ELECTROCHEMICAL SURFACE FINISHING OF ADDITIVELY MANUFACTURED PARTS H








- Slides: 22

ELECTROCHEMICAL SURFACE FINISHING OF ADDITIVELY MANUFACTURED PARTS H. Garich 1, T. Hall 1, E. J. Taylor 1, D. Lui 1, J. Porter 2, and B. Hayes 2 1 Faraday Technology (Dayton, OH) 2 UES, Inc. (Dayton, OH) 233 rd ECS Meeting, Seattle, WA USA May 16, 2018

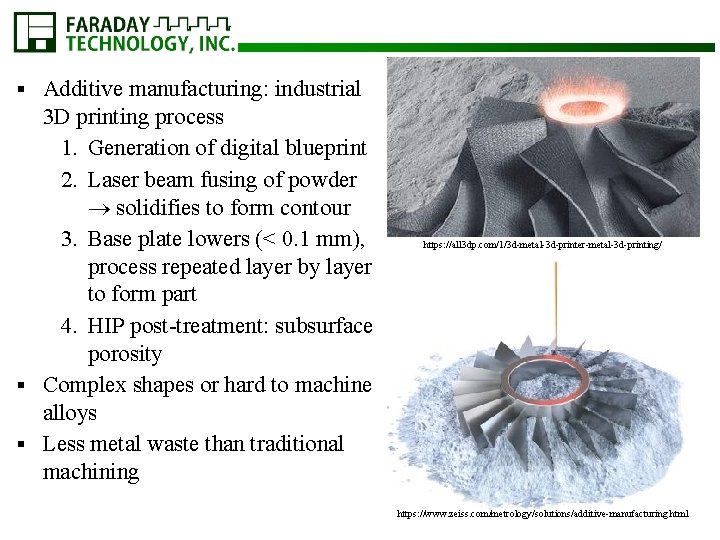
Additive manufacturing: industrial 3 D printing process 1. Generation of digital blueprint 2. Laser beam fusing of powder solidifies to form contour 3. Base plate lowers (< 0. 1 mm), process repeated layer by layer to form part 4. HIP post-treatment: subsurface porosity § Complex shapes or hard to machine alloys § Less metal waste than traditional machining § https: //all 3 dp. com/1/3 d-metal-3 d-printer-metal-3 d-printing/ https: //www. zeiss. com/metrology/solutions/additive-manufacturing. html

AM parts have inherent surface roughness - micro-roughness and large scale waviness § Surface finishing processes needed to ready part for use § Electrochemical polishing: ability to address complex features, internal channels/passages that are a challenge with conventional machining § Pulse reverse processes: enable polishing of oxide forming materials without use of harsh/toxic chemistry such as HF; control of mass removal rate, throwing power § A. Karme et al, Physics Procedia, 78, 347 -356 (2015)

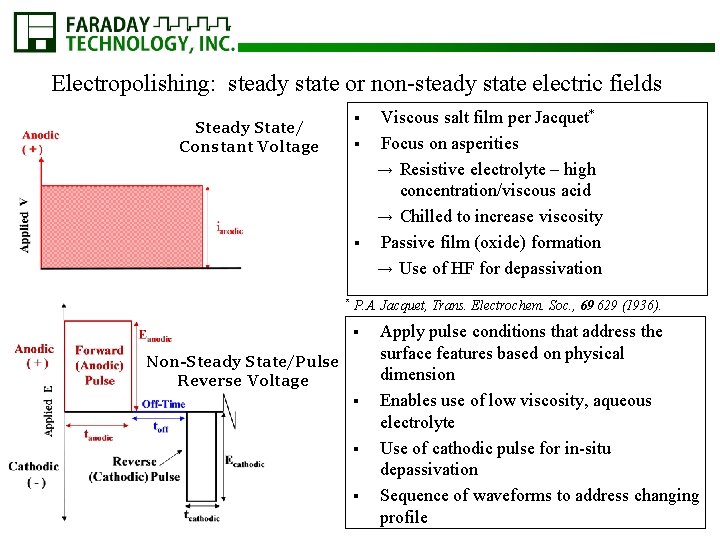
Electropolishing: steady state or non-steady state electric fields Viscous salt film per Jacquet* § Focus on asperities → Resistive electrolyte – high concentration/viscous acid → Chilled to increase viscosity § Passive film (oxide) formation → Use of HF for depassivation § Steady State/ Constant Voltage * P. A. Jacquet, Trans. Electrochem. Soc. , 69 629 (1936). § Non-Steady State/Pulse Reverse Voltage § § § Apply pulse conditions that address the surface features based on physical dimension Enables use of low viscosity, aqueous electrolyte Use of cathodic pulse for in-situ depassivation Sequence of waveforms to address changing profile

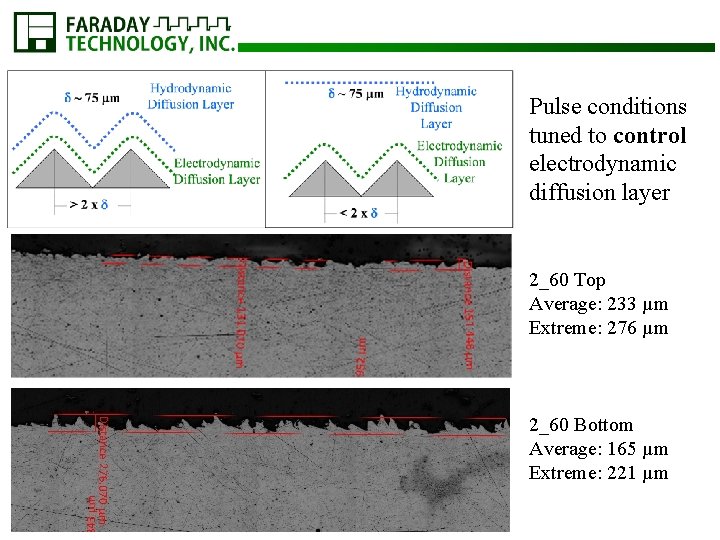
Pulse conditions tuned to control electrodynamic diffusion layer 2_60 Top Average: 233 µm Extreme: 276 µm 2_60 Bottom Average: 165 µm Extreme: 221 µm

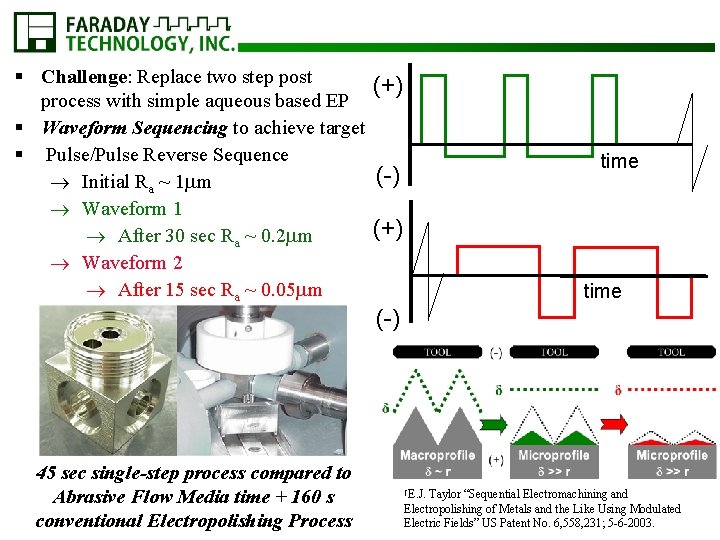
§ Challenge: Replace two step post (+) process with simple aqueous based EP § Waveform Sequencing to achieve target § Pulse/Pulse Reverse Sequence (-) Initial Ra ~ 1 m Waveform 1 (+) After 30 sec Ra ~ 0. 2 m Waveform 2 After 15 sec Ra ~ 0. 05 m time (-) 45 sec single-step process compared to Abrasive Flow Media time + 160 s conventional Electropolishing Process †E. J. Taylor “Sequential Electromachining and Electropolishing of Metals and the Like Using Modulated Electric Fields” US Patent No. 6, 558, 231; 5 -6 -2003.

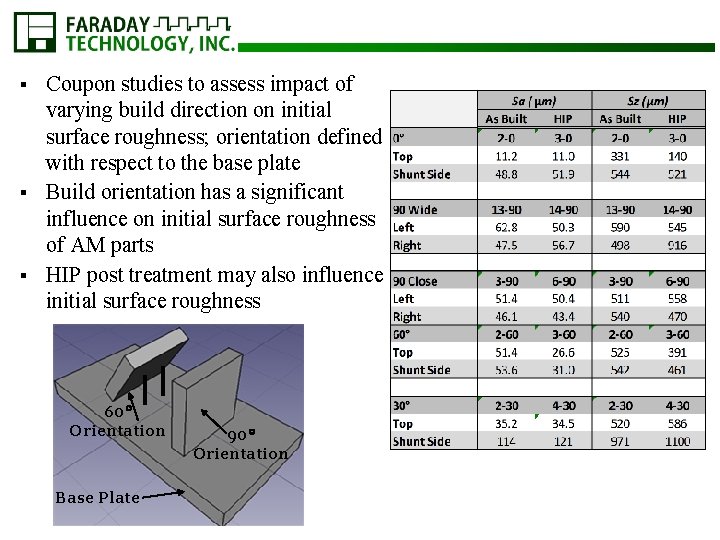
Coupon studies to assess impact of varying build direction on initial surface roughness; orientation defined with respect to the base plate § Build orientation has a significant influence on initial surface roughness of AM parts § HIP post treatment may also influence initial surface roughness § 60 Orientation Base Plate 90 Orientation

Electropolishing of Ti 64 demonstrated in multiple, aqueous electrolytes § Coupon: electrolyte and waveform development § Kazoo sample: electrode design; waveform optimization (throw) § Scaling and demonstration on full size part §

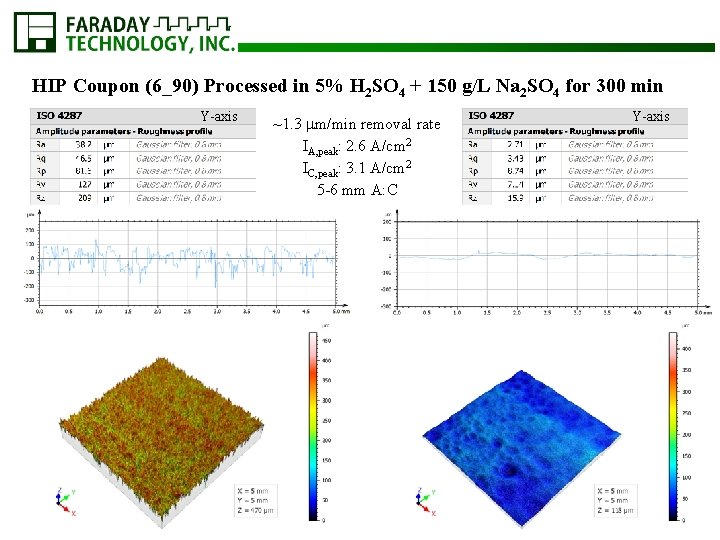
HIP Coupon (6_90) Processed in 5% H 2 SO 4 + 150 g/L Na 2 SO 4 for 300 min Y-axis ~1. 3 m/min removal rate IA, peak: 2. 6 A/cm 2 IC, peak: 3. 1 A/cm 2 5 -6 mm A: C Y-axis

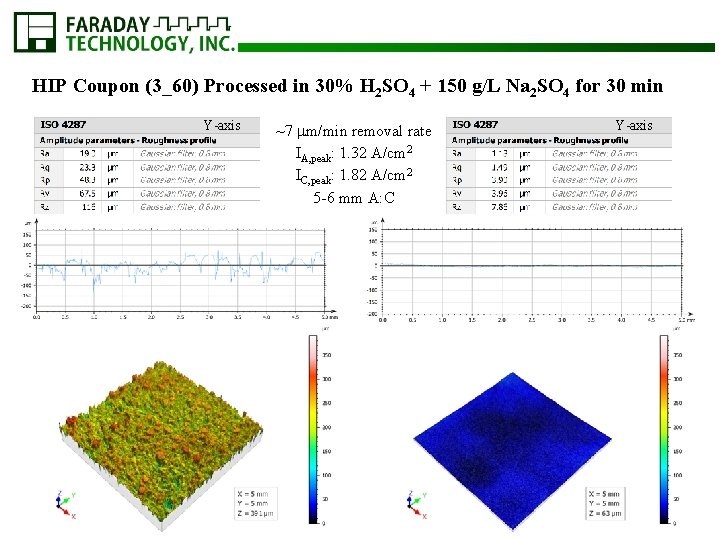
HIP Coupon (3_60) Processed in 30% H 2 SO 4 + 150 g/L Na 2 SO 4 for 30 min Y-axis ~7 m/min removal rate IA, peak: 1. 32 A/cm 2 IC, peak: 1. 82 A/cm 2 5 -6 mm A: C Y-axis

Summary of Tests in Sulfuric Acid Electrolytes In 5% (w/w) H 2 SO 4 + 150 g/L Na 2 SO 4: → Issues associated with smutting and slow removal rates → Low waveform frequencies and lower applied voltage to smooth rough surfaces → Waveform must be changed to address changing surface profile § In 30% (w/w) H 2 SO 4 + 150 g/L Na 2 SO 4: → Less luster than observed with 5% electrolyte → Issues of smutting are minimized → Ra decreases rapidly with less material losses → Cathodic pulse must be balanced with other parameters to ensure oxide removal and subsequent material removal § The high current densities observed with these electrolytes are prohibitive for scaling activities when decreasing anode-to-cathode spacing→ evaluate lower conductivity electrolyte §

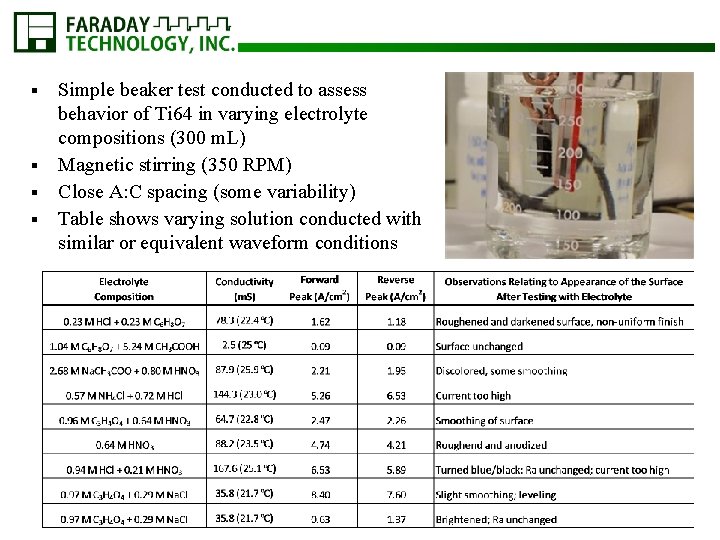
Simple beaker test conducted to assess behavior of Ti 64 in varying electrolyte compositions (300 m. L) § Magnetic stirring (350 RPM) § Close A: C spacing (some variability) § Table shows varying solution conducted with similar or equivalent waveform conditions §

Oxalic acid vs malonic acid: cost Evaluate waveform conditions in flow cell using 0. 66 M C 2 H 2 O 4 + 0. 19 M Na. Cl § A: C spacing of 2. 5 mm and flow rate of 1 GPM to mimic processing of flow channels § §

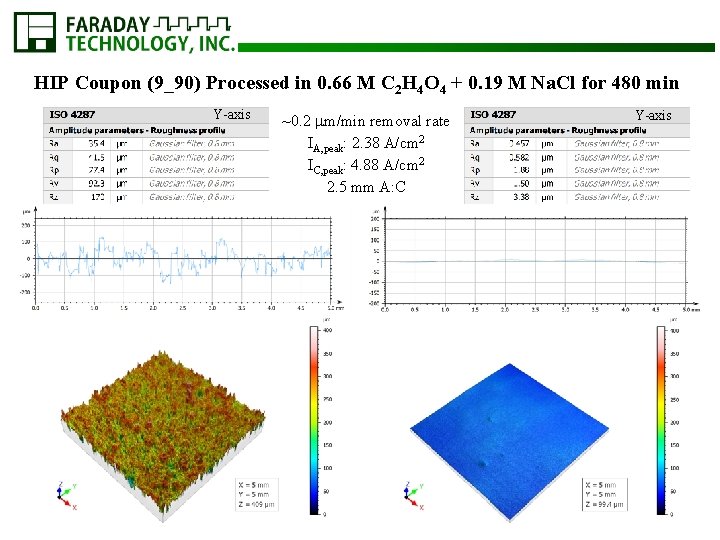
HIP Coupon (9_90) Processed in 0. 66 M C 2 H 4 O 4 + 0. 19 M Na. Cl for 480 min Y-axis ~0. 2 m/min removal rate IA, peak: 2. 38 A/cm 2 IC, peak: 4. 88 A/cm 2 2. 5 mm A: C Y-axis

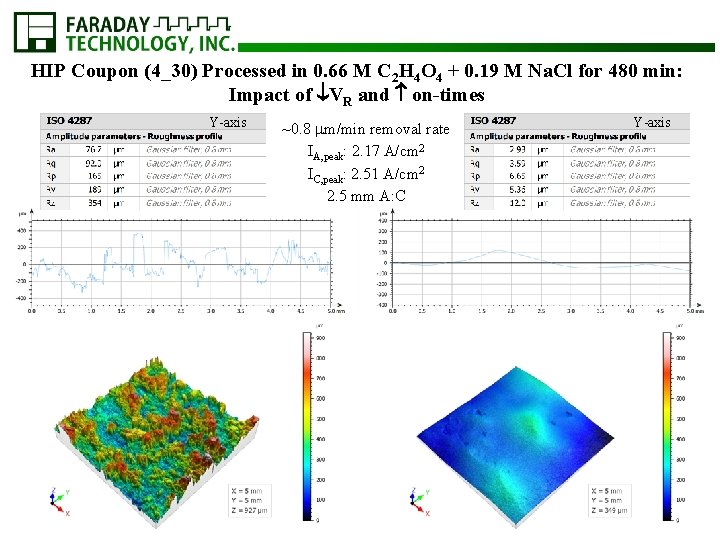
HIP Coupon (4_30) Processed in 0. 66 M C 2 H 4 O 4 + 0. 19 M Na. Cl for 480 min: Impact of VR and on-times Y-axis ~0. 8 m/min removal rate IA, peak: 2. 17 A/cm 2 IC, peak: 2. 51 A/cm 2 2. 5 mm A: C Y-axis

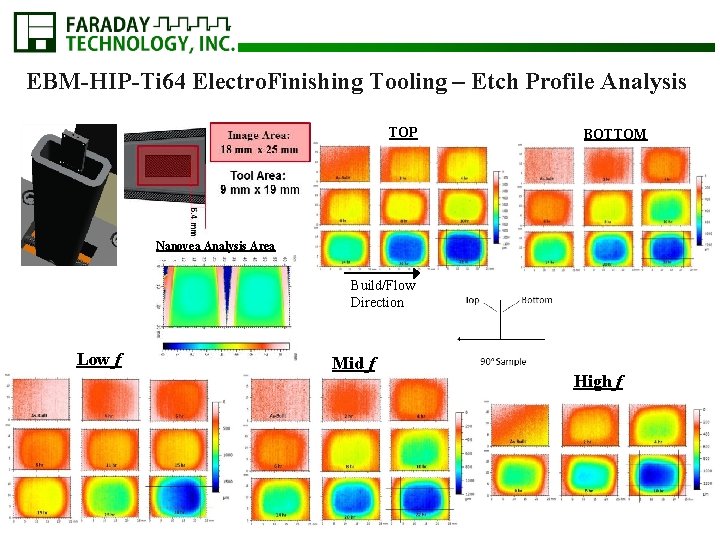
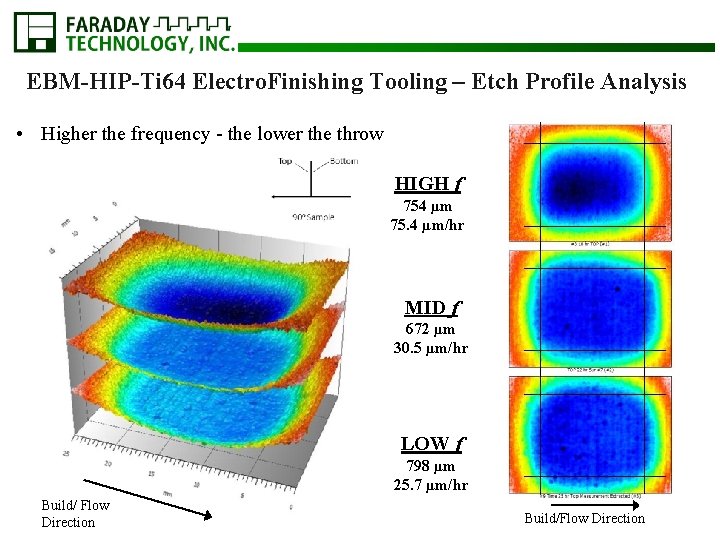
EBM-HIP-Ti 64 Electro. Finishing Tooling – Etch Profile Analysis TOP BOTTOM Nanovea Analysis Area Build/Flow Direction Low f Mid f High f

EBM-HIP-Ti 64 Electro. Finishing Tooling – Etch Profile Analysis • Higher the frequency - the lower the throw HIGH f 754 µm 75. 4 µm/hr MID f 672 µm 30. 5 µm/hr LOW f 798 µm 25. 7 µm/hr Build/ Flow Direction Build/Flow Direction

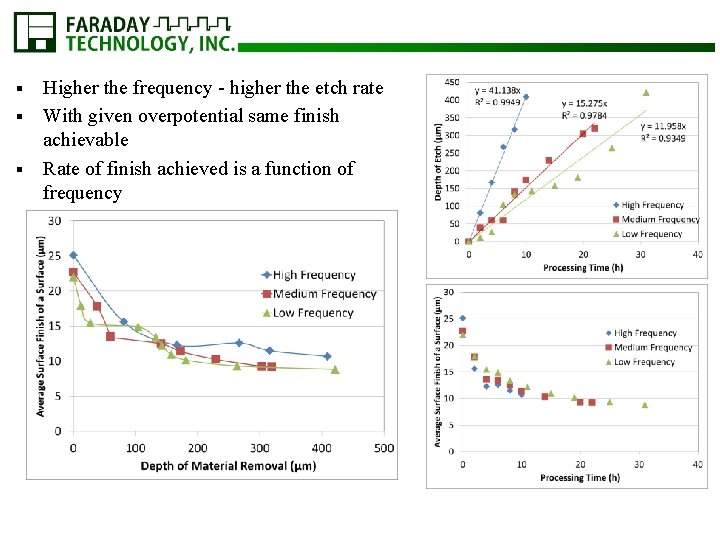
Higher the frequency - higher the etch rate § With given overpotential same finish achievable § Rate of finish achieved is a function of frequency §

EBM-Ti 64 Electro. Finishing Tooling – Shrouded Impeller

EBM-Ti 64 Electro. Finishing Tooling – Shrouded Impeller Post Finish Surface Upward Facing As-Built Surface (Separate Part) Upward Facing Downward Facing

o Demonstrated potential to use P/PR Electro. Finishing techniques to finish various Conventionally Manufactured and Additively Manufactured materials in water based HF-free electrolytes § Ti 64, Hast-X, IN 718, etc… o Demonstrated potential to selectively remove dissimilar materials from surface feature o Demonstrated the potential of electrically controlled electrochemical processes and sequencing to produce the desired finish while limiting material removal o Applications: § Medical, Automotive, Aerospace, Energy Markets, Even Superconducting Nb structures

ACKNOWLEDGEMENTS THANK YOU FOR YOUR ATTENTION! QUESTIONS? Financial support: USAF RIF, NASA, Client Confidential Contact Information: Tim Hall, Maria Inman, or EJ Taylor Ph: 937 -836 -7749