ELECTROLYTES DISTURBANCES Oleh Dr Husnil Kadri M Kes

ELECTROLYTES DISTURBANCES Oleh: Dr. Husnil Kadri, M. Kes Bagian Biokimia Fakultas Kedokteran Universitas Andalas Padang
![Change in [Electrolyte] Can Occur By… 1. ) Increase/Decrease in amount of electrolyte 2. Change in [Electrolyte] Can Occur By… 1. ) Increase/Decrease in amount of electrolyte 2.](http://slidetodoc.com/presentation_image/38c37cea950ec63d4a91aeeca3dddeb2/image-2.jpg)
Change in [Electrolyte] Can Occur By… 1. ) Increase/Decrease in amount of electrolyte 2. ) Increase/Decrease in amount of water Remember Concentration = amount of solute volume of solution

Sodium (Na+) Key role (s): plasma osmolality and water balance Regulation: Thirst Kidney Na+/K+ ATPase pumps Na+/H+ pumps Blood volume status ADH (saves water) when blood volume or plasma osmolality renin when arteriolar pressure or Na+ aldosterone (saves salt) when Na+ ( renin) Clinical: Na disorders = water disorders Hyponatremia Hypernatremia
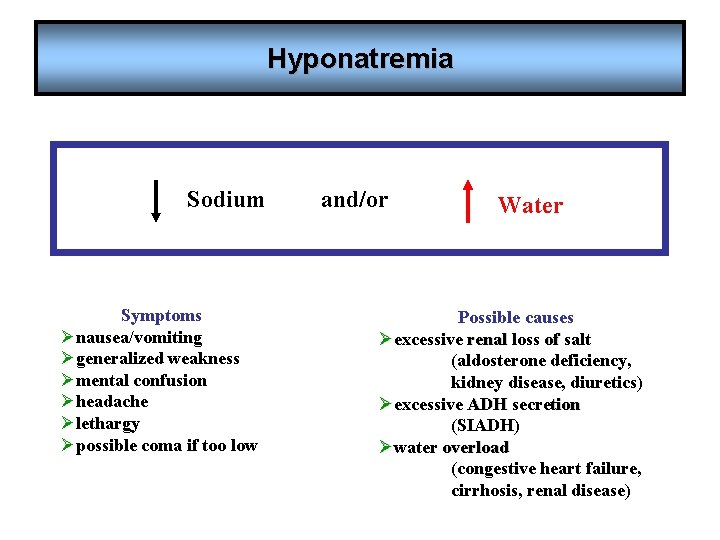
Hyponatremia Sodium Symptoms Ønausea/vomiting Øgeneralized weakness Ømental confusion Øheadache Ølethargy Øpossible coma if too low and/or Water Possible causes Øexcessive renal loss of salt (aldosterone deficiency, kidney disease, diuretics) Øexcessive ADH secretion (SIADH) Øwater overload (congestive heart failure, cirrhosis, renal disease)
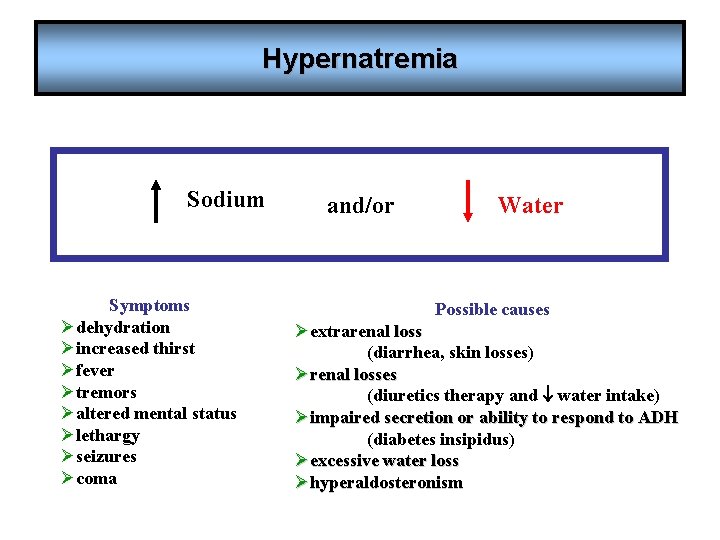
Hypernatremia Sodium Symptoms Ødehydration Øincreased thirst Øfever Øtremors Øaltered mental status Ølethargy Øseizures Øcoma and/or Water Possible causes Øextrarenal loss (diarrhea, skin losses) Ørenal losses (diuretics therapy and water intake) Øimpaired secretion or ability to respond to ADH (diabetes insipidus) Øexcessive water loss Øhyperaldosteronism

Potassium (K+) Key role (s): Regulate cardiac contraction and rhythm, muscle contraction Regulation: Kidneys Na+/K+ -ATPase pump Acid/Base balance (i. e. , K+/H+ pumps) Aldosterone results in K+ excretion and shift extracellular to intracellular. Clinical: Hypokalemia Hyperkalemia
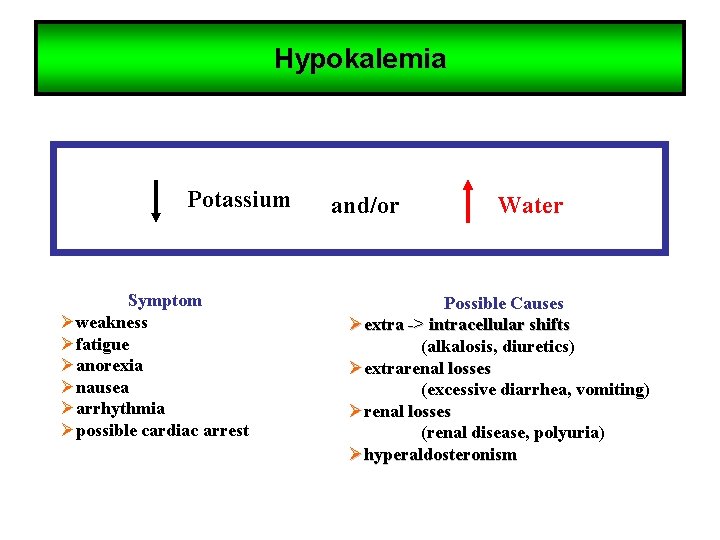
Hypokalemia Potassium Symptom Øweakness Øfatigue Øanorexia Ønausea Øarrhythmia Øpossible cardiac arrest and/or Water Possible Causes Øextra -> intracellular shifts (alkalosis, diuretics) Øextrarenal losses (excessive diarrhea, vomiting) Ørenal losses (renal disease, polyuria) Øhyperaldosteronism
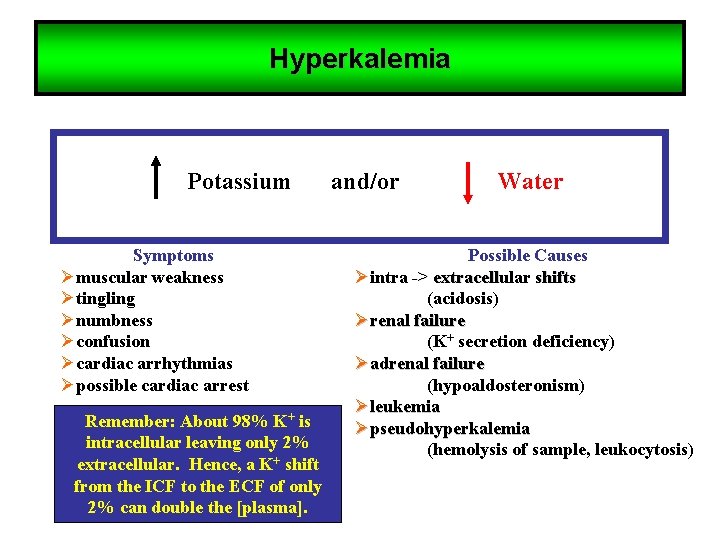
Hyperkalemia Potassium Symptoms Ømuscular weakness Øtingling Ønumbness Øconfusion Øcardiac arrhythmias Øpossible cardiac arrest Remember: About 98% K+ is intracellular leaving only 2% extracellular. Hence, a K+ shift from the ICF to the ECF of only 2% can double the [plasma]. and/or Water Possible Causes Øintra -> extracellular shifts (acidosis) Ørenal failure (K+ secretion deficiency) Øadrenal failure (hypoaldosteronism) Øleukemia Øpseudohyperkalemia (hemolysis of sample, leukocytosis)

Calcium (Ca 2+) Key role (s): primarily resides in bone, muscular contraction, neurotransmission, membrane transport, enzymes, and blood coagulation Regulation: Kidney (reabsorbed in the proximal tubules) Parathyroid hormone (PTH) Vitamin D – active form controls homeostasis Calcitonin – exact mechanism not known Clinical: Hypocalcemia – hypoparathyroidism, malabsorption of calcium or Vit. D, renal failure Hypercalcemia – hyperparathyroidism, excess Vit. D, tumors Serum Calcium = Ca 2+ionized (45%) + Caprotein-bound (45%) + Cacomplexed to anions (10%)

Magnesium (Mg 2+) Key role (s): enzyme cofactor; calcium and bone homeostasis Regulation: Kidney PTH, serum Mg 2+ aldosterone Clinical: hypomagnesemia – decreased intake (malabsorption, malnutrition), increased loss (renal disease, hyperaldosteronism, hyperparathyroidism) hypermagnesemia – usually increased intake of magnesium or renal disease

Chloride (Cl-) Key role (s): Maintains osmolality, blood volume, electric neutrality Regulation: kidneys (reabsorbed /w Na+ in the proximal tubules), aldosterone Clinical: Hypochloremia – similar causes as hyponatremia, prolonged vomiting, high [bicarbonate] associated metabolic alkalosis Hyperchloremia – similar causes as hypernatremia, dehydration, low [bicarbonate] associated with prolonged diarrhea or metabolic acidosis

Bicarbonate (HCO 3 -) Key role (s): determines p. H (along with H+); buffering the blood and maintaining acid/base equilibrium Regulation: kidneys (reabsorption in the tubules) lungs Clinical: Acid/Base disorders

Disorders of Water Balance: Dehydration • Water loss exceeds water intake and the body is in negative fluid balance • Causes include: hemorrhage, severe burns, prolonged vomiting or diarrhea, profuse sweating, and diuretic abuse • Signs and symptoms: thirst, dry flushed skin, and oliguria • Other consequences include hypovolemic shock and loss of electrolytes 13
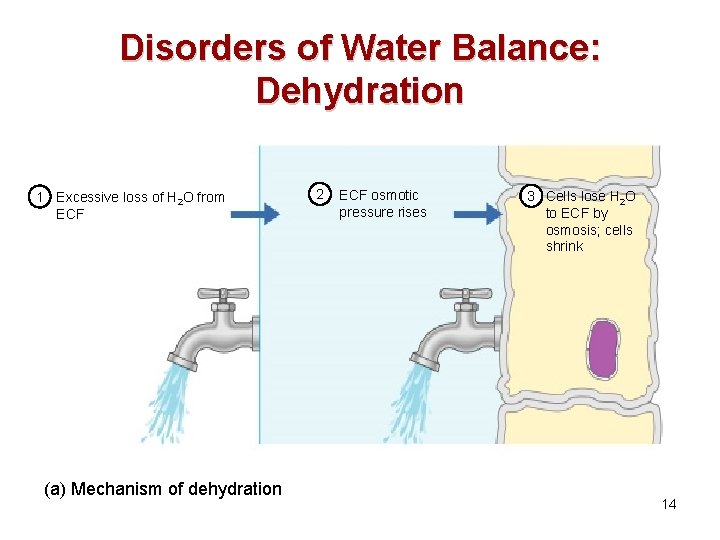
Disorders of Water Balance: Dehydration 1 Excessive loss of H 2 O from ECF (a) Mechanism of dehydration 2 ECF osmotic pressure rises 3 Cells lose H 2 O to ECF by osmosis; cells shrink 14

Disorders of Water Balance: Hypotonic Hydration • Amount of water ingested quickly can lead to cellular overhydration or water intoxication • ECF is diluted – sodium content is normal but excess water is present • The resulting hyponatremia promotes net osmosis into tissue cells, causing swelling 15
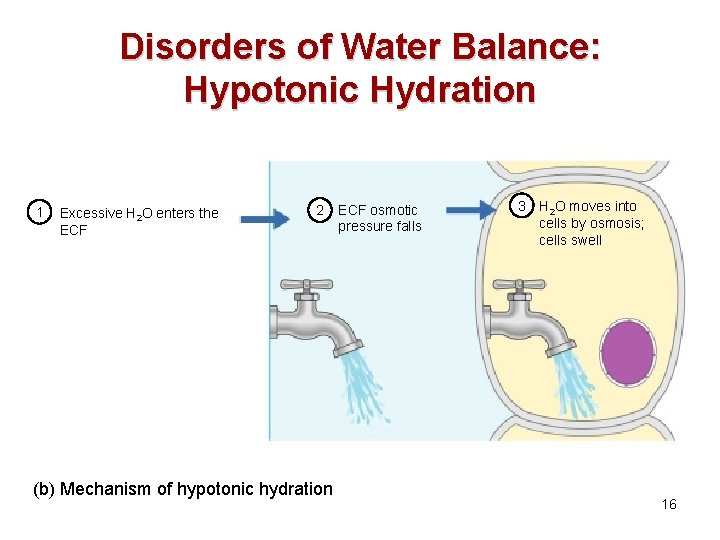
Disorders of Water Balance: Hypotonic Hydration 1 Excessive H 2 O enters the ECF 2 (b) Mechanism of hypotonic hydration ECF osmotic pressure falls 3 H 2 O moves into cells by osmosis; cells swell 16

Protein Imbalances • Plasma proteins(especially albumin) are important determinants of plasma volume • Hyperproteinemia is rare – Occurs with dehydration-induced hemoconcentration

Hypoproteinemia • Caused by – Anorexia – Malnutrition – Starvation – Fad dieting – Poorly balanced vegetarian diets

Hypoproteinemia • Poor absorption d/t GI malabsorptive diseases • Inflammation → protein can shift out of intravascular space • Hemorrhage

Hypoproteinemia: Clinical Manifestations • Edema (b/c insufficient oncotic pressure to “hold” water in vascular space) • • • Slow healing Anorexia Fatigue Anemia Muscle loss Ascites (same reason as edema)

Bioavailabilitas • Dalam makanan, mineral terdapat dalam bentuk garam yang sukar larut, kecuali K & Na. • Absorpsinya pada usus halus & besar. • Transportasi & penyimpanan memerlukan protein pengemban spesifik, contoh; Fe 3+-transferin Cu 2+-albumin

Kalsium & Fosfor • Keduanya membentuk garam appatite didalam tulang & gigi (80 -90%). • Absorpsi paling baik jika perbandingan dalam lumen usus Ca : P = 1 : 1 s/d 1 : 3 • Bila perbandingan > 3, maka absorpsi Ca terhambat (Rachitis). • Makanan penyebabnya disebut rachitogenik

Interaksi yang Menghambat • Beras mengandung asam fitat (P) sehingga mengikat Ca membentuk Ca-fitat. • Sayuran & buah yang mengandung asam oksalat juga akan menghambat absorpsi Ca.

Fungsi P • Ikatan fosfat berenergi tinggi ATP, ADP, kreatin-P, PEP, dll. • Komponen membran sel fosfolipid • Membentuk hidroksiapatit pada tulang dan gigi

Defisiensi Ca • • Rickets pada anak-anak Osteomalacia (osteoporosis) pada dewasa Tetani / kejang Postmenopause (estrogen rendah)

Defisiensi P • • Gangguan absorpsi di usus Ekskresi berlebihan melalui ginjal Sindrom Milkman Sindrom de Toni Fanconi

Penyakit yang Berhubungan dgn Na & K • Penyakit Addison - hipoaktif kelenjar kortek adrenal - hiponatremia & hiperkalemia • Penyakit Cushing - hiperaktif kelenjar kortek adrenal - hipernatremia & hipokalemia

Konsumsi Na. Cl berlebihan • Hipertensi • Diabetes • Gangguan ginjal kronik

Besi • Komponen penting pada: hemoglobin, sitokrom, katalase, peroksidase • Terdapat dalam makanan terutama daging sebagai ion Fe 3+ (Ferri). • Pengaturan absorpsi Fe dikenal sebagai mucosal block system

Mucosal Block System • Dalam lumen lambung, reduktor (asam askorbat, HCl, dll) mereduksi ferri menjadi ferro. • Ferro akan diabsorpsi mukosa usus. • Dalam sel usus, ferro dioksidasi kembali menjadi ferri. • Ion ferri diikat apoferritin membentuk ferritin.

Mucosal Block System • Bila tubuh tidak membutuhkan Fe, apoferritin menjadi jenuh. • Akibatnya ion ferro di lumen usus tidak bisa masuk ke dalam sel usus. • Fe akan dibuang bersama feses. • Bila tubuh butuh Fe, ferritin melepas ferri, • Ferri direduksi menjadi ferro.

Mucosal Block System • Dalam sirkulasi darah, ferro dioksidasi lagi oleh peroksidase (dalam ceruloplasmin). • Ion ferri kemudian diikat oleh apotransferin membentuk transferin. • Transferin ditranspor ke berbagai jaringan yang membutuhkan besi. • Dalam jangka panjang, besi disimpan sebagai hemosiderin jaringan.

Defisiensi Besi • Anemia mikrositer hipokrom, disebabkan oleh: - infeksi cacing tambang - perdarahan • Pil KB meningkatkan pembuangan besi

Kelebihan Besi • Hemosiderosis, disebabkan oleh: - Pemberian preparat besi - Transfusi darah • Bronze diabetikum (gangguan Mucosal Block System ).

Seng/Zinc • Berhubungan dengan fungsi enzim dan hormon; - karbonat anhidrase - laktat dehidrogenase - glutamat dehidrogenase - hormon insulin

Seng/Zinc • Faktor pengikat Zn dari sekret pankreas membantu absorpsi Zn di usus. • Absorpsi Zn berkompetisi dengan Cu. • Ekskresi melalui ; - empedu untuk keluar dengan feses - keringat - urine

Fluor (F) • • • Komponen jaringan keras, tu gigi. Melindungi email gigi. Fluor bersifat racun thd enolase (glikolisis) Air PAM mengandung fluor 1 -2 ppm. Defisiensi fluor --> karies dentis. Kelebihan fluor --> fluorosis (mottled enamel) = cekungan-cekungan kuning kecoklatan pada email & dentin

Sources 1. Beaudoin, D. Electrolytes and ion sensitive electrodes. PPT. 2003. 2. Hardjasasmita, P. 1993. Ikhtisar: biokimia dasar B. Balai Penerbit FKUI. Jakarta: 50 - 6. 3. Marieb, EN. Fluid, electrolyte, and acidbase balance. PPT. Pearson Education, Inc. 2004 38
- Slides: 38