the electrons are arranged is better than the electrons cloud model as it allows us to understand why some elements are reactive and why others are not. • It also allows us to predict what sort of compound or molecules different elements will make. • And it give us more of an idea of the location of electrons in an atom
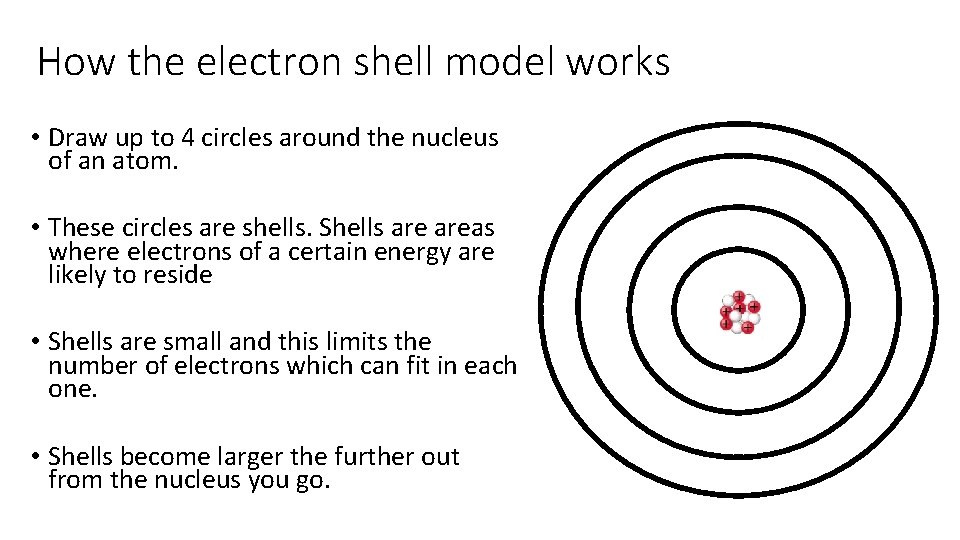
How the electron shell model works • Draw up to 4 circles around the nucleus of an atom. • These circles are shells. Shells areas where electrons of a certain energy are likely to reside • Shells are small and this limits the number of electrons which can fit in each one. • Shells become larger the further out from the nucleus you go.
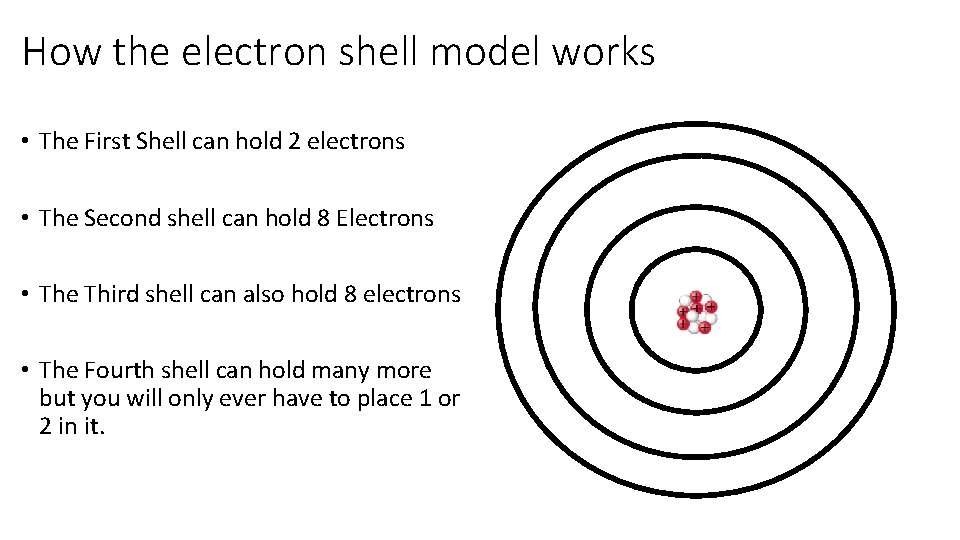
How the electron shell model works • The First Shell can hold 2 electrons • The Second shell can hold 8 Electrons • The Third shell can also hold 8 electrons • The Fourth shell can hold many more but you will only ever have to place 1 or 2 in it.
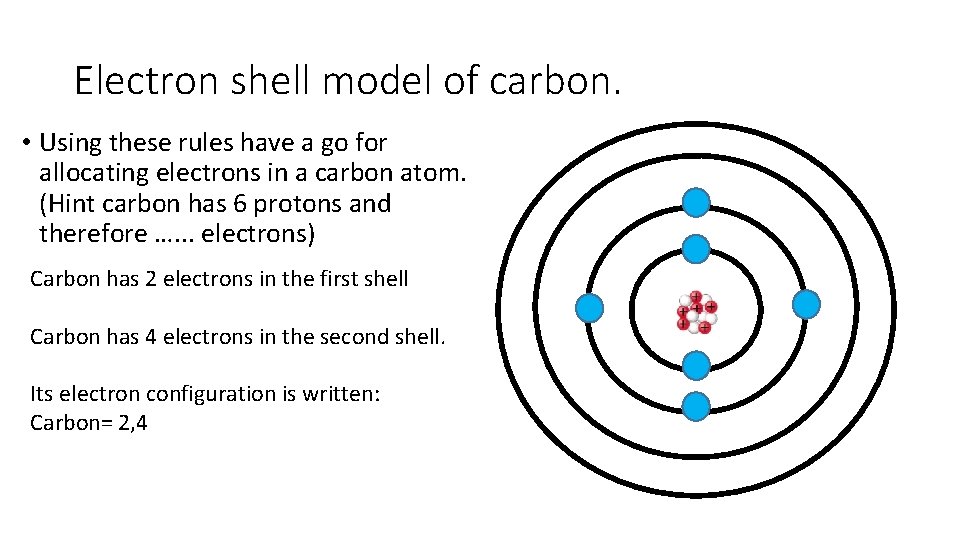
Electron shell model of carbon. • Using these rules have a go for allocating electrons in a carbon atom. (Hint carbon has 6 protons and therefore …. . . electrons) Carbon has 2 electrons in the first shell Carbon has 4 electrons in the second shell. Its electron configuration is written: Carbon= 2, 4
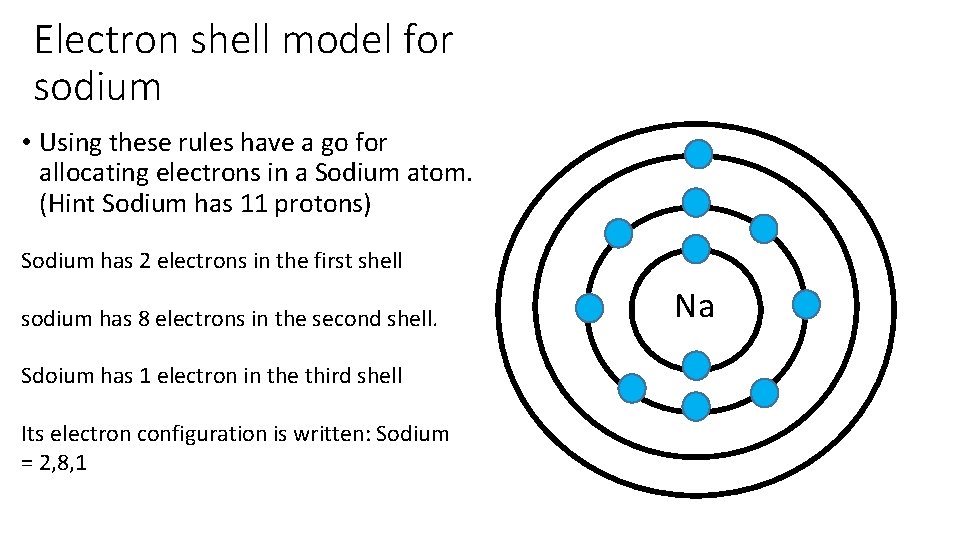
Electron shell model for sodium • Using these rules have a go for allocating electrons in a Sodium atom. (Hint Sodium has 11 protons) Sodium has 2 electrons in the first shell sodium has 8 electrons in the second shell. Sdoium has 1 electron in the third shell Its electron configuration is written: Sodium = 2, 8, 1 Na

Now complete electron shell diagrams for all of the atoms in your handout. This will be examined at the end of class and failure to complete will result in a read stamp. No alternatives! Once you have completed the atoms then draw electron shell diagrams for the ions of these atoms and answer the questions
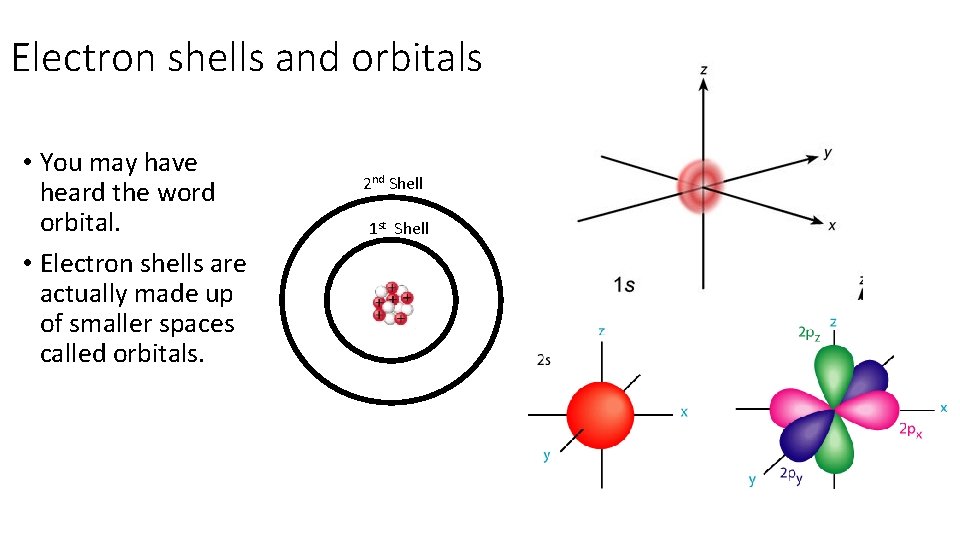
Electron shells and orbitals • You may have heard the word orbital. • Electron shells are actually made up of smaller spaces called orbitals. 2 nd Shell 1 st Shell
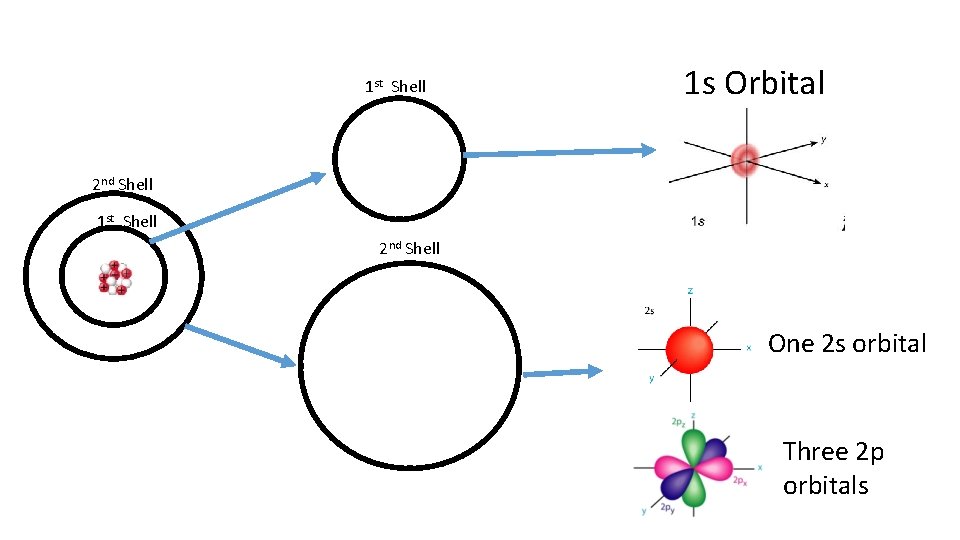
1 st Shell 1 s Orbital 2 nd Shell 1 st Shell 2 nd Shell One 2 s orbital Three 2 p orbitals
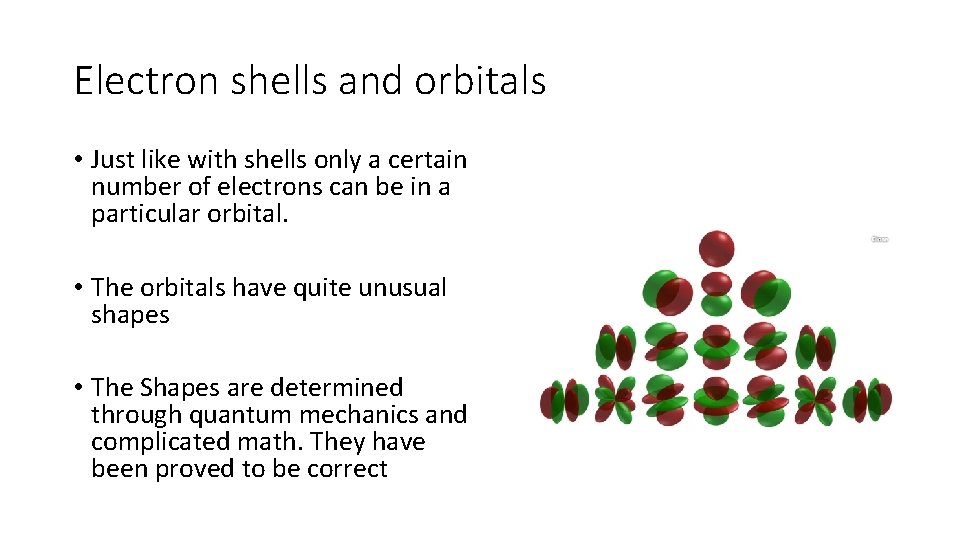
Electron shells and orbitals • Just like with shells only a certain number of electrons can be in a particular orbital. • The orbitals have quite unusual shapes • The Shapes are determined through quantum mechanics and complicated math. They have been proved to be correct

The difference between Shells and orbitals • Shells explain the amount of energy an electron in an atom will have. • Orbitals tell us how much energy the electrons have and also give us a good idea of where they are likely to be found in an atom. • Orbitals are a better description of how things are but we will talk about shells as they are sufficient to explain year 10 chemistry

- Slides: 14