Electrochemistry Chapter 20 Oxidation State and Oxidation Reduction





















- Slides: 26

Electrochemistry Chapter 20

Oxidation State and Oxidation. Reduction Reactions Section 20. 1 n Redox reactions are one of the reaction types covered in Ch. 4 ¡ ¡ n For the rules associated with assigning oxidation numbers see Pg. 137 Analogous to acid-base reactions Oxidant Reductant Ex: Zn(s) + 2 H+(aq) → Zn 2+(aq) + H 2(g)

Identifying Oxidizing and Reducing Agents Determine the oxidation numbers of the elements in the following reaction. Identify the oxidant and reductant. 5 H 2 C 2 O 4(aq) + 6 H 3 O+(aq) + 2 Mn. O 4 -(aq) → 10 CO 2(g) + 2 Mn+(aq) + 14 H 2 O(l) See Sample Exercise 20. 1 (Pg. 845)

Balancing Oxidation-Reduction Reactions Section 20. 2 n When balancing typical chemical reactions, mass must be conserved ¡ n However, charge must be conserved as well Redox reactions must be broken up into two half reactions which must be balanced separately ¡ Two half reactions: One for oxidation; one for reduction

Balancing Redox Reactions (cont. ) Two types of solutions: acidic and basic n ¡ Divide equation into two half reactions Balance each half reaction 1. 2. 3. 4. 3. 4. Rules are only slightly different All elements other than O and H Balance O by adding H 2 O Balance H by adding H+ Balance charge by adding electrons, e- Multiply each half reactions with integer values in order to balance the # of electrons Combine both half reactions

Redox Reactions in Acidic Solution Balance the following redox reaction which takes place in acidic solution: Fe 2+(aq) + Cr 2 O 72 -(aq) → Cr 3+(aq) + Fe 3+(aq) See Sample Exercise 20. 2 (Pg. 849)

Redox Reactions in Basic Solution n Exactly the same as acidic solution ¡ Only difference is that OH- must be used to “neutralize” H+ added to balance H’s n n No appreciable amount of H+ can exist in basic solution Ex: Mn. O 4 -(aq) + Br-(aq) → Mn. O 2(s) + Br. O 3 -(aq)

Voltaic Cells Section 20. 3 n Spontaneous redox reactions are used to perform electrical work using a voltaic cell ¡ Voltaic cells (sometimes called galvanic cells) require an oxidant and reductant and some sort of medium for the electrons to travel through in order to produce current

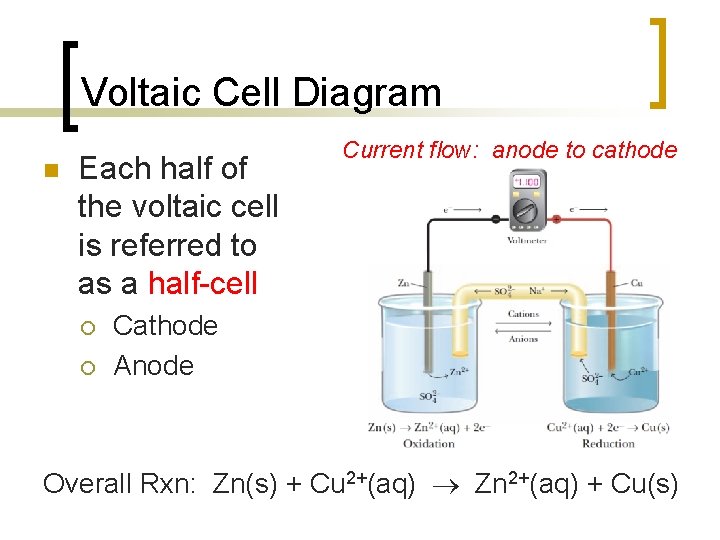
Voltaic Cell Diagram n Each half of the voltaic cell is referred to as a half-cell ¡ ¡ Current flow: anode to cathode Cathode Anode Overall Rxn: Zn(s) + Cu 2+(aq) Zn 2+(aq) + Cu(s)

Describing a Voltaic Cell Consider the cell shown below. a. ) Write the half-reaction that occurs in each half cell b. ) Write the equation for the overall chemical change taking place c. ) Identify cathode and anode d. ) Identify the signs of the electrodes e. ) Indicate the direction that the nitrate ions flow through the salt bridge See Sample Exercise 20. 4 (Pg. 853)

Cell EMF Under Standard Conditions Section 20. 4 n In a voltaic cell, there is a potential difference between the two electrodes (electromotive force or cell potential) ¡ ¡ For a spontaneous reaction Ecell will have a positive value Under standard conditions (1 M or 1 atm) it is referred to as standard cell potential, E cell

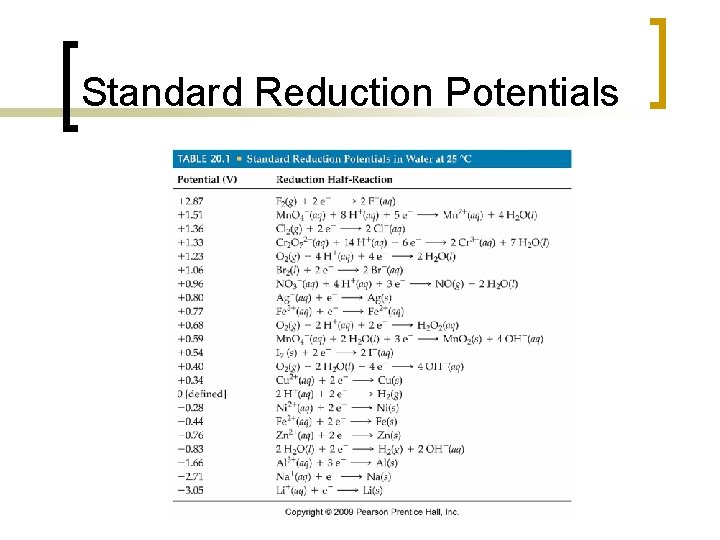
Standard Reduction Potentials n n In theory, we could experimentally determine the Ecell values for every possible redox combination (dumb idea—too much work) More practically, we can determine the voltage required for reduction to occur at an electrode ¡ Referred to as standard reduction potentials and are referenced to the standard hydrogen electrode (SHE) n E red = 0

Calculating Ecell n The cell potential, E cell, can be calculated using tabulated standard reduction potentials: E cell = E red(cathode) - E red(anode) n For all reactions, the reaction is spontaneous if E cell is positive ¡ The more positive the value, the more driving force there is for the reaction to occur

Standard Reduction Potentials

Calculating E cell from Standard Reduction Potentials Calculate the standard potential and See Sample state the direction in which the Exercise reaction proceeds spontaneously for: 20. 6 (Pg. 858) 2 Cr+3(aq) + 2 Br-(aq) → 2 Cr+2(aq) + Br 2(l)

Determining Half-Reactions at Electrodes and Calculating Cell EMF A voltaic cell is based on a Co 2+/Co half cell and an Ag. Cl/Ag half cell. a. ) What half-reaction occurs at the anode? b. ) What is the standard cell potential? See Sample Exercise 20. 7 (Pg. 859)

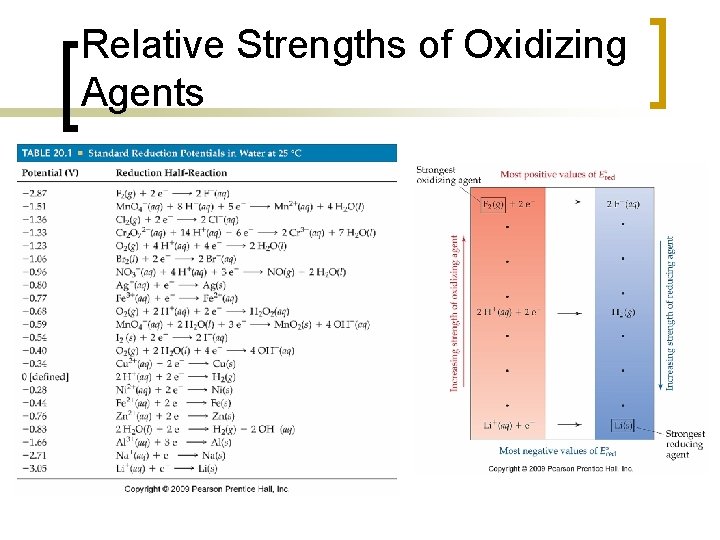
Relative Strengths of Oxidizing Agents

Free Energy and Redox Reactions Section 20. 5 n We know that a negative G value indicates a spontaneous reaction, and we also know that a positive emf, E, also indicates spontaneity Therefore there must be a link between the two: G = -n. FE or G = -n. FE (std. cond. ) F = Faraday’s constant = 96, 485 J/V mol n = # of electrons transferred in the reaction ¡

Determining G Calculate the standard free energy change for See the reaction below: Sample Exercise 20. 10 (Pg. Ag. Cl(s) + Fe+2(aq) Ag(s) + Fe+3(aq) + Cl-(aq) 864)

Determining G and K Calculate the equilibrium constant for the reaction below: Sn 2+(aq) + Ni(s) Sn(s) + Ni 2+(aq) See Sample Exercise 20. 10 (Pg. 864)

Cell emf Under Nonstandard Conditions Section 20. 6 n Cell potential under nonstandard conditions can be calculated using the Nernst equation which is derived from the equation describing free energy change: G = G + RT ln(Q) Nernst equation:

Voltaic Cell EMF under Nonstandard Conditions A voltaic cell consists of a half-cell of iron ions in a solution with [Fe 2+] = 2. 0 M and [Fe 3+] = 0. 75 M, and a half-cell of copper metal immersed in a solution containing Cu 2+ at a concentration of 3. 6 x 10 -4 M. What is the voltage of this cell? See Sample Exercise 20. 11 (Pg. 866)

Calculating Concentration in a Voltaic Cell What is the p. H of the solution in the See Sample cathode compartment of the reaction Exercise 20. 12 shown below when PH 2 = 1. 0 atm, (Pg. 866) [Zn 2+] in the anode compartment is 0. 10 M, and the cell emf (Ecell) is 0. 542 V? Zn(s) + 2 H+(aq) → Zn 2+(aq) + H 2(g)

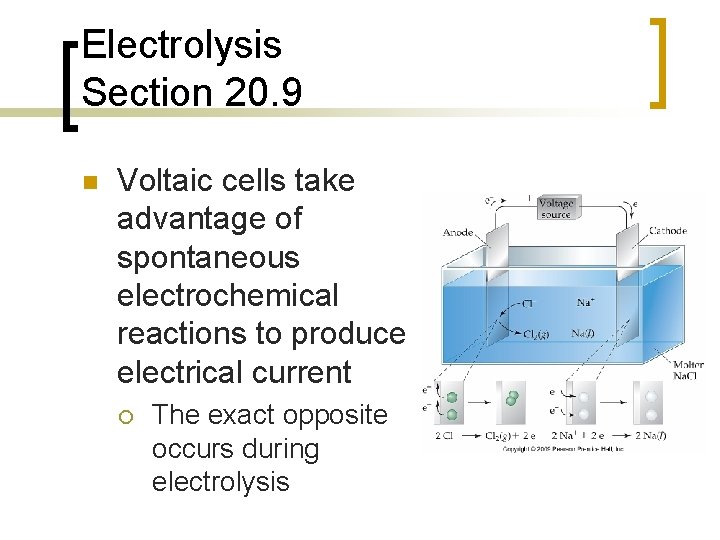
Electrolysis Section 20. 9 n Voltaic cells take advantage of spontaneous electrochemical reactions to produce electrical current ¡ The exact opposite occurs during electrolysis

Quantitative Aspects of Electrolysis n Consider the reactions below: Li+ → Li(s) Mg 2+ → Mg(s) n If the amount of charge applied to an electrolytic cell is known, the mass of the electroplated material can be calculated Coulombs = amperes x seconds

Relating Electrical Charge and Quantity of Electrolysis A constant current of 0. 500 A passes through a silver nitrate solution for 90. 0 minutes. What mass of silver metal is deposited at the anode? See Sample Exercise 20. 14 (Pg. 878)