Electrolysis Electrolysis Process by which an electric current

Electrolysis

Electrolysis �Process by which an electric current is passed through a substance to cause a chemical change. �The chemical change is one in which the substance loses or gains an electron.

Electrolysis �The process uses an apparatus consisting of positive and negative electrodes which are separated from each other in a solution. �Electric current enters through the negatively charged electrode (cathode). �Positively charged parts of the solution travel to the cathode, combine with the electrons, and are transformed into neutral molecules. �The negatively charged parts of the solution travel to the positive electrode (anode), give up electrons, and are transformed into neutral molecules.
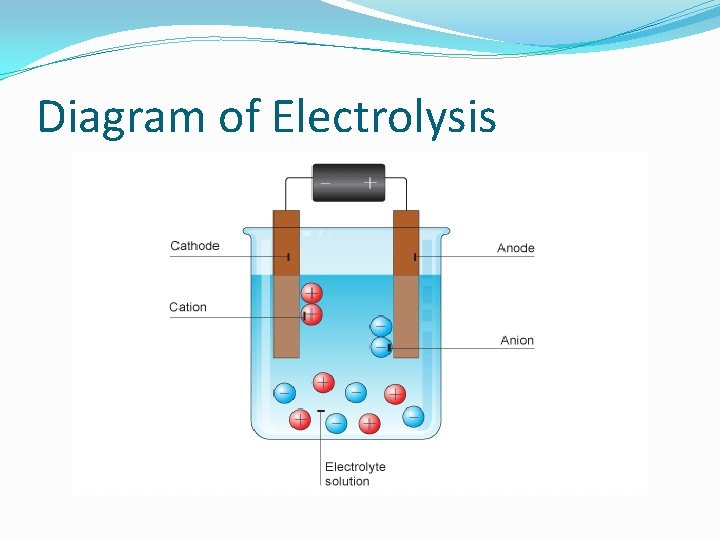
Diagram of Electrolysis

Electrolysis Apparatus
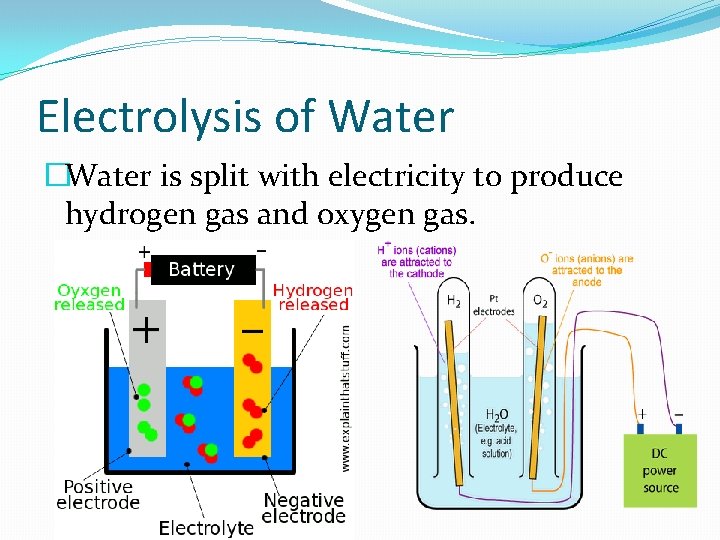
Electrolysis of Water �Water is split with electricity to produce hydrogen gas and oxygen gas.
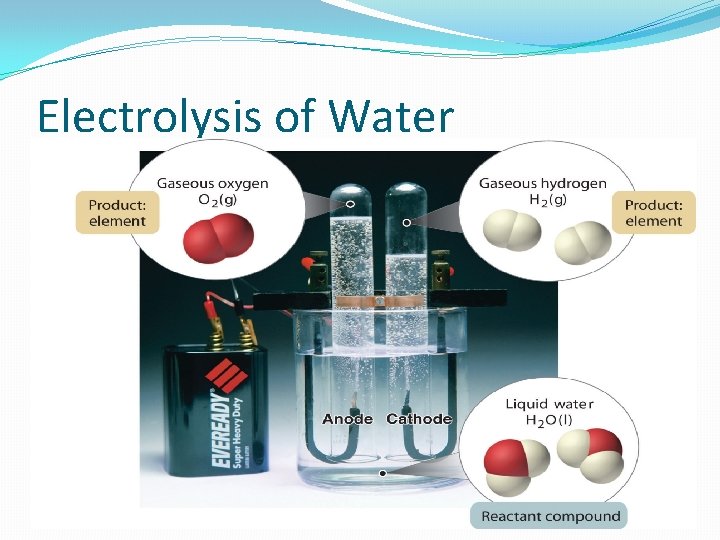
Electrolysis of Water

Electrolysis of Copper Chloride �Copper chloride is split with an electrical current to produce copper and chlorine gas. �During electrolysis, copper loses electrons, has an overall positive charge, and becomes a cation, which is attracted to the cathode or negative electrode. �Chlorine gains electrons, has an overall negative charge, and becomes an anion, which is attracted to the anode or positive electrode.

Electrolysis of Copper Chloride For more detail contact us
- Slides: 9