Electrochemistry The study of the interchange between chemical

Electrochemistry The study of the interchange between chemical and electrical energy

Oxidation-Reduction Reaction Review • “Redox” reactions involve the transfer of electrons (e-) • We use oxidation states to keep track of electron transfer • Some rules for assigning oxidation numbers (or states): • Oxidation numbers are roughly equivalent to hypothetical charge in compounds (there are some exceptions) • H is almost always +1 in compounds (Group 1 element) • O is almost always -2 in compounds (Group 6 element) • F is always -1 in compounds (Group 7 element) • For elements (H 2, O 2, F 2, Ca, K, Mn, etc. ) the oxidation state is always 0

Oxidation-Reduction Review Con’t • The overall charge of a compound = the sum of the oxidation states of all atoms in it – Neutral (i. e. H 2 O, CO 2, CH 4) • H 2 O : H = +1, O = -2 – The overall charge is 2(1) + -2 = 0 • CO 2: What is the oxidation state of C? – Since C + 2 (O) = 0 – C + 2(-2) = 0, thus C = +4 • CH 4: Is C still +4? – H is always +1 – Therefore to remain neutral, 4(1) + C = 0 – C must = -4

Oxidation-Reduction Review Con’t • Charged compounds (NO 3 -, CO 32 -, NH 4+) – NO 3 - or (NO 3)- : What is the oxidation # of N? • O is -2, and the overall charge is -1 • Thus, N + 3(O) = -1 or N + 3(-2) = -1 • N = +5 – (CO 3)2 -: What is the oxidation # of C? • O is -2, and the overall charge is -2 • Thus, C + 3(O) = -2 or C + 3(-2) = -2 • C = +4 – The oxidation # of charged atoms = charge • Mn 3+ has an oxidation # of +3 • S 2 - has an oxidation # of -2

Oxidation-Reduction Review Con’t • Try these…Mn. O 4 -, Cr 2 O 72 -, C 2 O 42 • (Mn. O 4)– O = -2, so [4(-2) + Mn = -1] – Mn = +7 • (Cr 2 O 7)2– O = -2, so [7(-2) + 2 Cr = -2] – 2 Cr = 12, therefore Cr = +6 • (C 2 O 4)2– O = -2, so [2 C + 4(-2) = -2] – 2 C = 6, therefore C = +3

Assigning Oxidation # Practice • Assign oxidation numbers to each atom – Cl 2 – Fe 2+ – Cl. O 3– Cl. O 4– IO 2– Cr. O 42– Fe 3(PO 4)2 – Co. SO 4 – H 2 CO 3

Assigning Oxidation # Practice • Assign oxidation numbers to each atom – Cl 2: element → oxidation # = 0 – Fe 2+ ion → oxidation # same as charge… +2 – Cl. O 3 - : O = -2, Cl = +5 (Cl is usually -1) – Cl. O 4 - : O = -2, Cl = +7 – IO 2 - : O = -2, I = +3 – Cr. O 42 - : O = -2, Cr = +6 – Fe 3(PO 4)2 : O = -2, P = +5, Fe = +2 – Co. SO 4 : O = -2, S = +6, Co = +2 – H 2 CO 3 : O = -2, C = +4, H = +1

Oxidation-Reduction Reactions • Two separate reactions occurring simultaneously – Oxidation: oxidation # of an atom increases • i. e. Fe(s) → Fe 3+(aq) oxidation state goes from 0 → +3 – Reduction: oxidation # of an atom is “reduced” • i. e. O 2 → O 2 - (oxidation state goes from 0 → -2) – When occurring together… • Fe + O 2 → Fe 3+ + O 2 • This common redox reaction is the formation of rust • But, how do we balance this?

Balancing by Half-Reactions *in acidic solution CH 3 OH (aq) + Cr 2 O 72 -(aq) → CH 2 O(aq) + Cr 3+(aq) 1. Assign oxidation states to all atoms within the reaction C-2 H 4+O 2 - + (Cr 26+O 72 -)2 - → C 0 H 2+O 2 - + Cr 3+ 2. Determine which atom is being reduced and which is oxidized and write separate reactions Ox: C-2 H 4+O 2 - → C 0 H 2+O 2 - (C is going from -2 to 0) Red: (Cr 26+O 72 -)2 - → Cr 3+ (Cr is being reduced from +6 to +3)

Balancing by Half-Reactions *in acidic solution 3. For each half-reaction: • Disregard all oxidation states except those that are changing (all others will cancel each other out) C-2 H 4+O 2 - → C 0 H 2+O 2 - into C-2 H 4 O → C 0 H 2 O • Balance elements (except H and O) by using coefficients (like normal) C-2 H 4 O → C 0 H 2 O Carbon is already balanced • Balance O’s by adding H 2 O to either side C-2 H 4 O → C 0 H 2 O Oxygen is already balanced

Balancing by Half-Reactions *in acidic solution 3. For each half-reaction: • Balance H’s by adding H+ to either side (in acidic solution) – Where do we add H+? C-2 H 4 O → C 0 H 2 O + 2 H+ Double check that all atoms are balanced • Balance charge by adding electrons to either side – Remember that electrons are negative charges – Use changing oxidation state as a guide

C-2 H 4 O → C 0 H 2 O + 2 H+ • Now that all elements are balanced, we have to balance the charges • Balance charges by adding e- to either side – All charges stay the same, except C – Therefore add e- to make the charge equal on each side – C-2 H 4 O → C 0 H 2 O + 2 H+ + 2 e-2 charge = -2 charge ≠ 0 Now we have to use the same procedure on the reduction reaction!!!!!!! I’ll bet you just can’t wait!!!!!

Time to balance the reduction half • Reduction half reaction: (Cr 26+O 72 -)2 - → Cr 3+ • Disregard all other oxidation states (the ones that aren’t changing): (Cr 26+O 7)2 - → Cr 3+ • Balance all elements except H and O (Cr 26+O 7)2 - → 2 Cr 3+ • Balance O by adding H 2 O if necessary (Cr 26+O 7)2 - → 2 Cr 3+ + 7 H 2 O • Balance H by adding H+ if necessary 14 H+ + (Cr 26+O 7)2 - → 2 Cr 3+ + 7 H 2 O • Balance charge by adding e 6 e- + 14 H+ + (Cr 26+O 7)2 - → 2 Cr 3+ + 7 H 2 O

Adding Half-Reactions *in acidic solution Now that we have balanced each half rxn, we need to add them together to get the full rxn Ox: C-2 H 4 O → C 0 H 2 O + 2 H+ + 2 e. Red: 6 e- + 14 H+ + (Cr 26+O 7)2 - → 2 Cr 3+ + 7 H 2 O 4. In order to add the rxn’s, e- must be equal So, multiply rxn’s by integers to equalize electrons 3 (C-2 H 4 O → C 0 H 2 O + 2 H+ + 2 e- ) 6 e- + 14 H+ + (Cr 26+O 7)2 - → 2 Cr 3+ + 7 H 2 O 3 C-2 H 4 O → 3 C 0 H 2 O + 6 H+ + 6 e-

Let the adding begin… 6 e- + 14 H+ + (Cr 26+O 7)2 - → 2 Cr 3+ + 7 H 2 O 3 C-2 H 4 O → 3 C 0 H 2 O + 6 H+ + 6 e 3 CH 4 O + 8 H+ + Cr 2 O 72 - → 3 CH 2 O + 2 Cr 3+ + 7 H 2 O …and the reaction is now balanced! 5. Double check that all atoms and charges are balanced

Balancing by Half-Reactions (in basic solution) 1. Write oxidation and reduction half-reactions 2. Balance each half-reaction • Balance elements except for O, H • Balance O by adding H 2 O • Balance H by adding H+ • Add enough OH- to both sides to cancel out any H+ and form H 2 O • • Recall that H+ + OH- → H 2 O Balance charge by adding electrons 3. Equalize electrons and add half-reactions 4. Check elements and charges for balance

Balancing by Half-Reactions (in basic solution) • Let’s balance a previous example in basic solution – Remember, it is all the same steps up to this point 14 H+ + (Cr 26+O 7)2 - → 2 Cr 3+ + 7 H 2 O + 14 OH 14 H 2 O + (Cr 26+O 7)2 - → 2 Cr 3+ + 7 H 2 O + 14 OH- 7 H 2 O + (Cr 26+O 7)2 - → 2 Cr 3+ + 14 OHNow, continue as you normally would…

Practice Balancing Redox Reactions Unbalanced reaction (in acid): Mn. O 4 + Fe 2+ Mn 2+ + Fe 3+ Balanced Reduction half-reaction: 8 H+ + Mn. O 4 + 5 e Mn 2+ + 4 H 2 O Balanced Oxidation half-reaction: 5( Fe 2+ Fe 3+ + e ) Balanced overall reaction: 8 H+ + Mn. O 4 + 5 Fe 2+ Mn 2+ + 5 Fe 3+ + 4 H 2 O

Redox Vocabulary Mn. O 4 + Fe 2+ Mn 2+ + Fe 3+ • Oxidized species (atom, ion, molecule, or compound) – whichever species is being oxidized (↑ oxidation #) • Reduced species – whichever species is being reduced (↓ oxidation #) • Oxidizing agent – Whichever species CAUSES oxidation to occur—in other words, the oxidizing agent IS the reduced species • Reducing agent – Whichever species CAUSES reduction to occur—in other words, the reducing agent IS the oxidized species
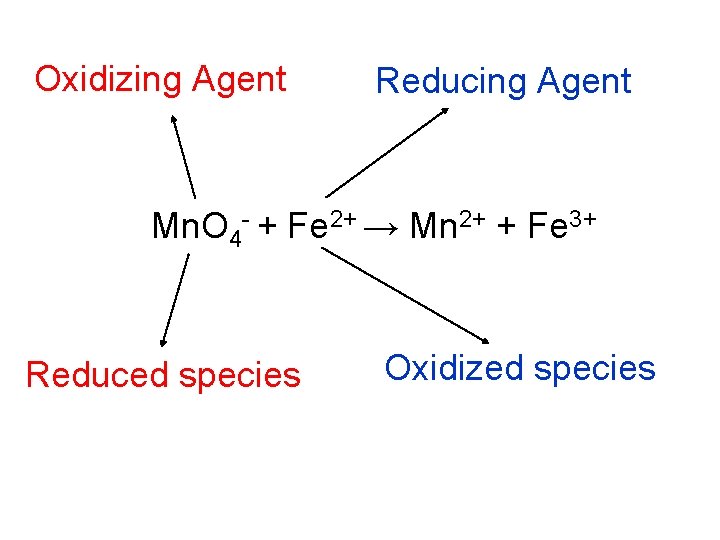
Oxidizing Agent Reducing Agent Mn. O 4 - + Fe 2+ → Mn 2+ + Fe 3+ Reduced species Oxidized species
- Slides: 20