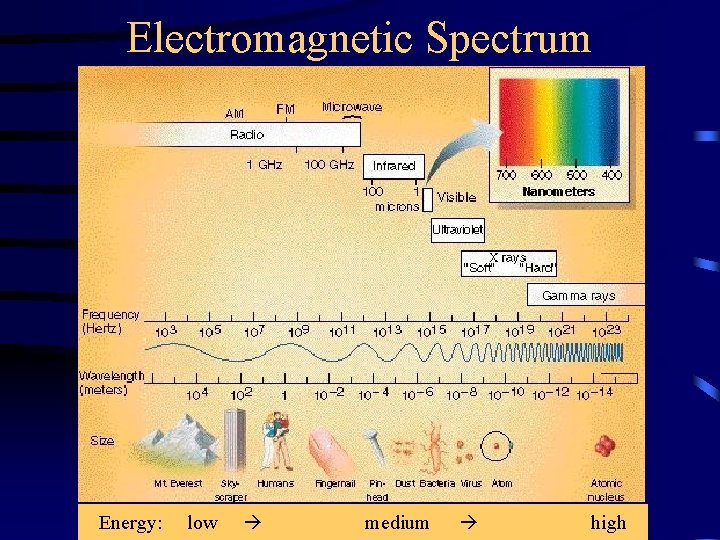
Electromagnetic Spectrum Energy low medium high Electromagnetic Radiation








- Slides: 17

Electromagnetic Spectrum Energy: low medium high

Electromagnetic Radiation: Quick Facts • There are different types of EM radiation, visible light is just one of them • EM waves can travel in vacuum, no medium needed • The speed of EM radiation “c” is the same for all types and very high ( light travels to the moon in 1 sec. ) • The higher the frequency, the smaller the wavelength ( f = c) • The higher the frequency, the higher the energy of EM radiation (E= h f, where h is a constant)

Visible Light • Color of light determined by its wavelength • White light is a mixture of all colors • Can separate individual colors with a prism

Three Things Light Tells Us • Temperature – from black body spectrum • Chemical composition – from spectral lines • Radial velocity – from Doppler shift

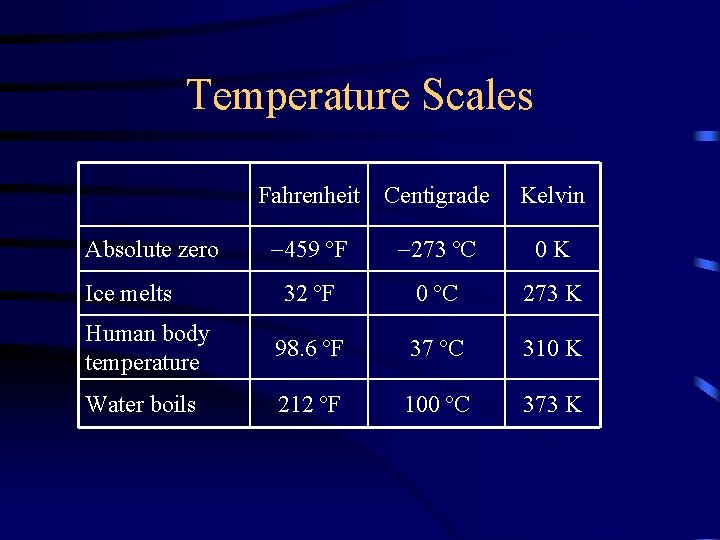
Temperature Scales Fahrenheit Centigrade Kelvin 459 ºF 273 ºC 0 K 32 ºF 0 ºC 273 K Human body temperature 98. 6 ºF 37 ºC 310 K Water boils 212 ºF 100 ºC 373 K Absolute zero Ice melts

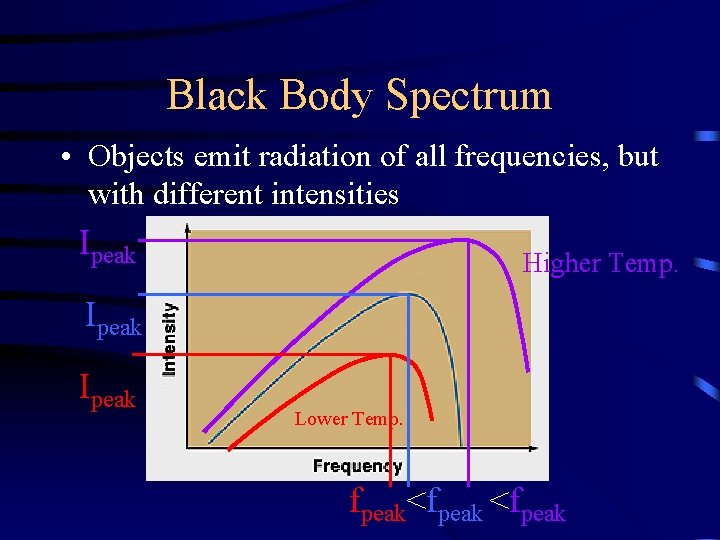
Black Body Spectrum • Objects emit radiation of all frequencies, but with different intensities Ipeak Higher Temp. Ipeak Lower Temp. fpeak<fpeak

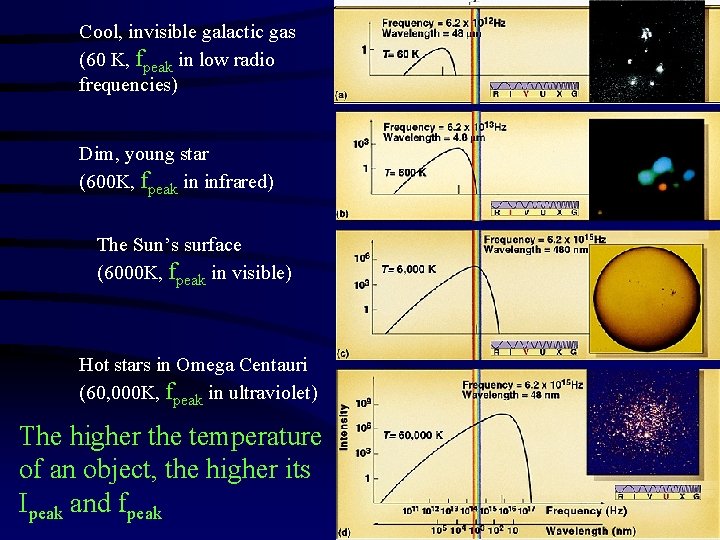
Cool, invisible galactic gas (60 K, fpeak in low radio frequencies) Dim, young star (600 K, fpeak in infrared) The Sun’s surface (6000 K, fpeak in visible) Hot stars in Omega Centauri (60, 000 K, fpeak in ultraviolet) The higher the temperature of an object, the higher its Ipeak and fpeak

Wien’s Law • The peak of the intensity curve will move with temperature, this is Wien’s law: Temperature * wavelength = constant = 0. 0029 K*m So: the higher the temperature T, the smaller the wavelength, i. e. the higher the energy of the electromagnetic wave

Example • Peak wavelength of the Sun is 500 nm, so T = (0. 0029 K*m)/(5 x 10 -7 m) = 5800 K • Instructor temperature: roughly 100 F = 37 C = 310 K, so wavelength = (0. 0029 K*m)/310 K = 9. 35 * 10 -6 m = 9350 nm infrared radiation

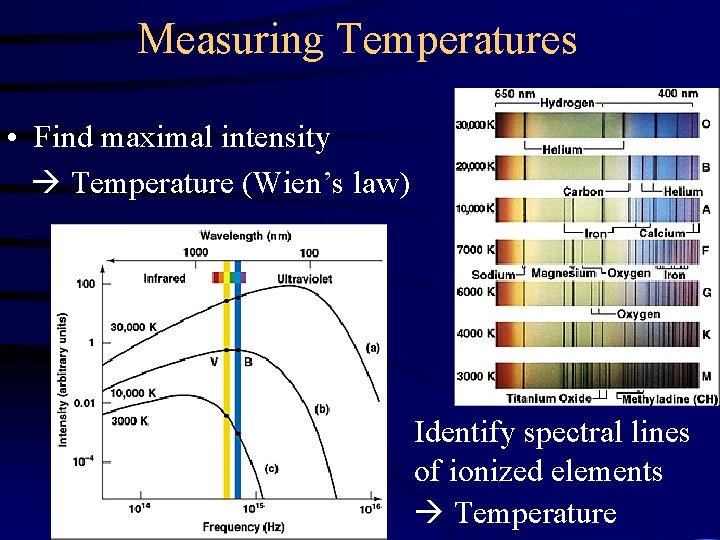
Measuring Temperatures • Find maximal intensity Temperature (Wien’s law) Identify spectral lines of ionized elements Temperature

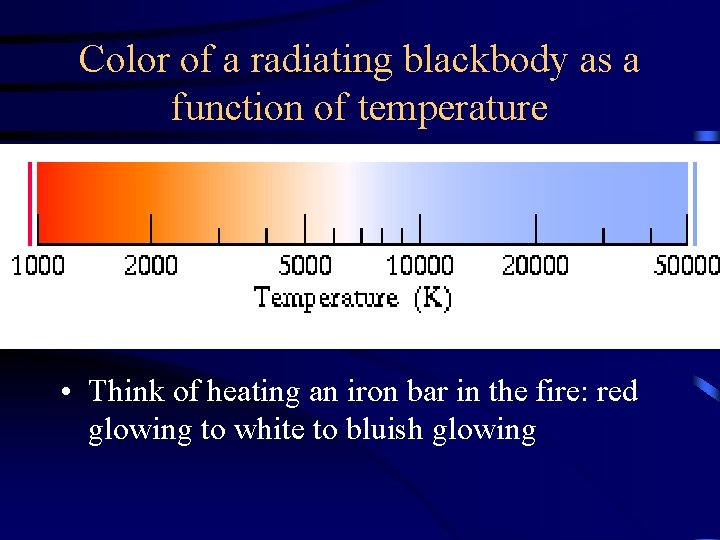
Color of a radiating blackbody as a function of temperature • Think of heating an iron bar in the fire: red glowing to white to bluish glowing

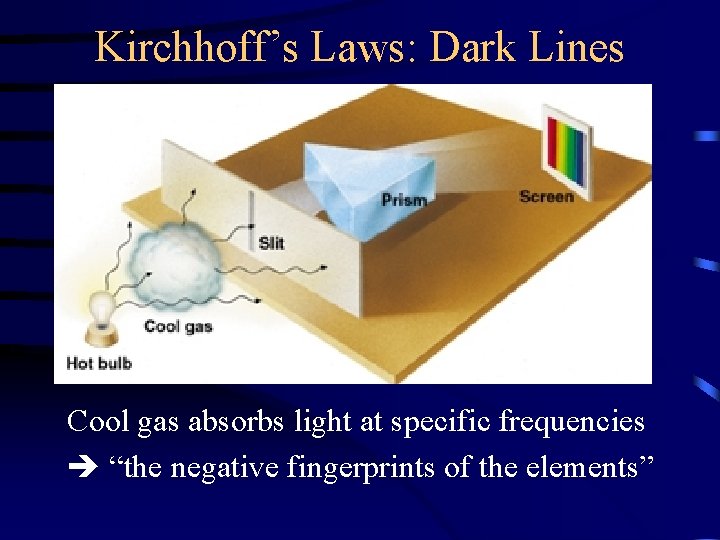
Kirchhoff’s Laws: Dark Lines Cool gas absorbs light at specific frequencies “the negative fingerprints of the elements”

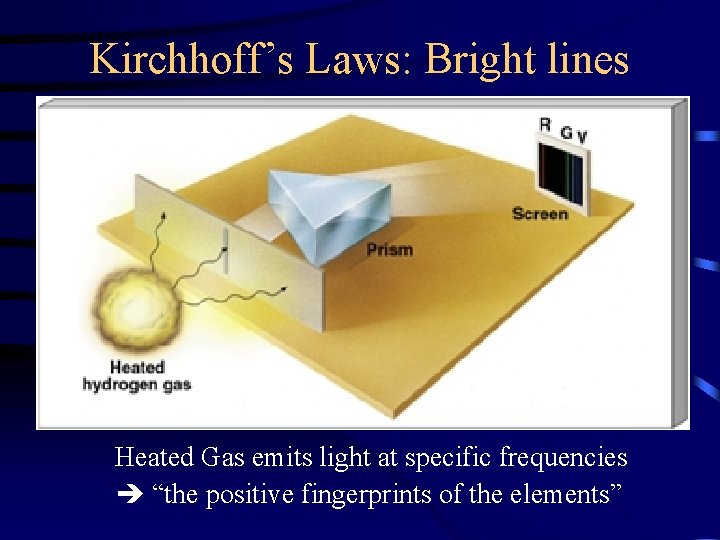
Kirchhoff’s Laws: Bright lines Heated Gas emits light at specific frequencies “the positive fingerprints of the elements”

Kirchhoff’s Laws 1. A luminous solid or liquid (or a sufficiently dense gas) emits light of all wavelengths: the black body spectrum 2. Light of a low density hot gas consists of a series of discrete bright emission lines: the positive “fingerprints” of its chemical elements! 3. A cool, thin gas absorbs certain wavelengths from a continuous spectrum dark absorption ( “Fraunhofer”) lines in continuous spectrum: negative “fingerprints” of its chemical elements, precisely at the same wavelengths as emission lines.

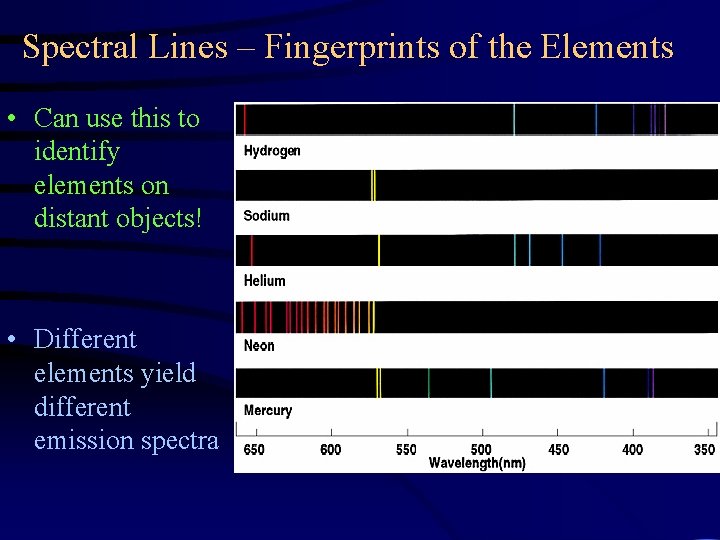
Spectral Lines – Fingerprints of the Elements • Can use this to identify elements on distant objects! • Different elements yield different emission spectra

Spectral Lines • Origin of discrete spectral lines: atomic structure of matter • Atoms are made up of electrons and nuclei – Nuclei themselves are made up of protons and neutrons • Electrons orbit the nuclei, as planets orbit the sun • Only certain orbits allowed Quantum jumps!

• The energy of the electron depends on orbit • When an electron jumps from one orbital to another, it emits (emission line) or absorbs (absorption line) a photon of a certain energy • The frequency of emitted or absorbed photon is related to its energy E=hf (h is called Planck’s constant, f is frequency)