ELECTROCHEMISTRY Electrochemistry the study of the exchange between

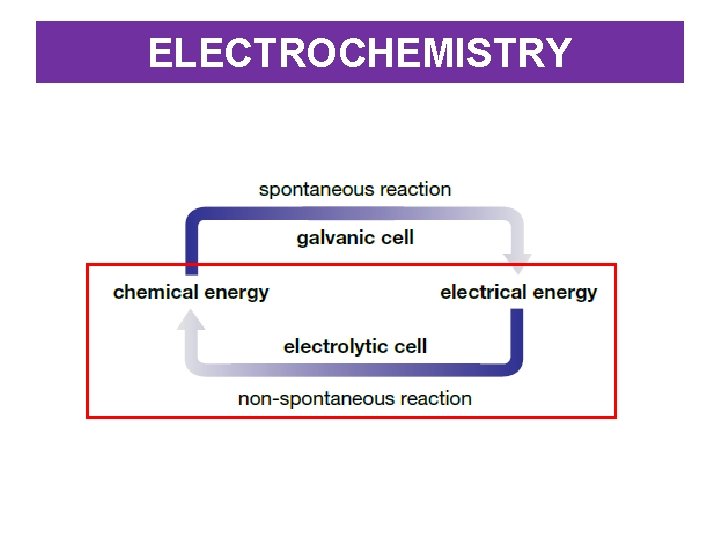
ELECTROCHEMISTRY • Electrochemistry: the study of the exchange between electrical energy and chemical energy • Chemical energy can be used to generate electrical energy. – Electrical energy (electricity) is the movement of electrons. The movement of electrons (also called the current) can be harnessed to do work. – This is how batteries operate – chemicals stored inside the battery undergo a chemical reaction causing electrons to move, producing electrical energy which can be used to power something (torch, radio, phone etc).

ELECTROCHEMISTRY • In redox reactions, electrons move from one atom to another. – We can use this movement of electrons to generate electricity by separating the reactants and forcing the electrons to move through a wire, which is how we generate the electrical energy. • Electrochemical cell: a device that converts chemical energy into electrical energy – Two types of electrochemical cell: voltaic cell and electrolytic cell.

VOLTAIC CELLS (Galvanic Cells) • Voltaic Cell: a type of electrochemical cell where electrical energy (movement of electrons) is generated from a spontaneous chemical reaction. – The type of chemical reaction is a redox reaction
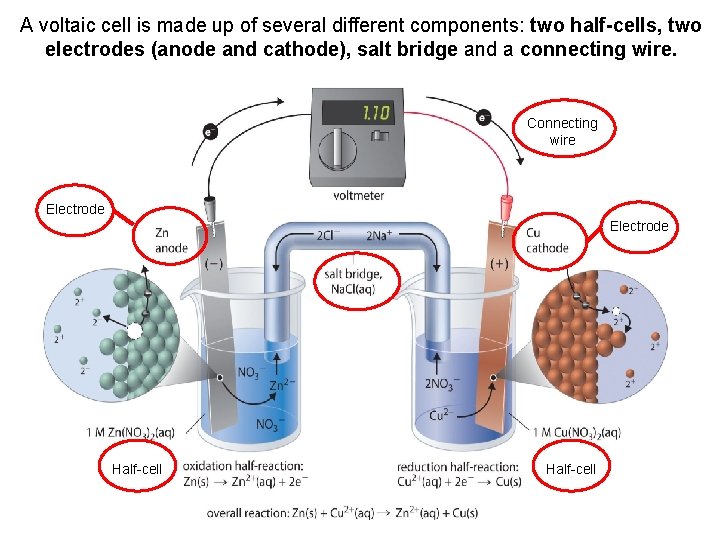
A voltaic cell is made up of several different components: two half-cells, two electrodes (anode and cathode), salt bridge and a connecting wire. Connecting wire Electrode Half-cell

EXAMPLE: VOLTAIC CELLS • Zn and Cu 2+ can undergo a redox reaction. We can use the movement of electrons between Zn and Cu 2+ to generate electricity Reduction Zn + Cu 2+ → Zn 2+ + Cu Oxidation: Zn → Zn 2+ + 2 e. Reduction: Cu 2+ + 2 e- → Cu
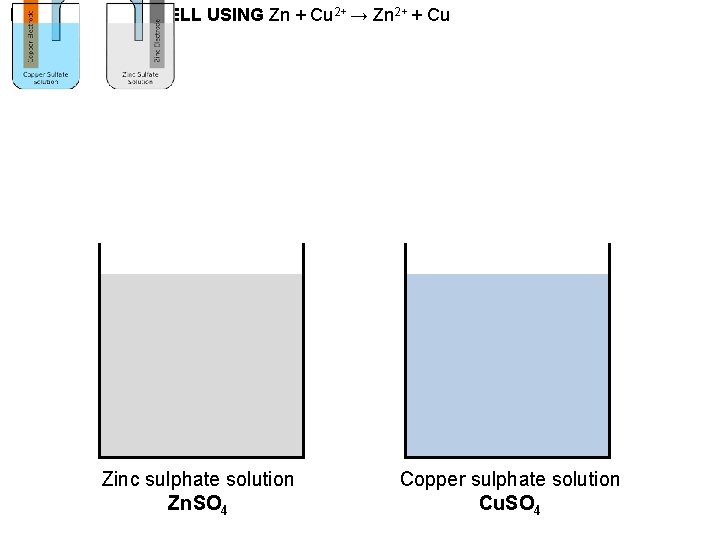
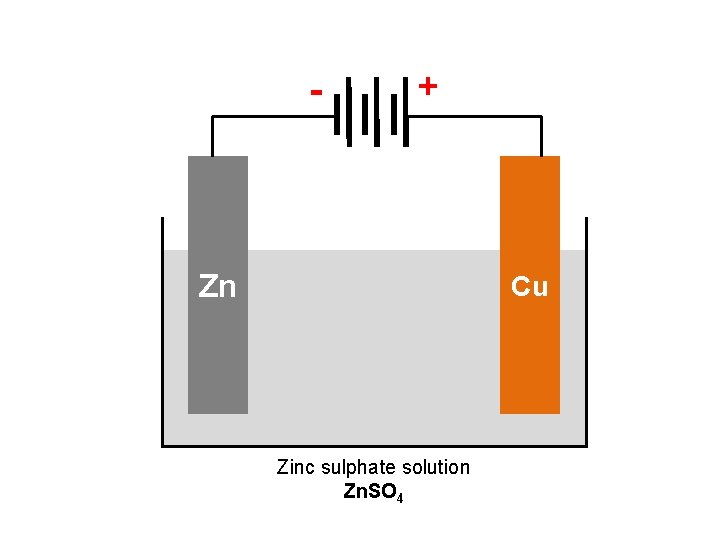
BASIC VOLTAIC CELL USING Zn + Cu 2+ → Zn 2+ + Cu Zinc sulphate solution Zn. SO 4 Copper sulphate solution Cu. SO 4
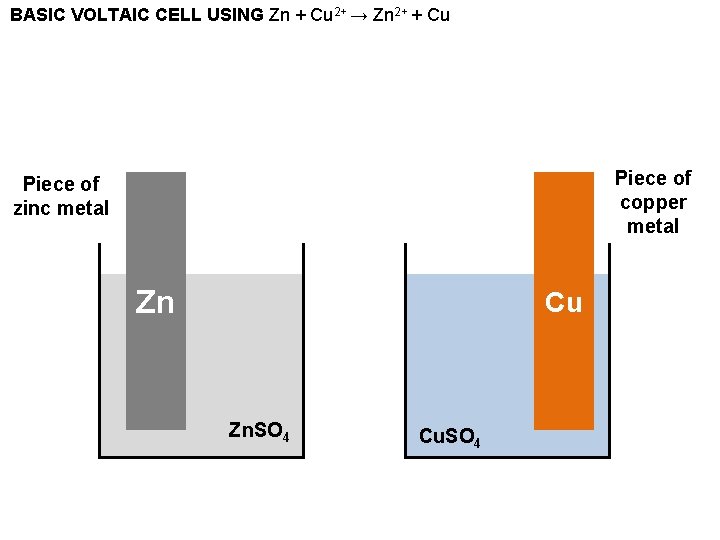
BASIC VOLTAIC CELL USING Zn + Cu 2+ → Zn 2+ + Cu Piece of copper metal Piece of zinc metal Zn Cu Zn. SO 4 Cu. SO 4
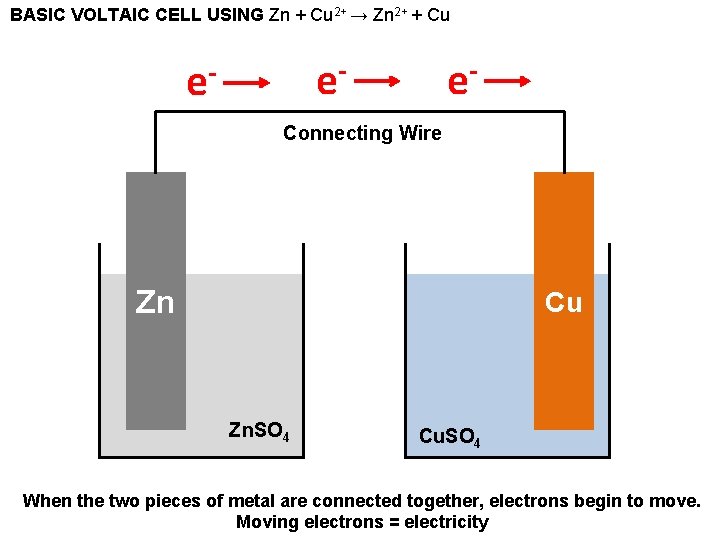
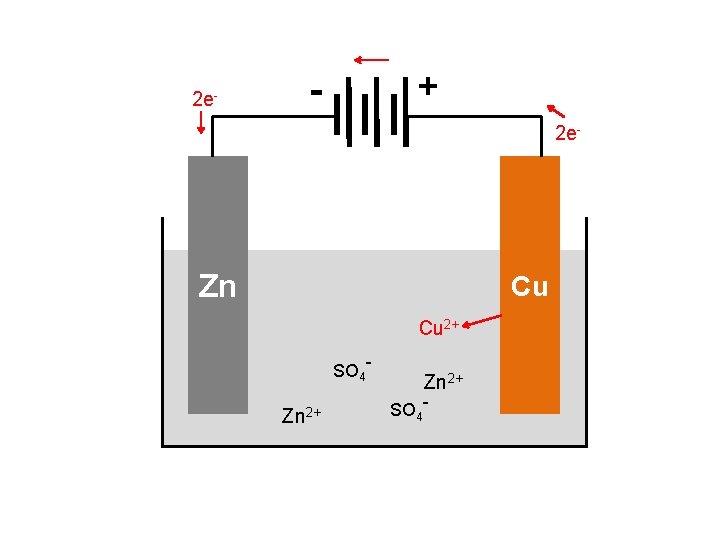
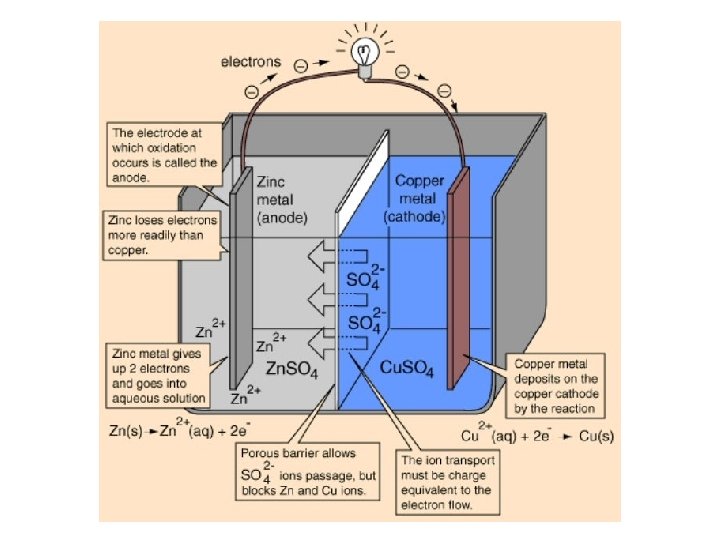
BASIC VOLTAIC CELL USING Zn + Cu 2+ → Zn 2+ + Cu e- e- e- Connecting Wire Zn Cu Zn. SO 4 Cu. SO 4 When the two pieces of metal are connected together, electrons begin to move. Moving electrons = electricity
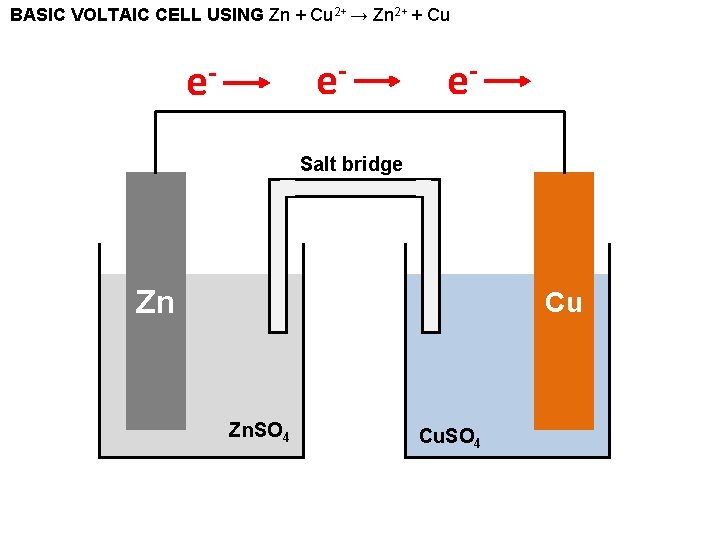
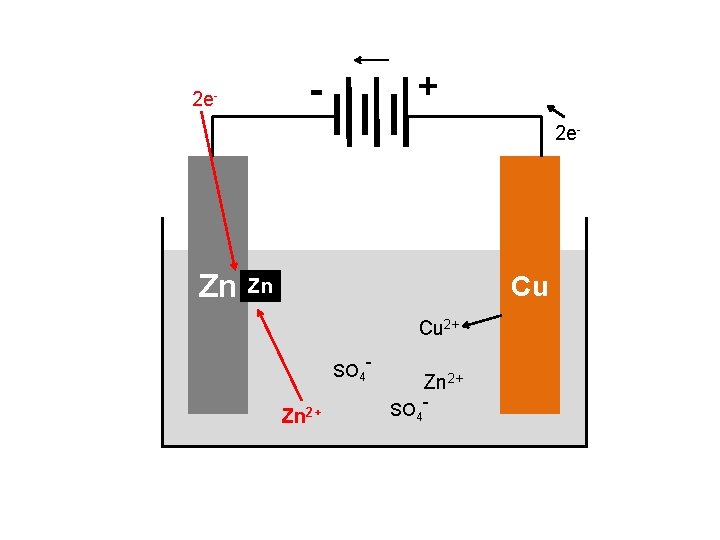
BASIC VOLTAIC CELL USING Zn + Cu 2+ → Zn 2+ + Cu e- e- e- Salt bridge Zn Cu Zn. SO 4 Cu. SO 4
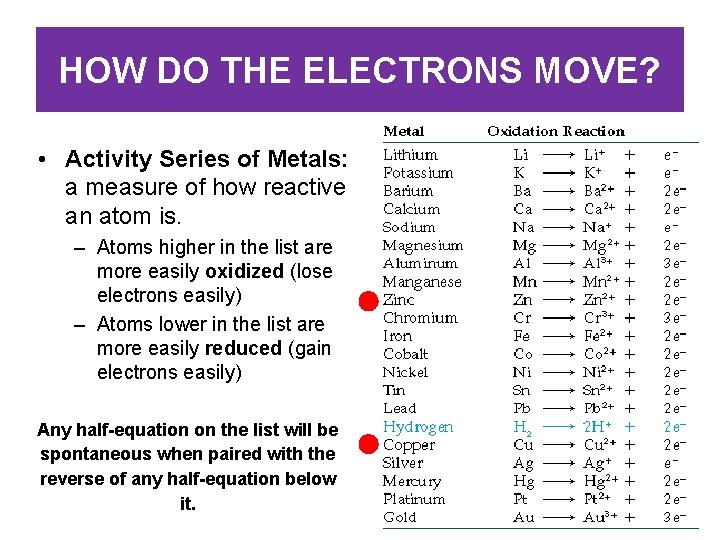
HOW DO THE ELECTRONS MOVE? • Activity Series of Metals: a measure of how reactive an atom is. – Atoms higher in the list are more easily oxidized (lose electrons easily) – Atoms lower in the list are more easily reduced (gain electrons easily) Any half-equation on the list will be spontaneous when paired with the reverse of any half-equation below it.

DEFINITIONS TO KNOW • Cathode: the site where reduction is occurring • Anode: the site where oxidation is occurring – Remember using “An Ox, Red Cat” – Electrons always flow from the anode to the cathode • Half-cell: one half of the voltaic cell. Each voltaic cell is made up of two half-cells – an oxidation half -cell and a reduction half-cell. • Salt Bridge: connects the two half-cells together and balances the charge between them.
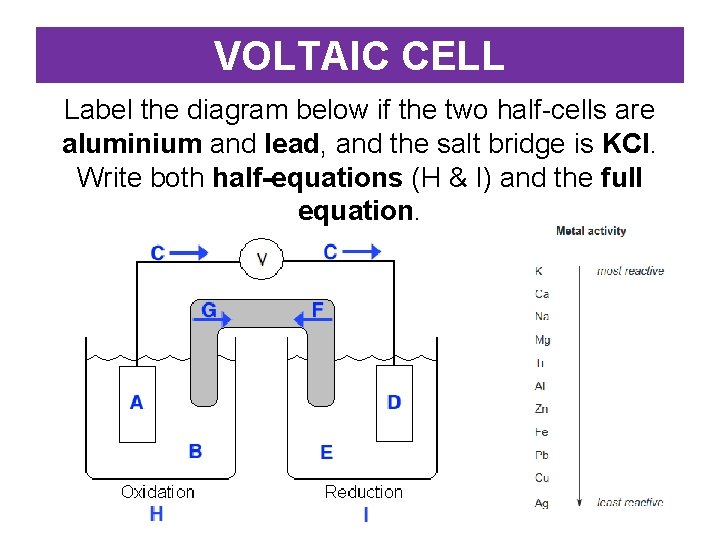
VOLTAIC CELL Label the diagram below if the two half-cells are aluminium and lead, and the salt bridge is KCl. Write both half-equations (H & I) and the full equation.
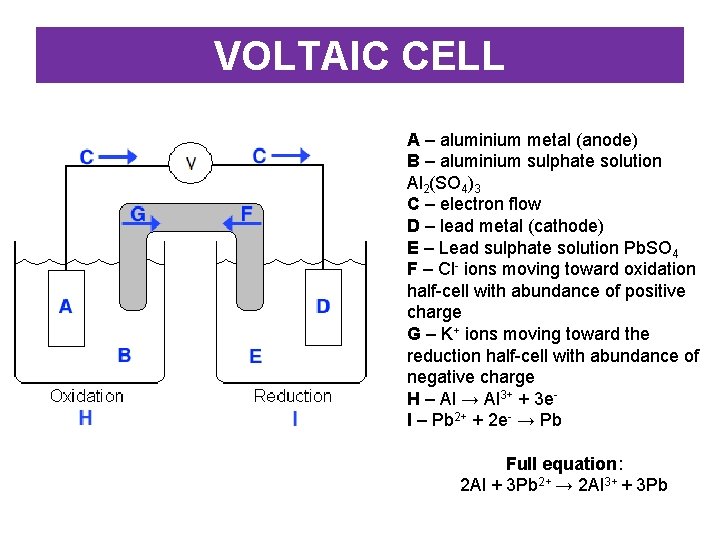
VOLTAIC CELL A – aluminium metal (anode) B – aluminium sulphate solution Al 2(SO 4)3 C – electron flow D – lead metal (cathode) E – Lead sulphate solution Pb. SO 4 F – Cl- ions moving toward oxidation half-cell with abundance of positive charge G – K+ ions moving toward the reduction half-cell with abundance of negative charge H – Al → Al 3+ + 3 e. I – Pb 2+ + 2 e- → Pb Full equation: 2 Al + 3 Pb 2+ → 2 Al 3+ + 3 Pb
ELECTROCHEMISTRY

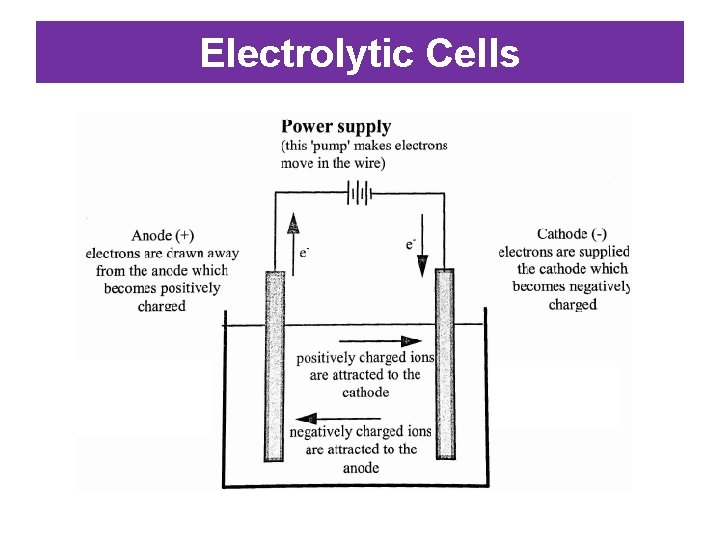
Electrolytic Cells • Electrolytic Cell: an electrochemical cell in which an electrical energy is used to drive a non-spontaneous reaction (doesn’t happen on its own) – The process of using electrical energy to drive a nonspontaneous reaction is called electrolysis. • An electrolytic cell typically contains: – An electrolyte solution that takes part in the reaction – Two electrodes where the electrolysis reactions occur – An external source of electrical energy (eg battery)
Electrolytic Cells
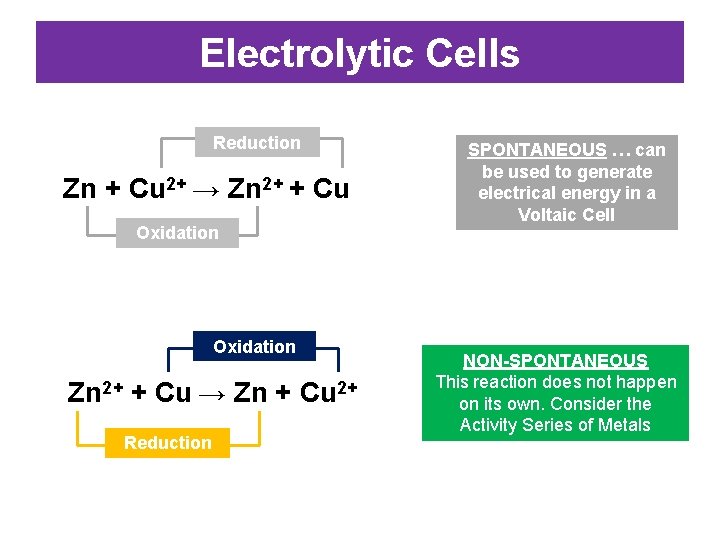
Electrolytic Cells Reduction Zn + Cu 2+ → Zn 2+ + Cu Oxidation Zn 2+ + Cu → Zn + Cu 2+ Reduction SPONTANEOUS … can be used to generate electrical energy in a Voltaic Cell NON-SPONTANEOUS This reaction does not happen on its own. Consider the Activity Series of Metals
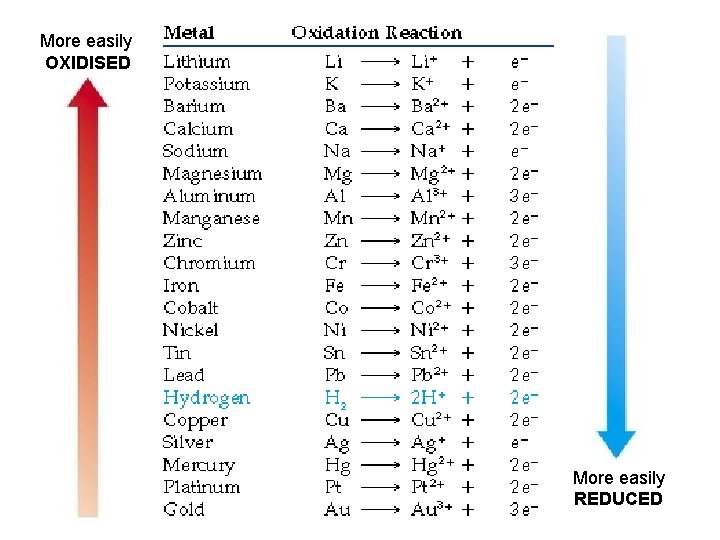
More easily OXIDISED More easily REDUCED
- + Zn Cu Zinc sulphate solution Zn. SO 4
2 e- + - 2 e- Zn Cu Cu 2+ SO 4 - Zn 2+ SO 4 -
+ - 2 e- Zn Cu 2+ SO 4 - Zn 2+ SO 4 -
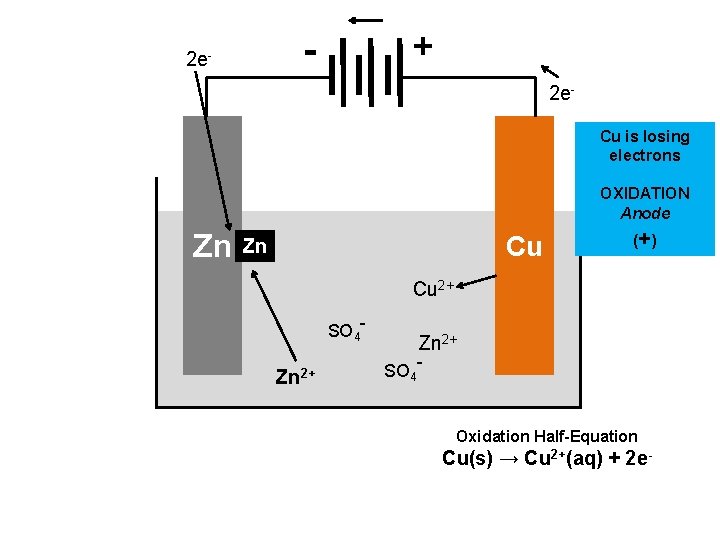
+ - 2 e. Cu is losing electrons OXIDATION Anode Zn Cu Zn (+ ) Cu 2+ SO 4 - Zn 2+ SO 4 - Oxidation Half-Equation Cu(s) → Cu 2+(aq) + 2 e-
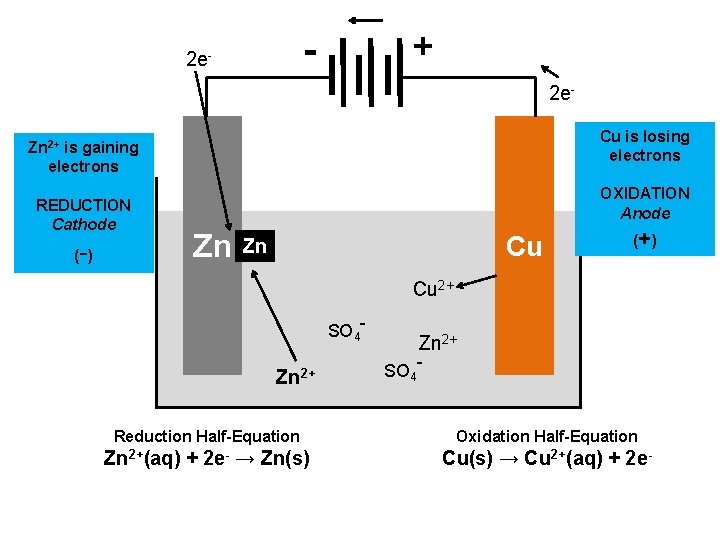
+ - 2 e. Cu is losing electrons Zn 2+ is gaining electrons REDUCTION Cathode - ( ) OXIDATION Anode Zn Cu Zn (+ ) Cu 2+ SO 4 - Zn 2+ SO 4 - Reduction Half-Equation Oxidation Half-Equation Zn 2+(aq) + 2 e- → Zn(s) Cu(s) → Cu 2+(aq) + 2 e-
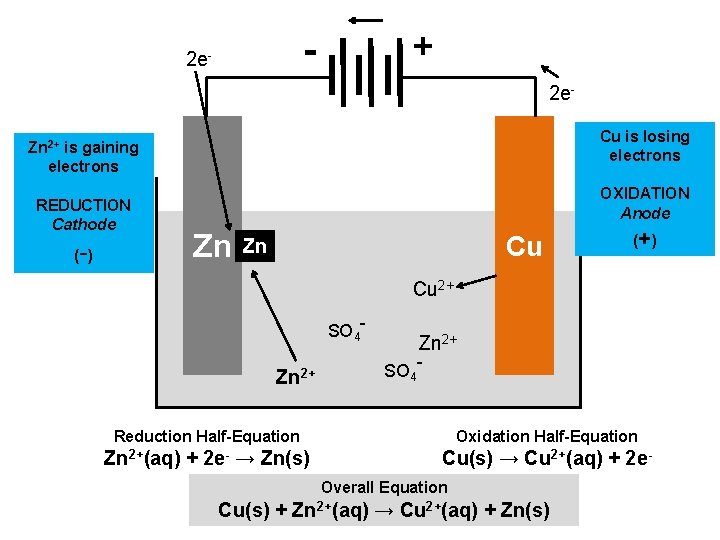
+ - 2 e. Cu is losing electrons Zn 2+ is gaining electrons REDUCTION Cathode - ( ) OXIDATION Anode Zn Cu Zn (+ ) Cu 2+ SO 4 - Zn 2+ SO 4 - Reduction Half-Equation Oxidation Half-Equation Zn 2+(aq) + 2 e- → Zn(s) Cu(s) → Cu 2+(aq) + 2 e. Overall Equation Cu(s) + Zn 2+(aq) → Cu 2+(aq) + Zn(s)

ELECTROLYTIC CELL Draw a diagram for an electrolytic cell that drives the following non-spontaneous reaction: Cu + Mg 2+ → Cu 2+ + Mg Label the anode, cathode, electrolytic solution, direction of electron flow, halfequation occurring at each electrode
Electrolytic vs Voltaic Cells Fill in the table below to compare the similarities and differences between Electrolytic and Voltaic Cells Voltaic Cell Type of redox reaction Type of energy change Energy produced or required Where oxidation occurs Where reduction occurs Negative terminal Positive terminal Flow of electrons Structure Electrolytic Cell
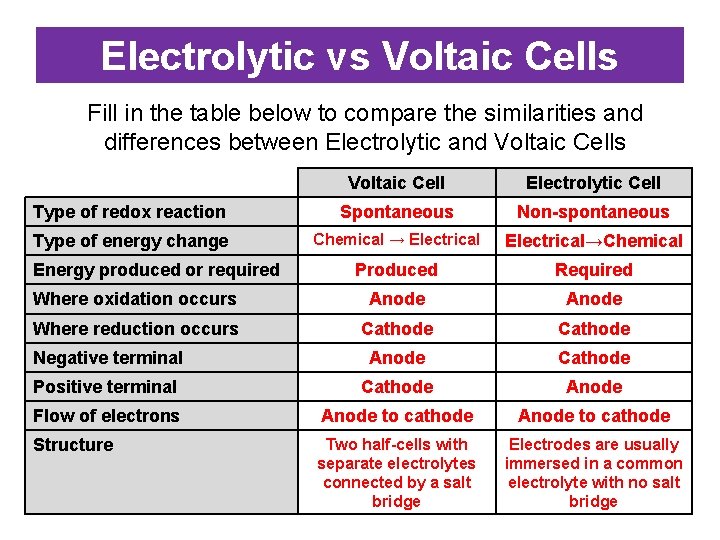
Electrolytic vs Voltaic Cells Fill in the table below to compare the similarities and differences between Electrolytic and Voltaic Cells Voltaic Cell Electrolytic Cell Type of redox reaction Spontaneous Non-spontaneous Type of energy change Chemical → Electrical→Chemical Produced Required Where oxidation occurs Anode Where reduction occurs Cathode Negative terminal Anode Cathode Positive terminal Cathode Anode Flow of electrons Anode to cathode Structure Two half-cells with separate electrolytes connected by a salt bridge Electrodes are usually immersed in a common electrolyte with no salt bridge Energy produced or required

Application of Electrolysis • The most common application of electrolytic cells is the rechargeable battery. – While the battery is being used in a device it is a voltaic cell function (using the redox reaction to produce electricity). – While the battery is charging it is an electrolytic cell function (using outside electricity to reverse the completed redox reaction).
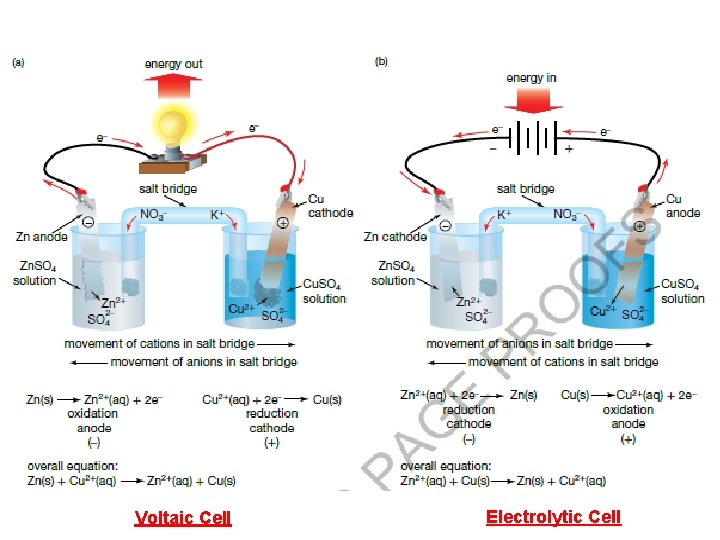
Voltaic Cell Electrolytic Cell
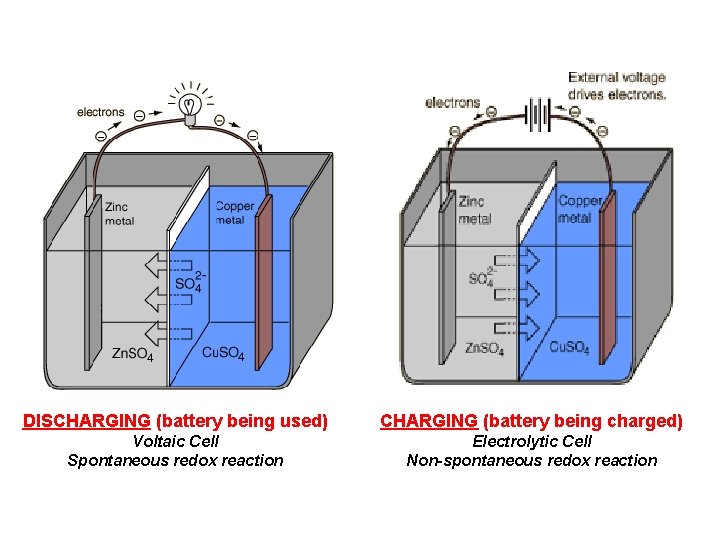
DISCHARGING (battery being used) CHARGING (battery being charged) Voltaic Cell Spontaneous redox reaction Electrolytic Cell Non-spontaneous redox reaction

Application of Electrolysis • Electrolysis can be used to separate a compound into the elements that it is made up of. • Example: electrolysis of sodium chloride Na. Cl → Na(s) + Cl 2(g) This is a non-spontaneous reaction.
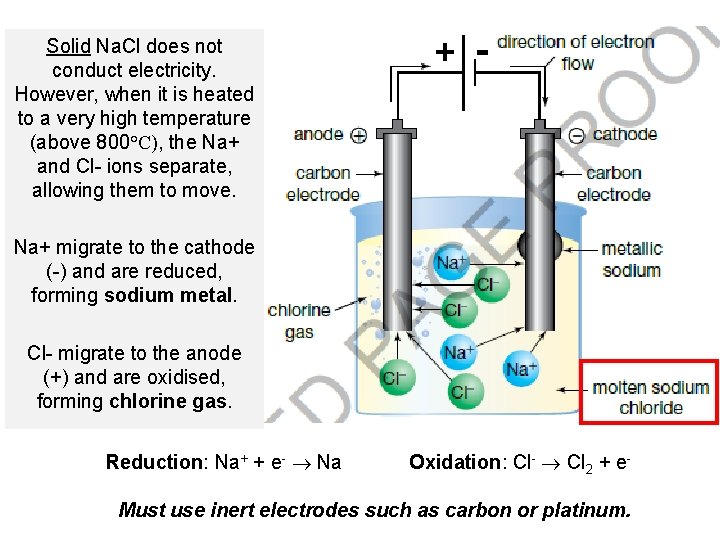
Solid Na. Cl does not conduct electricity. However, when it is heated to a very high temperature (above 800°C), the Na+ and Cl- ions separate, allowing them to move. + - Na+ migrate to the cathode (-) and are reduced, forming sodium metal. Cl- migrate to the anode (+) and are oxidised, forming chlorine gas. Reduction: Na+ + e- Na Oxidation: Cl- Cl 2 + e- Must use inert electrodes such as carbon or platinum.
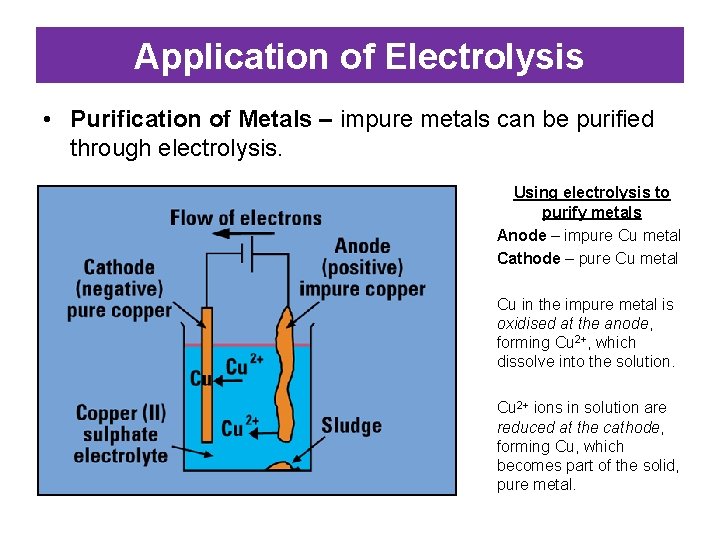
Application of Electrolysis • Purification of Metals – impure metals can be purified through electrolysis. Using electrolysis to purify metals Anode – impure Cu metal Cathode – pure Cu metal Cu in the impure metal is oxidised at the anode, forming Cu 2+, which dissolve into the solution. Cu 2+ ions in solution are reduced at the cathode, forming Cu, which becomes part of the solid, pure metal.

Application of Electrolysis • Metal Electroplating: depositing a thin layer of metal onto the surface of another metal. – This may be either for decorative purposes or to prevent corrosion • When electroplating: – Cathode – the metallic object to be electroplated. – Anode – a strip of the plating metal. – Electrolyte – a solution of the salt of the plating metal.
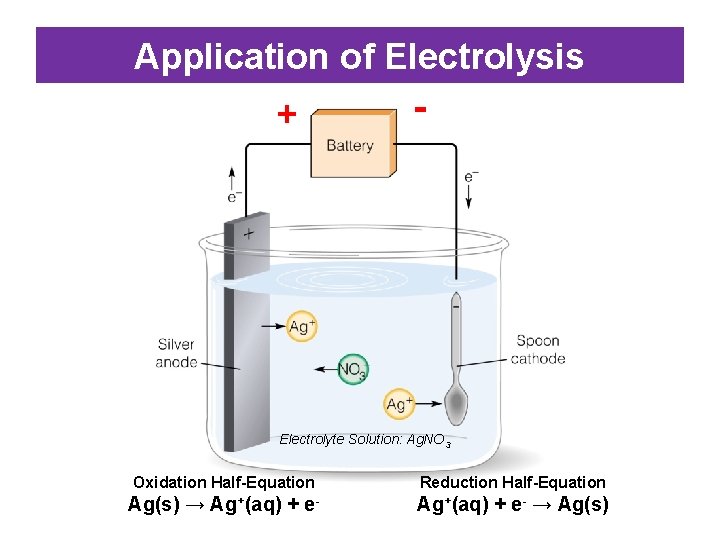
Application of Electrolysis + - Electrolyte Solution: Ag. NO 3 Oxidation Half-Equation Reduction Half-Equation Ag(s) → Ag+(aq) + e- → Ag(s)

Electroplating Practice Question The electroplating of a nickel knife with silver is an electrolytic process. Draw a diagram to show the: • Anode • Cathode • Electrolyte • Charge on electrodes • Direction of electron flow • Movement of silver ions in electrolyte • Anode half-equation • Cathode half-equation

Molten Electrolysis Practice Question During the electrolysis of molten potassium iodide using graphite electrodes, potassium metal is produced at that cathode and iodine gas at the anode. 1. 2. 3. 4. 5. Draw and label a diagram of the electrolytic cell. Write an equation for the reaction at each electrode. State why graphite (carbon) electrodes are used. Write the full equation for the reaction. State at which electrode oxidation occurs.
- Slides: 38