ELECTROLYSIS OVERVIEW ANIMATION D C SOURCE BATTERY 3
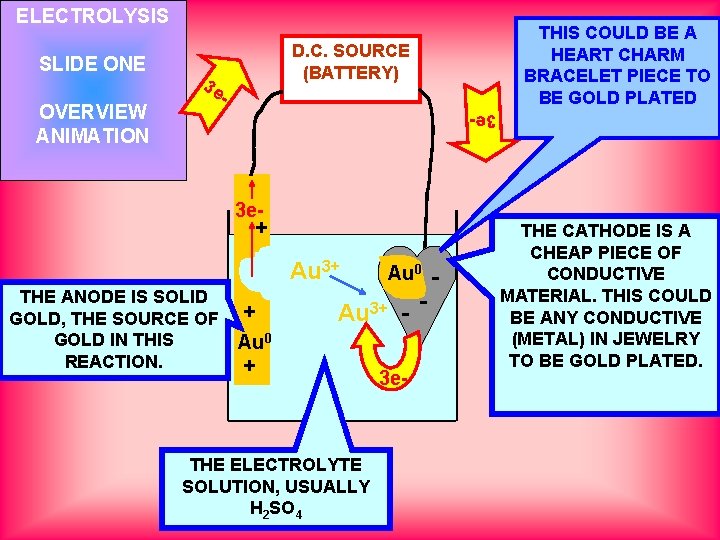
ELECTROLYSIS OVERVIEW ANIMATION D. C. SOURCE (BATTERY) 3 e 3 e- SLIDE ONE THIS COULD BE A HEART CHARM BRACELET PIECE TO BE GOLD PLATED 3 e+ Au THE ANODE IS SOLID GOLD, THE SOURCE OF GOLD IN THIS REACTION. + Au 0 + Au 3+ 0 -Au - Au 3+ - - THE ELECTROLYTE SOLUTION, USUALLY H 2 SO 4 3 e- THE CATHODE IS A CHEAP PIECE OF CONDUCTIVE MATERIAL. THIS COULD BE ANY CONDUCTIVE (METAL) IN JEWELRY TO BE GOLD PLATED.
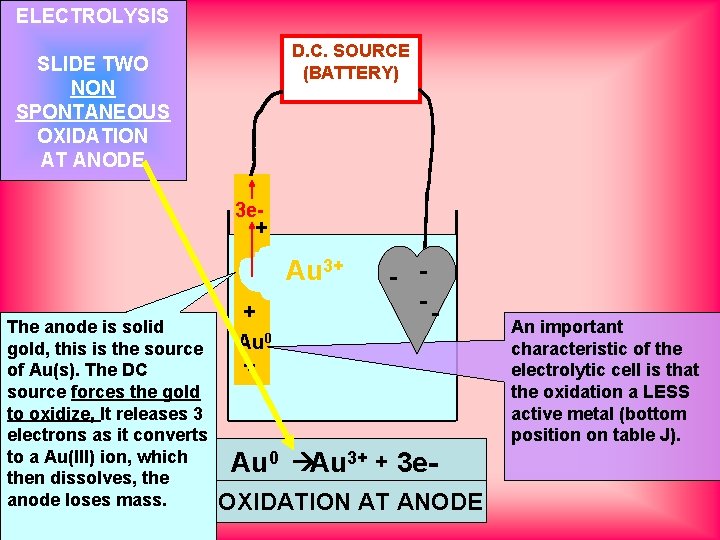
ELECTROLYSIS D. C. SOURCE (BATTERY) SLIDE TWO NON SPONTANEOUS OXIDATION AT ANODE 3 e+ Au The anode is solid gold, this is the source of Au(s). The DC source forces the gold to oxidize, It releases 3 electrons as it converts to a Au(III) ion, which then dissolves, the anode loses mass. + Au 0 + Au 3+ - -- Au 0 Au 3+ + 3 e. OXIDATION AT ANODE An important characteristic of the electrolytic cell is that the oxidation a LESS active metal (bottom position on table J).
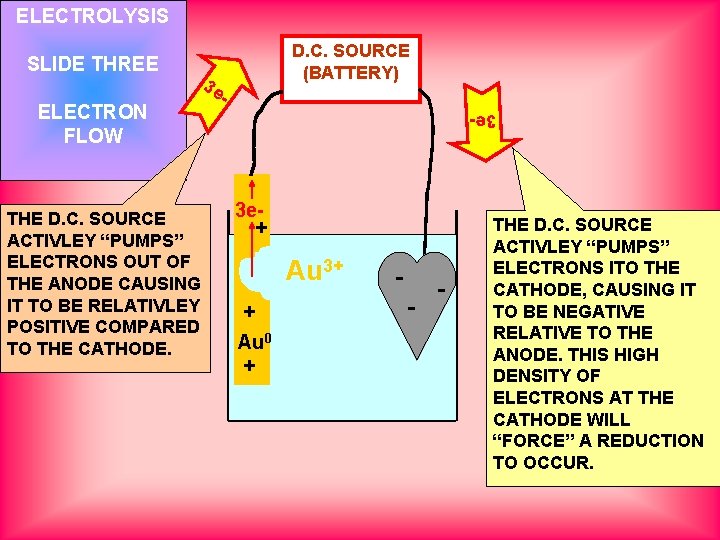
ELECTROLYSIS ELECTRON FLOW THE D. C. SOURCE ACTIVLEY “PUMPS” ELECTRONS OUT OF THE ANODE CAUSING IT TO BE RELATIVLEY POSITIVE COMPARED TO THE CATHODE. D. C. SOURCE (BATTERY) 3 e 3 e- SLIDE THREE 3 e+ Au 0 + Au 3+ -- - THE D. C. SOURCE ACTIVLEY “PUMPS” ELECTRONS ITO THE CATHODE, CAUSING IT TO BE NEGATIVE RELATIVE TO THE ANODE. THIS HIGH DENSITY OF ELECTRONS AT THE CATHODE WILL “FORCE” A REDUCTION TO OCCUR.
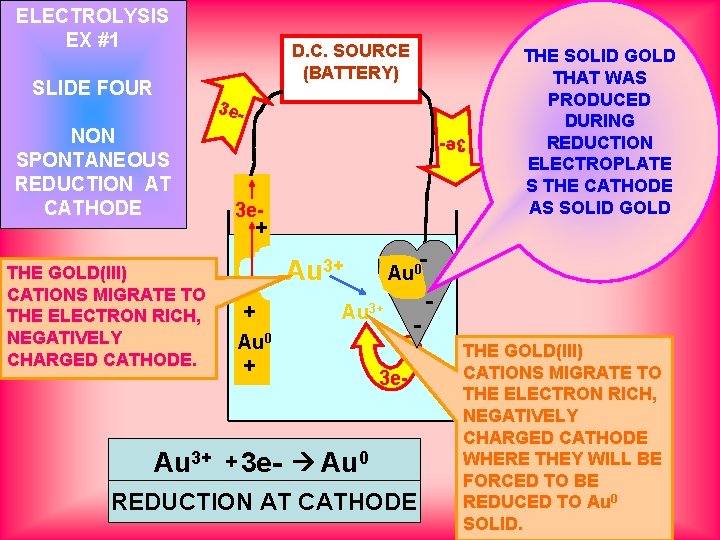
ELECTROLYSIS EX #1 SLIDE FOUR D. C. SOURCE (BATTERY) 3 e- THE GOLD(III) CATIONS MIGRATE TO THE ELECTRON RICH, NEGATIVELY CHARGED CATHODE. Au 3+ 3 e- NON SPONTANEOUS REDUCTION AT CATHODE 3 e+ Au 0 + + 3 e- THE SOLID GOLD THAT WAS PRODUCED DURING REDUCTION ELECTROPLATE S THE CATHODE AS SOLID GOLD - Au 3+ Au 0 Au 3+ - 3 e- Au 0 REDUCTION AT CATHODE THE GOLD(III) CATIONS MIGRATE TO THE ELECTRON RICH, NEGATIVELY CHARGED CATHODE WHERE THEY WILL BE FORCED TO BE REDUCED TO Au 0 SOLID.
- Slides: 4