Electrochemistry Applications of Redox Review o Oxidation reduction

































- Slides: 42

Electrochemistry Applications of Redox

Review o Oxidation reduction reactions involve a transfer of electrons. o OIL- RIG o Oxidation Involves Loss o Reduction Involves Gain o LEO-GER o Lose Electrons Oxidation o Gain Electrons Reduction

Applications o Moving electrons is electric current. o 8 H++Mn. O 4 -+ 5 Fe+2 +5 eo o ® Mn+2 + 5 Fe+3 +4 H 2 O Helps to break the reactions into half reactions. 8 H++Mn. O 4 -+5 e- ® Mn+2 +4 H 2 O 5(Fe+2 ® Fe+3 + e- ) In the same mixture it happens without doing useful work, but if separate

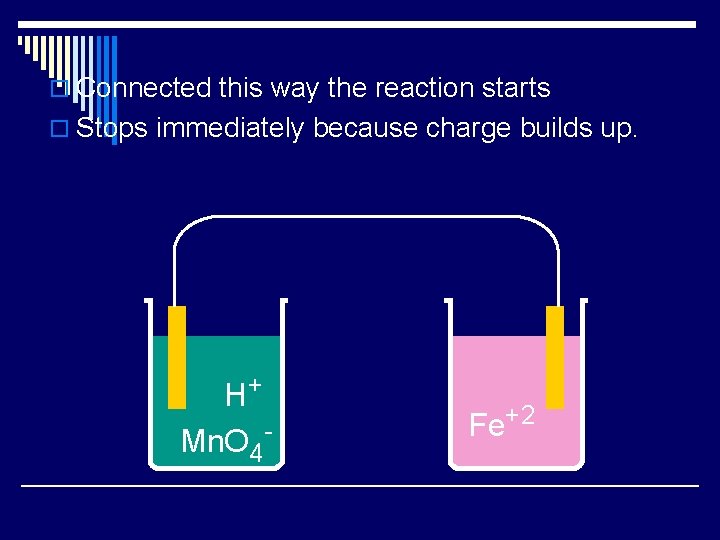
o Connected this way the reaction starts o Stops immediately because charge builds up. H+ Mn. O 4 - Fe+2

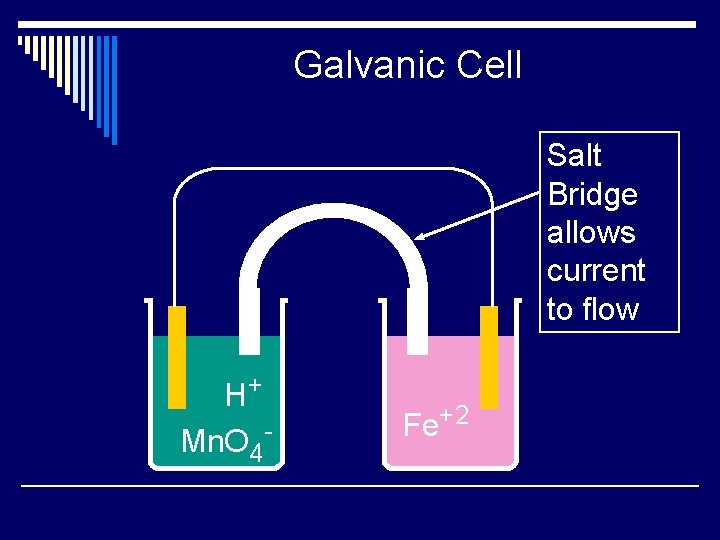
Galvanic Cell Salt Bridge allows current to flow H+ Mn. O 4 - Fe+2

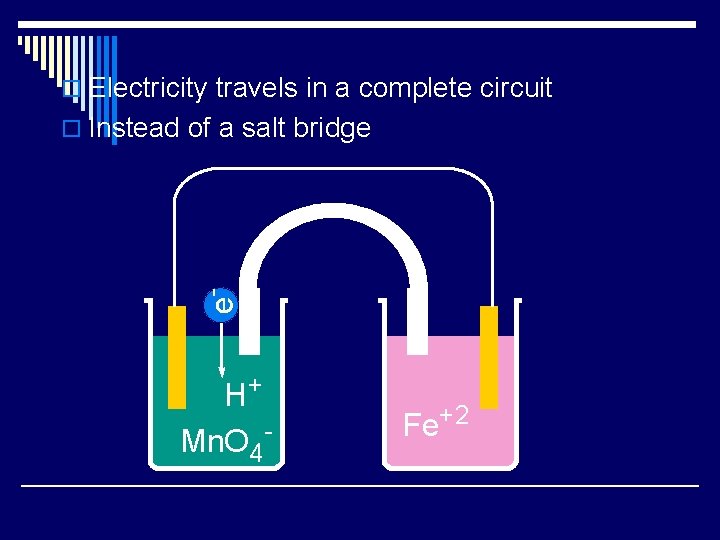
o Electricity travels in a complete circuit e- o Instead of a salt bridge H+ Mn. O 4 - Fe+2

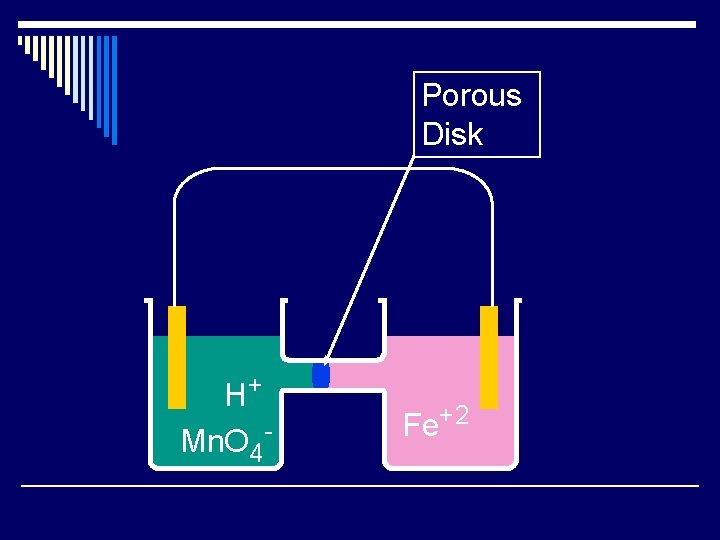
Porous Disk H+ Mn. O 4 - Fe+2

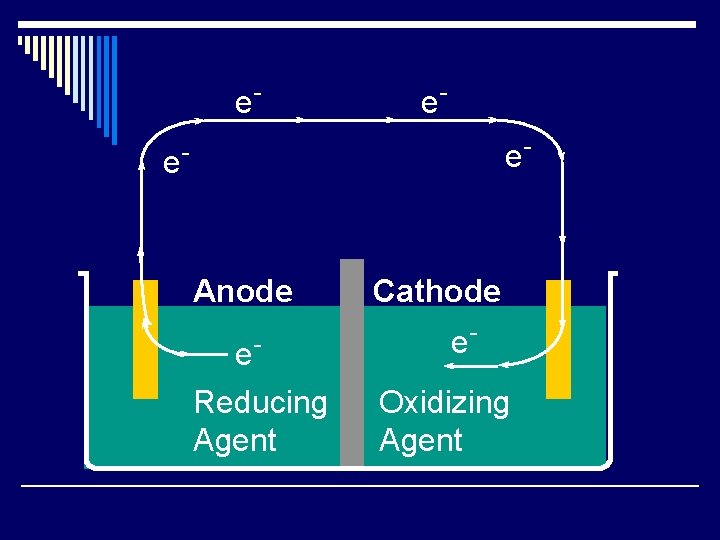
e- e- Anode e. Reducing Agent Cathode e. Oxidizing Agent

Cell Potential o Oxidizing agent pushes the electron. o Reducing agent pulls the electron. o The push or pull (“driving force”) is called the cell potential Ecell o Also called the electromotive force (emf) o Unit is the volt(V) o = 1 joule of work/coulomb of charge o Measured with a voltmeter

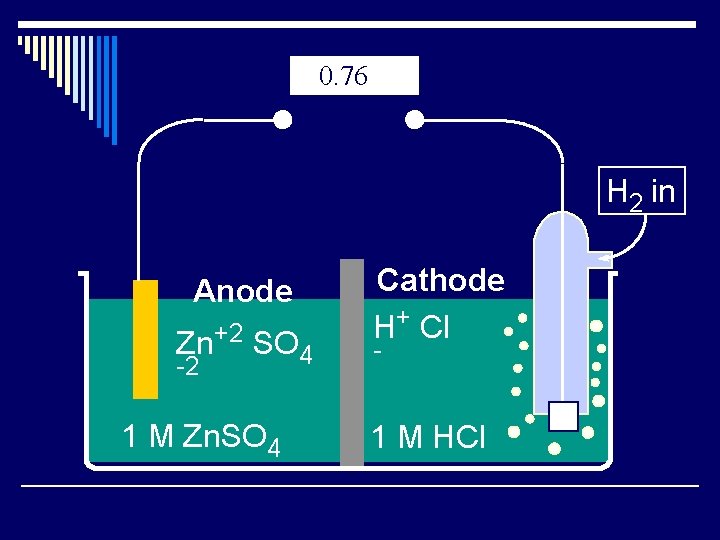
0. 76 H 2 in Anode Zn+2 SO 4 -2 1 M Zn. SO 4 Cathode H+ Cl - 1 M HCl

Standard Hydrogen Electrode o This is the reference all other oxidations are compared to o. E º = 0 + Cl H o º indicates standard - states of 25ºC, 1 atm, 1 M solutions. 1 M HCl H 2 in

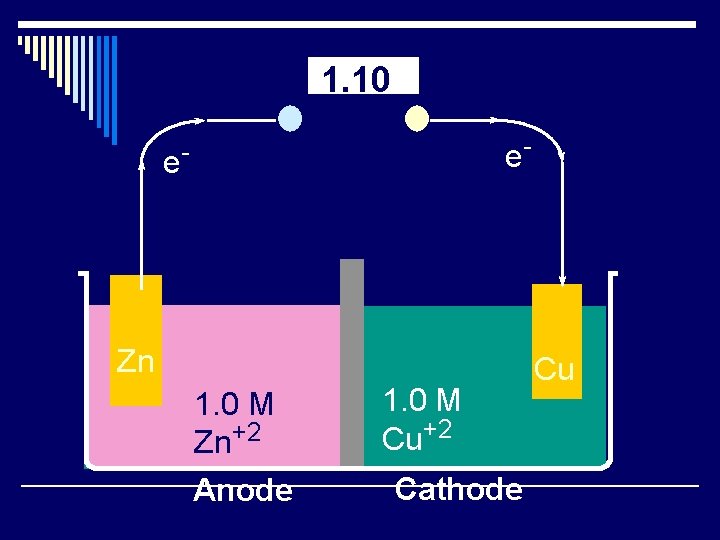
Cell Potential o Zn(s) + Cu+2 (aq) ® Zn+2(aq) + Cu(s) o The total cell potential is the sum of the potential at each electrode. o Eº cell = EºZn® Zn+2 + Eº Cu+2 ® Cu o We can look up reduction potentials in a table. o One of the reactions must be reversed, so change it sign.

Cell Potential o Determine the cell potential for a galvanic cell based on the redox reaction. o Cu(s) + Fe+3(aq) ® Cu+2(aq) + Fe+2(aq) o Fe+3(aq) + e-® Fe+2(aq) Eº = 0. 77 V o Cu+2(aq)+2 e- ® Cu(s) Eº = 0. 34 V o Cu(s) ® Cu+2(aq)+2 e. Eº = -0. 34 V o 2 Fe+3(aq) + 2 e-® 2 Fe+2(aq) Eº = 0. 77 V o 0. 77 V + - 0. 34 V = 0. 43 V

Homework o 17. 27 and 17. 28

Line Notation solid½Aqueous½solid o Anode on the left½½Cathode on the right o Single line different phases. o Double line porous disk or salt bridge. o If all the substances on one side are aqueous, a platinum electrode is indicated. o For the last reaction o Cu(s)½Cu+2(aq)½½Fe+2(aq), Fe+3(aq)½Pt(s) o

Galvanic Cell o o 1) 2) 3) 4) The reaction always runs spontaneously in the direction that produced a positive cell potential. Four things for a complete description. Cell Potential Direction of flow Designation of anode and cathode Nature of all the componentselectrodes and ions

Practice o Completely describe the galvanic cell based on the following half-reactions under standard conditions. o Mn. O 4 - + 8 H+ +5 e- ® Mn+2 + 4 H 2 O Eº=1. 51 o Fe+2 +2 e- ® Fe(s) Eº=0. 44 V o Reverse the smaller valued reaction o Fe(s) ® Fe+2 +2 eo Eº= - 0. 44 V

Solution o Balance the cell reaction: o Balance the electrons to balance the equation: The First reaction requires 5 e-, the second one supplies 2 e-, so 10 must be exchanged. o 2(Mn. O 4 - + 8 H+ +5 e- ® Mn+2 + 4 H 2 O) o 5(Fe(s) ® Fe+2 +2 e-) o Add the reactions together n

Solution Continued o 2 Mn. O 4 -(aq) + 16 H+ (aq)+5 Fe (s) ® 5 Fe +2 (aq) + 2 Mn+2 (aq) + 8 H 2 O (l) o Determine the Cell Potential: n 2(1. 51) + 5(-0. 44) = 3. 02 – 2. 2 = 0. 8 o Fe(s) l Fe+2(aq) ll Mn. O 4 -(aq), Mn+2(aq) l Pt(s)

More Homework o 17. 29 and 17. 30

Potential, Work and DG o emf = potential (V) = work (J) / Charge(C) o E = work done by system / charge o E = -w/q o Charge is measured in coulombs. o -w = q. E o Faraday = 96, 485 C/mol eo q = n. F = moles of e- x charge/mole eo w = -q. E = -n. FE = DG

Potential, Work and DG o DGº = -n. FE º o if E º < 0, then DGº > 0 nonspontaneous o if E º > 0, then DGº < 0 spontaneous o In fact, reverse is spontaneous. o Calculate DGº for the following reaction: o Cu+2(aq)+ Fe(s) ® Cu(s)+ Fe+2(aq) o Fe+2(aq) + 2 e-® Fe(s) o Cu+2(aq)+2 e- ® Cu(s) Eº = -0. 44 V Eº = 0. 34 V

Practice o Cu+2(aq)+ Fe(s) ® Cu(s)+ Fe+2(aq) o Fe(s) ® Fe+2(aq) + 2 e. Eº = 0. 44 V o Cu+2(aq)+2 e- ® Cu(s) Eº = 0. 34 V o 0. 44 V + 0. 34 V = 0. 78 V o DGº = -n. FE º o DGº = -(2 mol e-)96, 485 C/mol e-(0. 78 J/C) o DGº = -1. 5 x 105 J

Practice Predicting Spontaneity o Will 1 M HNO 3 dissolve gold metal to form a 1 M o o Au 3+ solution? HNO 3 half reaction: NO 3 - + 4 H+ + 3 e- NO + 2 H 2 O E=0. 96 V Au 3+ Half Reaction: Au 3+ + 3 e- E = -1. 50 V o Au(s) + NO 3 -(aq) + 4 H+(aq) Au 3+(s) + NO(g) + 2 H 2 O(l) o Ecell = 0. 96 V – 1. 50 V = -0. 54 V o Since Ecell is negative, it will NOT occur under standard conditions.

Homework o 17. 37, 17. 39, 17. 49

Cell Potential and Concentration o Qualitatively - Can predict direction of change in E from Le. Châtelier. o 2 Al(s) + 3 Mn+2(aq) ® 2 Al+3(aq) + 3 Mn(s) o Predict if Ecell will be greater or less than Eºcell if [Al+3] = 1. 5 M and [Mn+2] = 1. 0 M LESS o if [Al+3] = 1. 0 M and [Mn+2] = 1. 5 M GREATER o if [Al+3] = 1. 5 M and [Mn+2] = 1. 5 M SLIGHTLY GREATER

Homework o 17. 55

The Nernst Equation o DG = DGº +RTln(Q) o -n. FE = -n. FEº + RTln(Q) o E = Eº - RTln(Q) n. F o 2 Al(s) + 3 Mn+2(aq) ® 2 Al+3(aq) + 3 Mn(s) Eº = 0. 48 V o Always have to figure out n by balancing. o If concentration can gives voltage, then from voltage we can tell concentration.

The Nernst Equation o As reactions proceed concentrations of products increase and reactants decrease. o Reach equilibrium where Q = K and Ecell = 0 o 0 = Eº - RTln(K) n. F o Eº = RTln(K) n. F o n. FEº = ln(K) RT

Example Using Nernst o Describe the cell based on the following: o VO 2+ + 2 H+ + e- VO 22+ + H 2 O Eo = 1. 00 V o Zn 2+ + 2 e- Zn Eo = -0. 76 V o T = 25 C o [VO 2+] = 2. 0 M [H+] = 0. 50 M o [VO 22+] = 0. 01 M [Zn 2+] = 0. 1 M

Starting Solution o In Order to balance the electrons we must o o o double the first equation, and reverse the second equation, so we end up with: 2 VO 2+ + 4 H+ + 2 e- 2 VO 22+ + 2 H 2 O Zn 2+ + 2 e 2 VO 2+ (aq) + 4 H+ (aq) + Zn (s) 2 VO 22+ (aq) + 2 H 2 O (l) + Zn 2+ (aq) Ecell = 1. 76 V Note: Because we are using the Nernst Equation we use base values for E

Final Solution o At 25 C R = 0. 0521 o E = Ecell –R/n log (Q) o E = 1. 76 –(0. 0521/2) log ([Zn 2+][VO 22+]2) [VO 2+]2[H+]4 o E = 1. 76 –(0. 0521/2) log ([0. 10][0. 010]2) [2. 0]2[0. 50]4 o E = 1. 76 –(0. 0521/2) log 4. 0 x 10 -5 o E = 1. 76 + 0. 13 = 1. 89 V

Homework o 17. 61 and 17. 63

Batteries are Galvanic Cells o Car batteries are lead storage batteries. o Pb +Pb. O 2 +H 2 SO 4 ®Pb. SO 4(s) +H 2 O o Dry Cell + NH 4+ +Mn. O 2 ® Zn+2 + NH 3 + H 2 O o Alkaline +Mn. O 2 ® Zn. O+ Mn 2 O 3 (in base) o Ni. Cad o Ni. O 2 + Cd + 2 H 2 O ® Cd(OH)2 +Ni(OH)2 Zn Zn

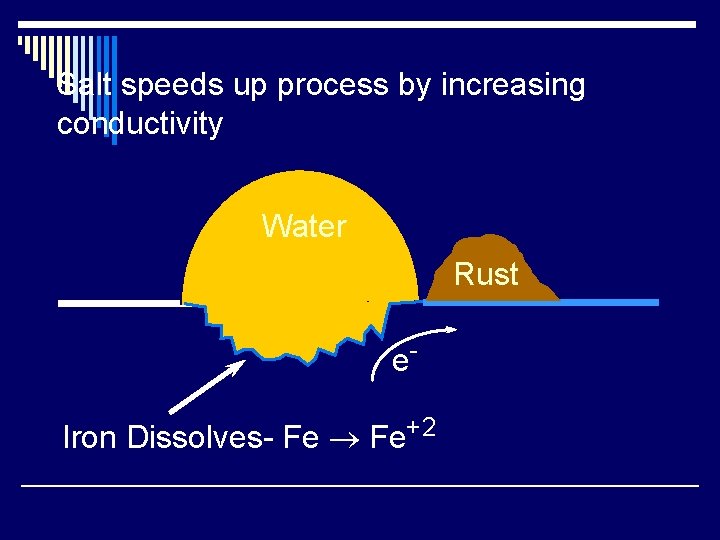
Corrosion o Rusting - spontaneous oxidation. o Most structural metals have reduction potentials that are less positive than O 2. o Fe ® Fe+2 +2 e. Eº= 0. 44 V o O 2 + 2 H 2 O + 4 e- ® 4 OH- Eº= 0. 40 V o Fe+2 + O 2 + H 2 O ® Fe 2 O 3 + H+ o Reaction happens in two places.

Salt speeds up process by increasing conductivity Water Rust e. Iron Dissolves- Fe ® Fe+2

Preventing Corrosion o Coating to keep out air and water. o Galvanizing - Putting on a zinc coat o Has a lower reduction potential, so it is more. easily oxidized. o Alloying with metals that form oxide coats. o Cathodic Protection - Attaching large pieces of an active metal like magnesium that get oxidized instead.

Electrolysis o Running a galvanic cell backwards. o Put a voltage bigger than the potential and reverse the direction of the redox reaction. o Used for electroplating.

1. 10 e- e- Zn 1. 0 M Zn+2 Anode 1. 0 M Cu+2 Cathode Cu

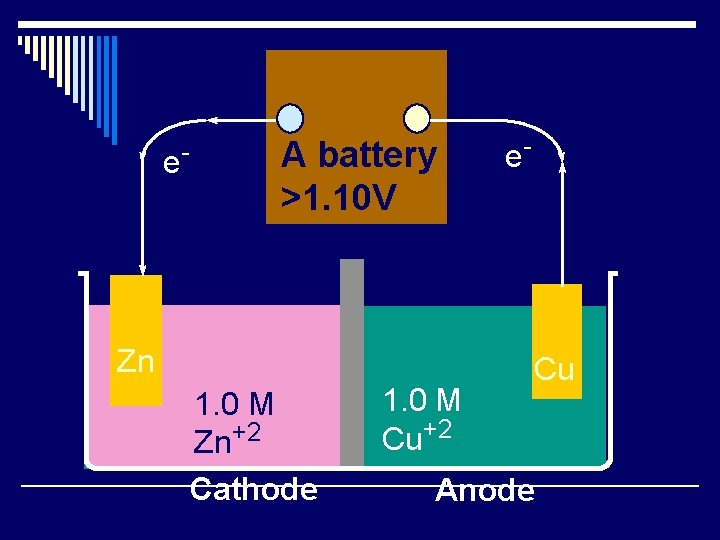
e- A battery >1. 10 V Zn 1. 0 M Zn+2 Cathode 1. 0 M Cu+2 e- Cu Anode

Calculating plating o Have to count charge. o Measure current I (in amperes) o 1 amp = 1 coulomb of charge per second o q=Ixt o q/n. F = moles of metal o Mass of plated metal o How long must 5. 00 amp current be applied to produce 15. 5 g of Ag from Ag+

Other uses o Electroysis of water. o Seperating mixtures of ions. o More positive reduction potential means the reaction proceeds forward. o We want the reverse. o Most negative reduction potential is easiest to plate out of solution.