Electron Configuration 4 f Sublevels 4 d Energy

Electron Configuration
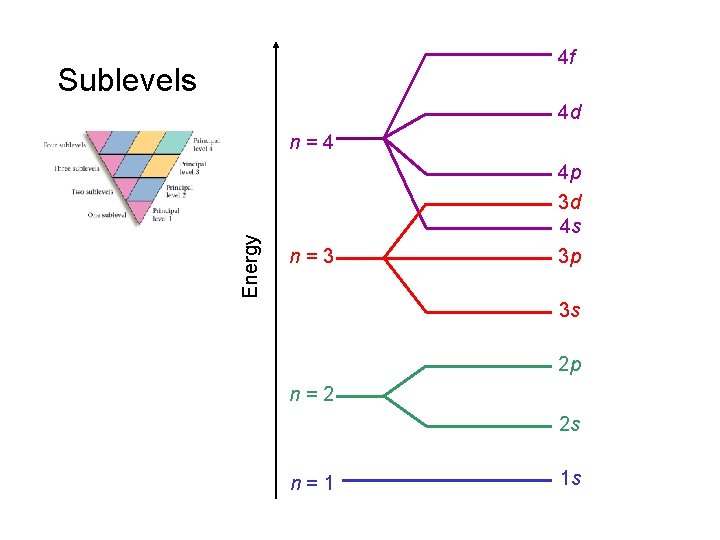
4 f Sublevels 4 d Energy n=4 n=3 4 p 3 d 4 s 3 p 3 s 2 p n=2 2 s n=1 1 s

4 f Sublevels 4 d s p s d p s n=4 f d p Energy s n=3 4 p 3 d 4 s 3 p 3 s 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 10… 2 p n=2 2 s n=1 1 s

Filling Rules for Electron Orbitals Aufbau Principle: Electrons are added one at a time to the lowest energy orbitals available until all the electrons of the atom have been accounted for. Pauli Exclusion Principle: An orbital can hold a maximum of two electrons. To occupy the same orbital, two electrons must spin in opposite directions. Hund’s Rule: Electrons occupy equal-energy orbitals so that a maximum number of unpaired electrons results. *Aufbau is German for “building up”
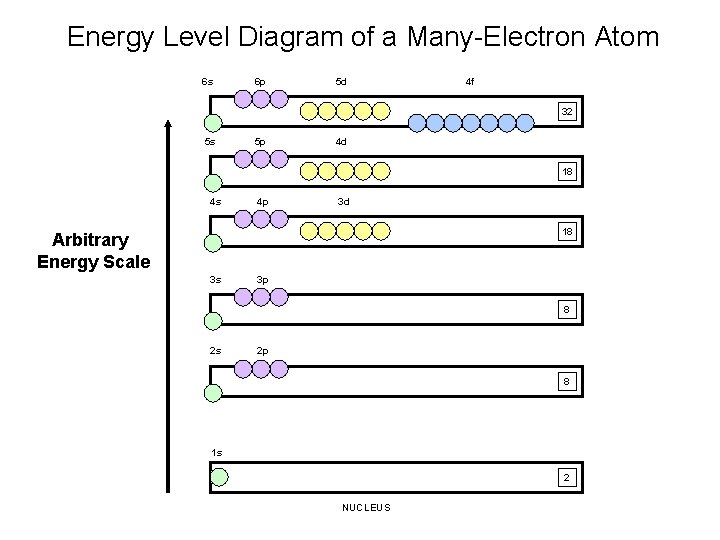
Energy Level Diagram of a Many-Electron Atom 6 s 6 p 5 d 4 f 32 5 s 5 p 4 d 18 4 s 4 p 3 d 18 Arbitrary Energy Scale 3 s 3 p 8 2 s 2 p 8 1 s 2 NUCLEUS
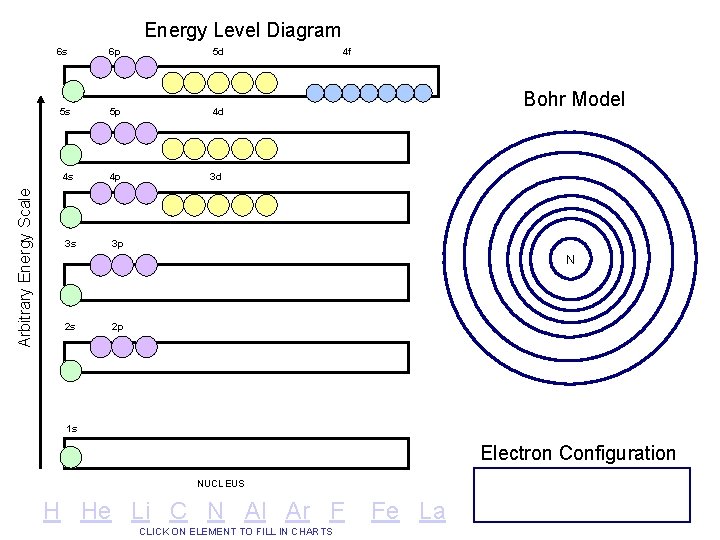
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La
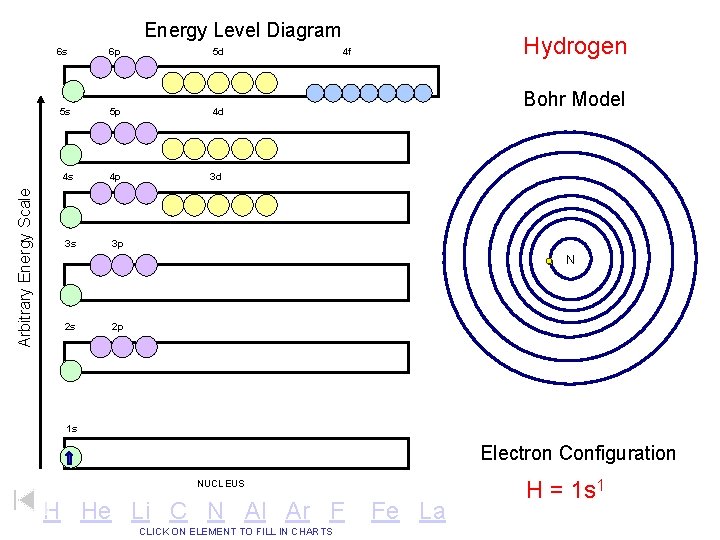
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Hydrogen 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La H = 1 s 1
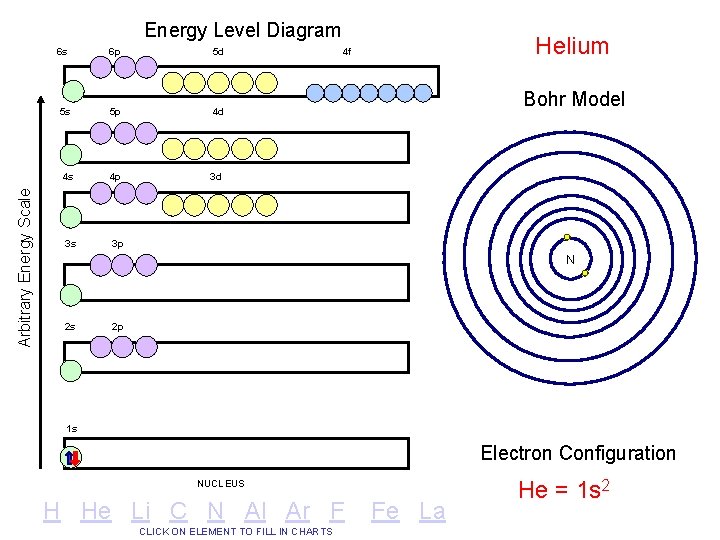
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Helium 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La He = 1 s 2
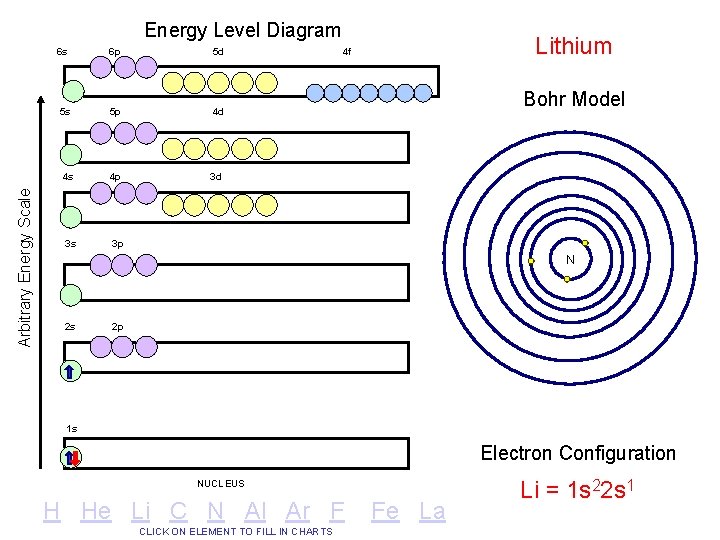
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Lithium 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La Li = 1 s 22 s 1
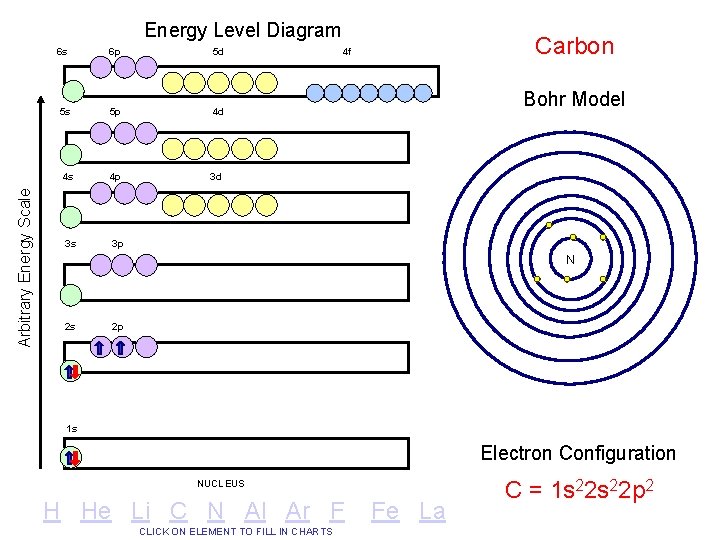
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Carbon 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La C = 1 s 22 p 2
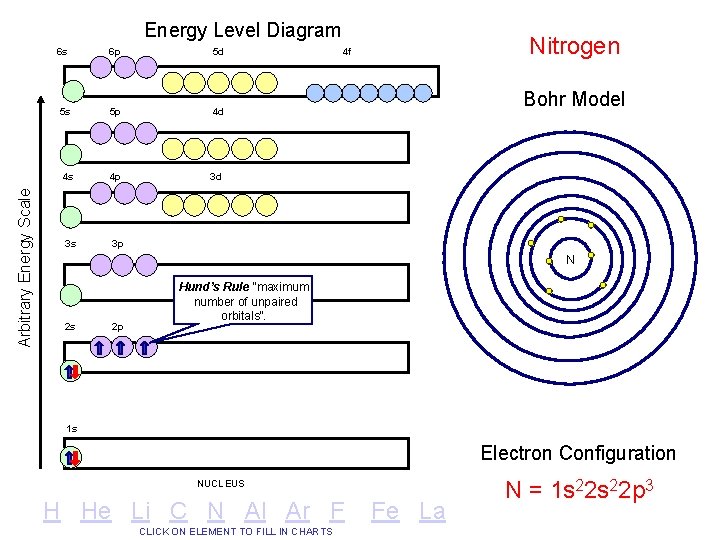
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Nitrogen 4 f Bohr Model N 2 s 2 p Hund’s Rule “maximum number of unpaired orbitals”. 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La N = 1 s 22 p 3
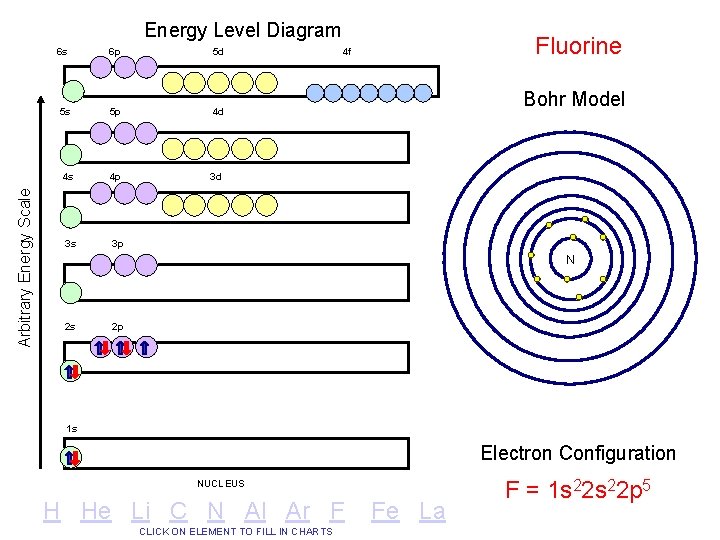
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Fluorine 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La F = 1 s 22 p 5
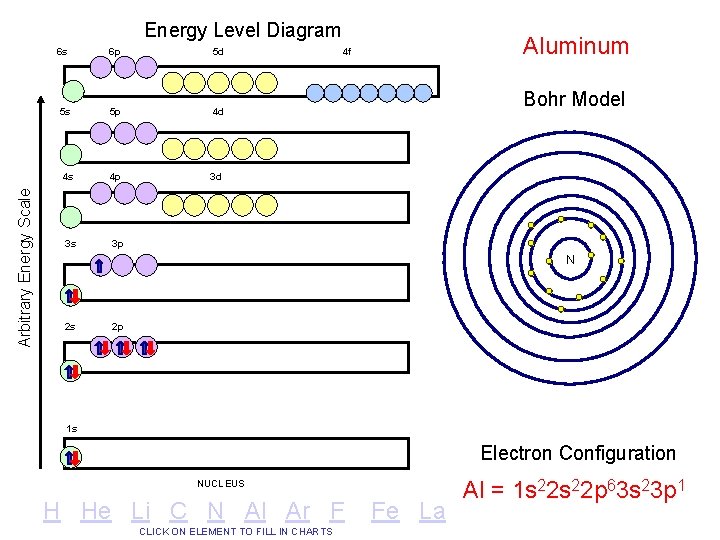
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Aluminum 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La Al = 1 s 22 p 63 s 23 p 1
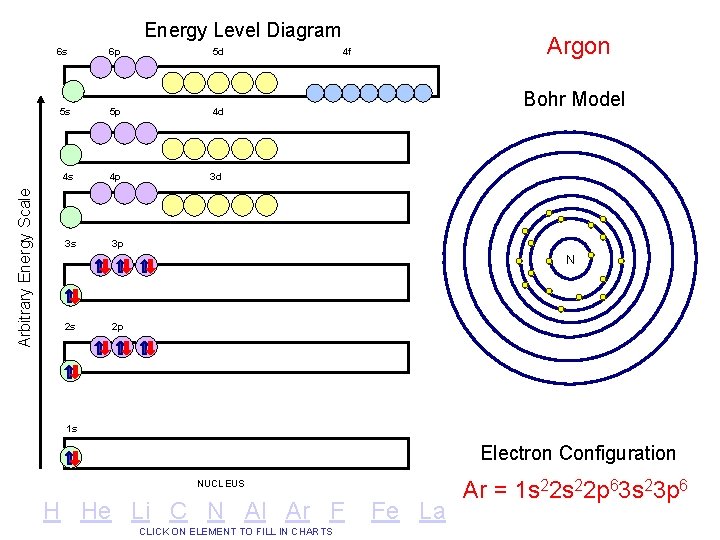
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Argon 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La Ar = 1 s 22 p 63 s 23 p 6
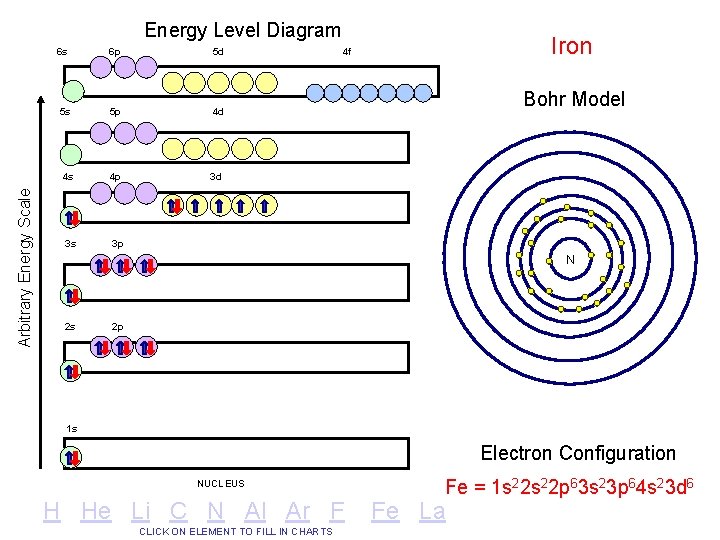
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Iron 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe = 1 s 22 p 63 s 23 p 64 s 23 d 6 Fe La
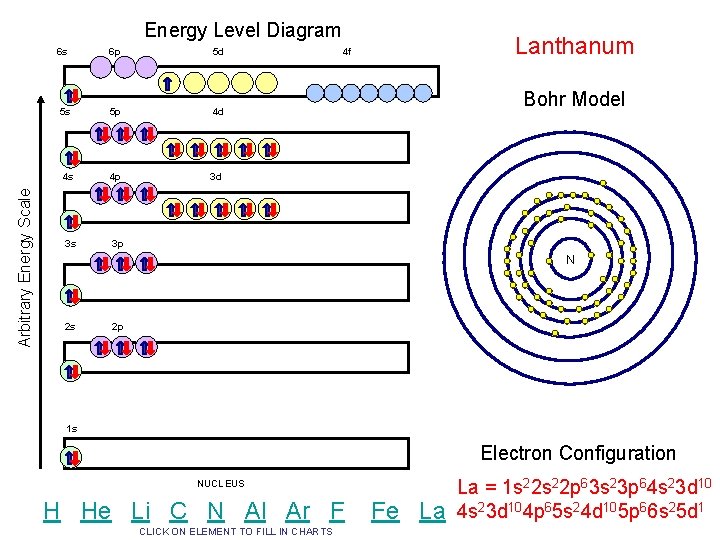
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Lanthanum 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La La = 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 66 s 25 d 1

Shorthand Configuration
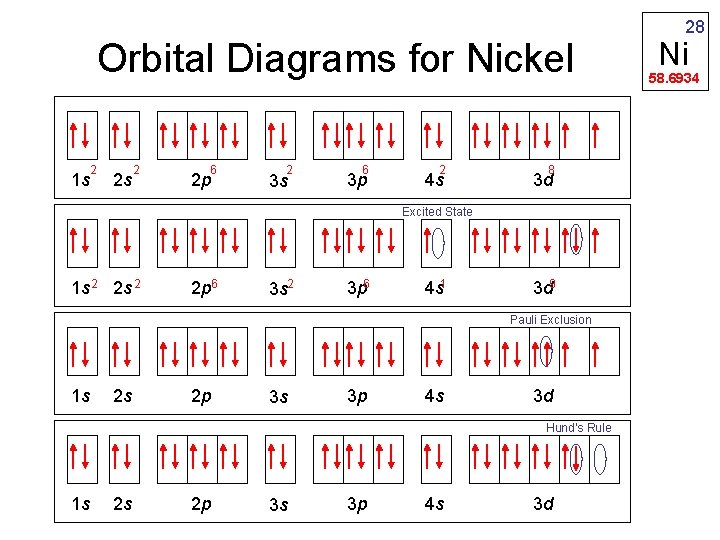
Orbital Diagrams for Nickel 2 1 s 2 s 2 6 2 p 2 3 s 6 3 p 2 4 s 8 3 d Excited State 1 s 2 2 p 6 3 s 2 3 p 6 4 s 1 3 d 9 Pauli Exclusion 1 s 2 s 2 p 3 s 3 p 4 s 3 d Hund’s Rule 1 s 2 s 2 p 3 s 3 p 4 s 3 d 28 Ni 58. 6934
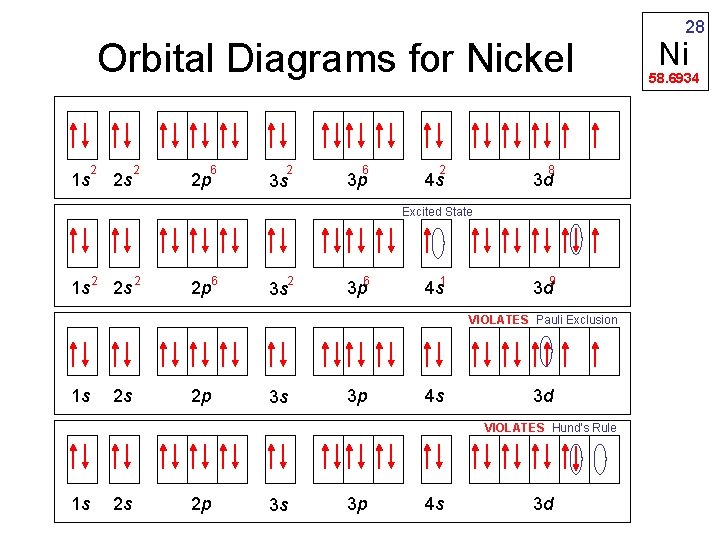
Orbital Diagrams for Nickel 2 1 s 2 s 2 6 2 p 2 3 s 6 3 p 2 8 4 s 3 d Excited State 1 s 2 2 p 6 2 3 s 3 p 6 4 s 1 3 d 9 VIOLATES Pauli Exclusion 1 s 2 s 2 p 3 s 3 p 4 s 3 d VIOLATES Hund’s Rule 1 s 2 s 2 p 3 s 3 p 4 s 3 d 28 Ni 58. 6934

Write out the complete electron configuration for the following: 1) An atom of nitrogen 2) An atom of silver POP QUIZ 3) An atom of uranium (shorthand) Fill in the orbital boxes for an atom of nickel (Ni) 1 s 2 s 2 p 3 s 3 p 4 s 3 d Which rule states no two electrons can spin the same direction in a single orbital? Extra credit: Draw a Bohr model of a Ti 4+ cation. Ti 4+ is isoelectronic to Argon.
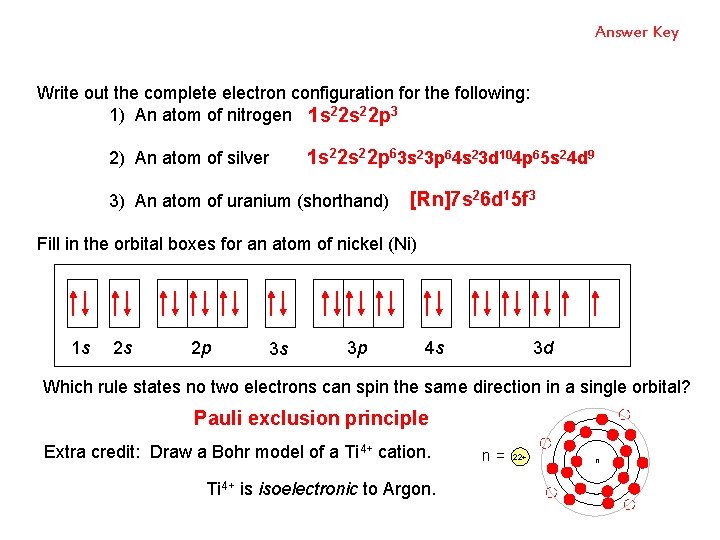
Answer Key Write out the complete electron configuration for the following: 1) An atom of nitrogen 1 s 22 p 3 2) An atom of silver 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 9 3) An atom of uranium (shorthand) [Rn]7 s 26 d 15 f 3 Fill in the orbital boxes for an atom of nickel (Ni) 1 s 2 s 2 p 3 s 3 p 4 s 3 d Which rule states no two electrons can spin the same direction in a single orbital? Pauli exclusion principle Extra credit: Draw a Bohr model of a Ti 4+ cation. Ti 4+ is isoelectronic to Argon. n= 22+ n

Periodic Table – Filling Order

Electron Configurations and the Periodic Table
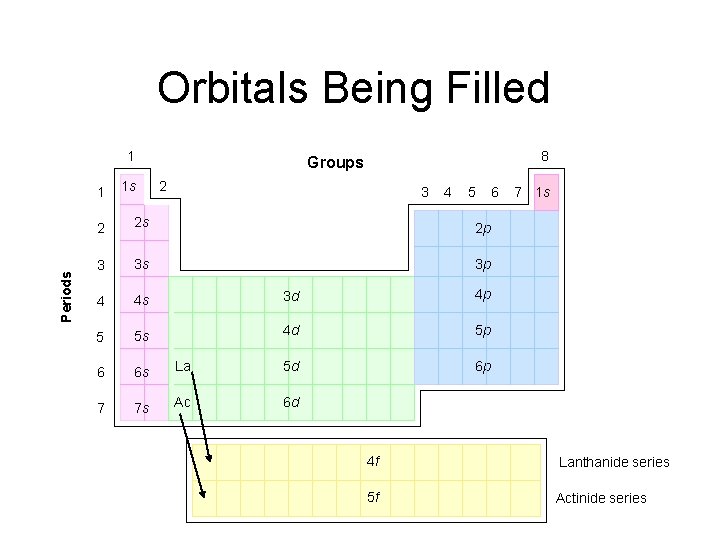
Orbitals Being Filled 1 Periods 1 1 s 8 Groups 2 3 4 5 2 2 s 2 p 3 3 s 3 p 4 4 s 3 d 4 p 5 5 s 4 d 5 p 6 6 s La 5 d 6 p 7 7 s Ac 6 d 6 7 1 s 4 f Lanthanide series 5 f Actinide series
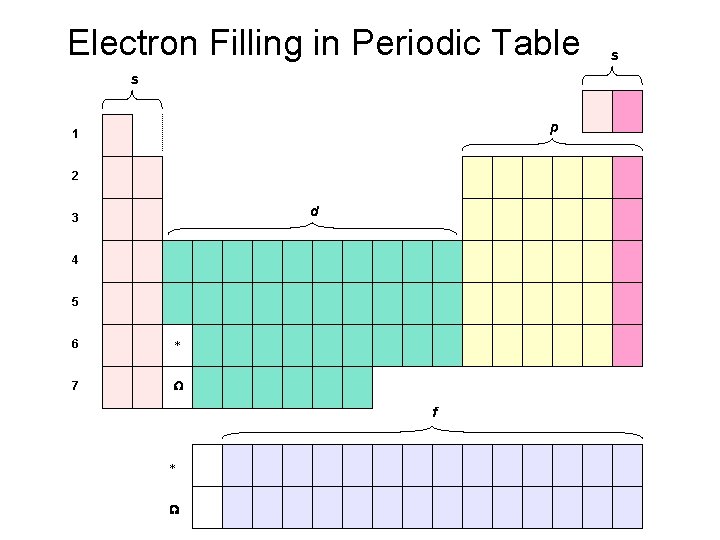
Electron Filling in Periodic Table s p 1 2 d 3 4 5 6 * 7 W f * W s
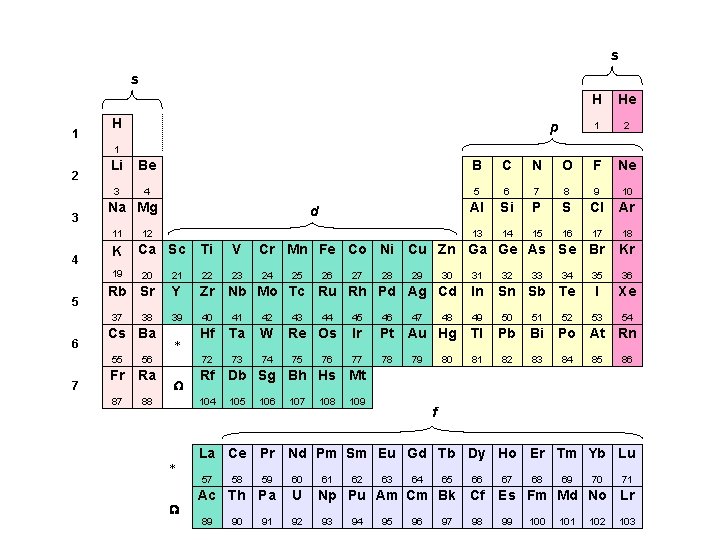
s s 1 H p H He 1 2 3 Li Be B C N O F Ne 3 4 5 6 7 8 9 10 Al Si P S Cl Ar 13 14 15 16 17 18 Na Mg 11 4 K 19 5 7 12 Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 23 24 35 36 I Xe 53 54 20 21 22 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In 39 40 41 42 49 50 51 Hf Ta W 72 73 74 37 6 d 38 Cs Ba 55 56 Fr Ra 87 88 * W 44 Re Os 75 76 27 28 29 30 47 48 31 32 33 Sn Sb Te 45 46 Ir Pt Au Hg Tl Pb Bi 77 78 81 82 83 79 80 34 52 Po At Rn 84 85 86 105 106 107 108 109 f La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 57 W 43 26 Rf Db Sg Bh Hs Mt 104 * 25 59 60 Ac Th Pa U 89 58 90 91 92 61 62 63 64 65 66 Np Pu Am Cm Bk Cf 93 94 95 96 97 98 67 68 69 70 71 Es Fm Md No Lr 99 100 101 102 103
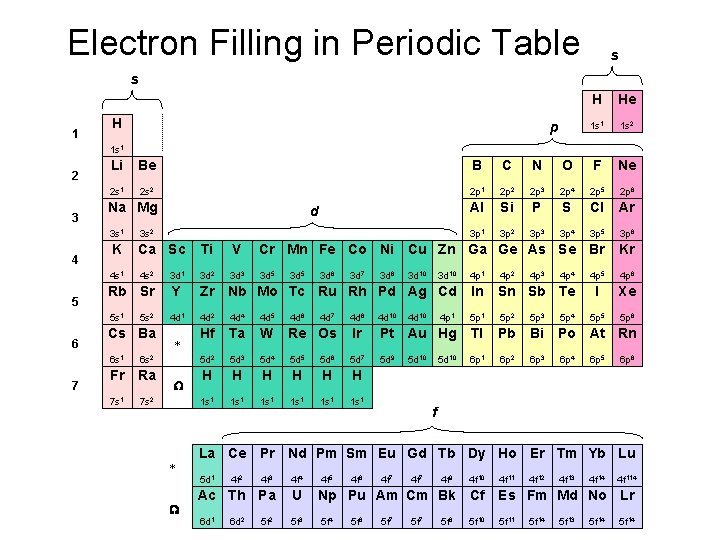
Electron Filling in Periodic Table s s 1 H p H He 1 s 1 1 s 2 1 s 1 2 3 4 5 6 7 Li Be B C N O F Ne 2 s 1 2 s 2 2 p 1 2 p 2 2 p 3 2 p 4 2 p 5 2 p 6 Al Si P S Cl Ar 3 p 1 3 p 2 3 p 3 3 p 4 3 p 5 3 p 6 Na Mg d 3 s 1 3 s 2 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 4 s 1 4 s 2 3 d 1 3 d 2 3 d 3 3 d 5 3 d 10 4 p 1 4 p 2 4 p 5 4 p 6 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe 5 s 1 4 d 2 4 d 4 4 d 5 4 d 6 4 d 7 4 d 8 4 d 10 4 p 1 5 p 2 5 p 3 5 p 4 5 p 5 5 p 6 Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn 5 d 2 5 d 3 5 d 4 5 d 5 5 d 6 5 d 7 5 d 9 6 p 1 6 p 2 6 p 3 6 p 4 H H H 1 s 1 1 s 1 5 s 2 Cs Ba 6 s 1 6 s 2 Fr Ra 7 s 1 7 s 2 * W * 3 d 6 3 d 7 3 d 8 3 d 10 4 d 10 5 d 10 4 p 3 4 p 4 6 p 5 6 p 6 f La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 5 d 1 W 3 d 5 4 f 2 4 f 3 4 f 4 Ac Th Pa U 6 d 1 5 f 3 6 d 2 5 f 2 4 f 5 4 f 6 4 f 7 4 f 9 4 f 10 Np Pu Am Cm Bk Cf 5 f 4 5 f 6 5 f 7 5 f 8 5 f 10 4 f 11 4 f 12 4 f 13 4 f 14 4 f 114 Es Fm Md No Lr 5 f 11 5 f 14 5 f 13 5 f 14
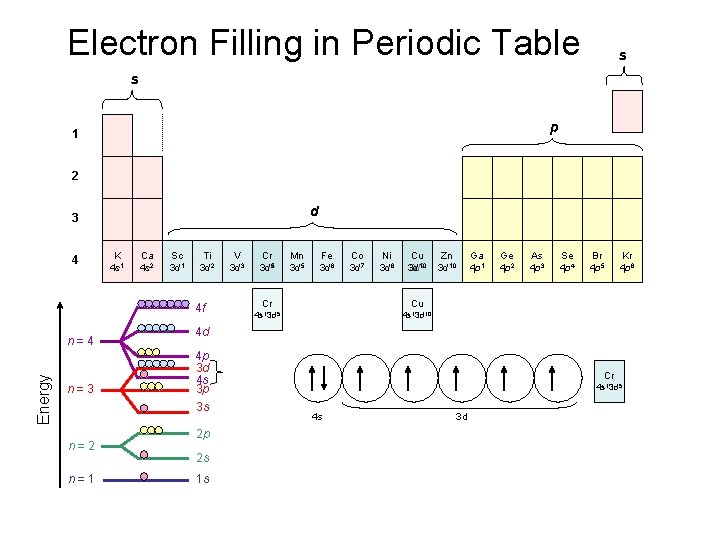
Electron Filling in Periodic Table s s p 1 2 d 3 4 K 4 s 1 Ca 4 s 2 Sc 3 d 1 Ti 3 d 2 4 f Energy n=4 n=3 n=2 n=1 V 3 d 3 Cr 3 d 54 Mn 3 d 5 Fe 3 d 6 Co 3 d 7 Ni 3 d 8 Cu 9 3 d 3 d 10 Cr Cu 4 s 13 d 5 4 s 13 d 10 Zn 3 d 10 Ga 4 p 1 Ge 4 p 2 As 4 p 3 Se 4 p 4 Br 4 p 5 Kr 4 p 6 4 d 4 p 3 d 4 s 3 p 3 s Cr 4 s 13 d 5 4 s 3 d 2 p 2 s Cu 1 s 4 s 13 d 10 4 s 3 d
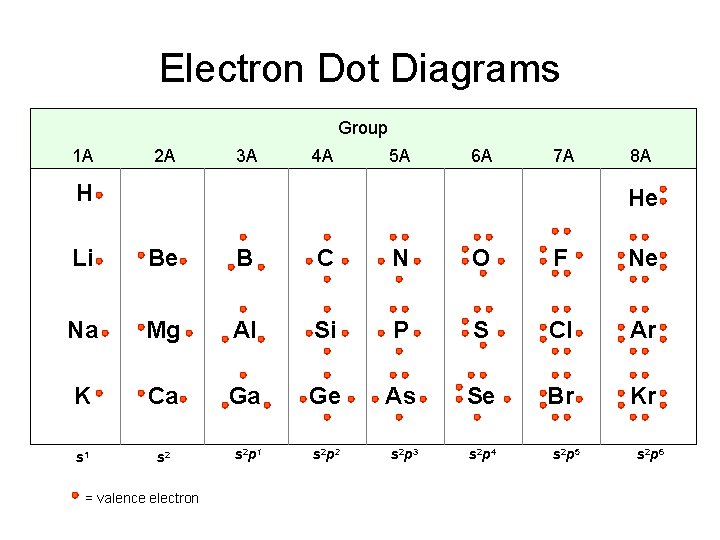
Electron Dot Diagrams Group 1 A 2 A 3 A 4 A 5 A 6 A 7 A H 8 A He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Ga Ge As Se Br Kr s 1 s 2 p 2 s 2 p 3 s 2 p 4 = valence electron s 2 p 5 s 2 p 6
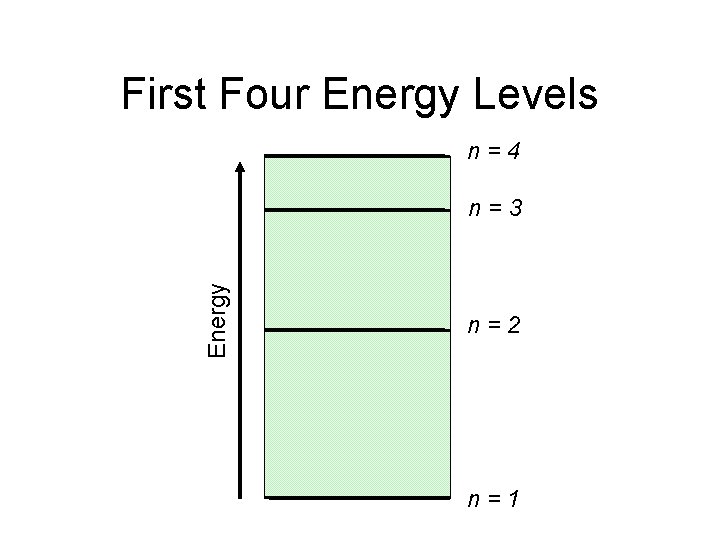
First Four Energy Levels n=4 Energy n=3 n=2 n=1
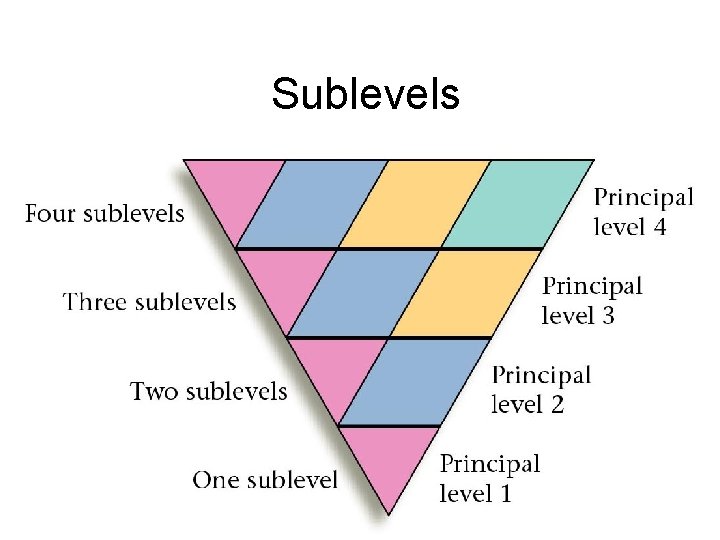
Sublevels
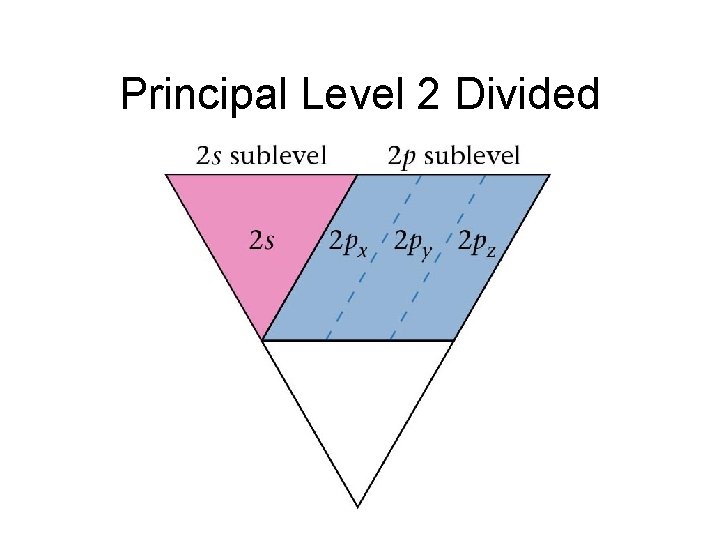
Principal Level 2 Divided
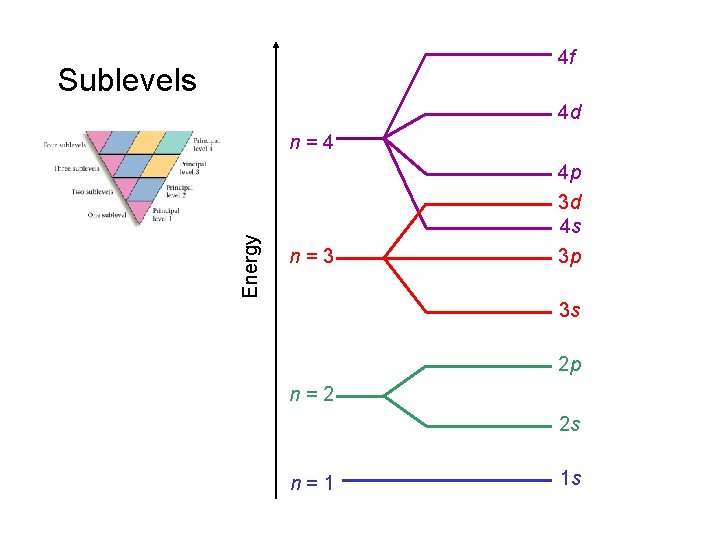
4 f Sublevels 4 d Energy n=4 n=3 4 p 3 d 4 s 3 p 3 s 2 p n=2 2 s n=1 1 s
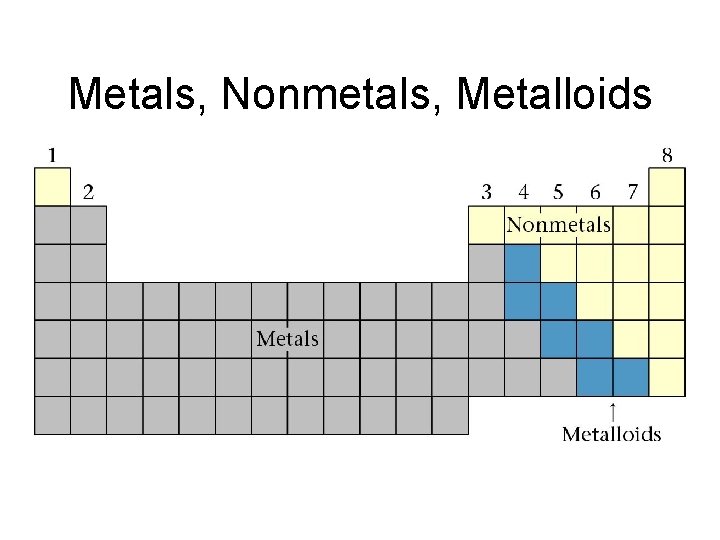
Metals, Nonmetals, Metalloids
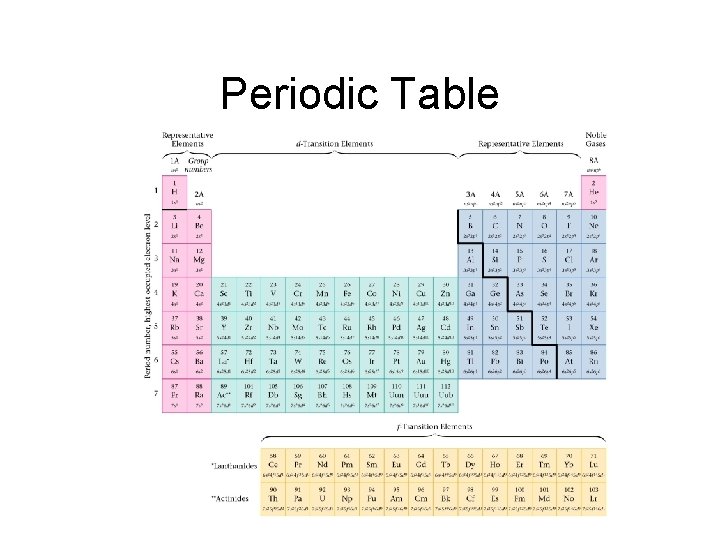
Periodic Table
- Slides: 35