Electrochemical Cells Batteries SNC 1 D Electrochemical Cells

Electrochemical Cells (Batteries) SNC 1 D

Electrochemical Cells Cell is another name for battery. n Cells are classified as either primary or secondary. n In a primary cell, chemical reactions use up some of the materials in the cell as electrons flow from it. They can’t be recharged. n When these materials have been used up, the cell is said to be discharged and cannot be recharged. * n

Primary Cells Primary cells can be further classified as either wet or dry. n The primary wet cell was first developed in 1800 by Italian scientist, Alessandro Volta. n This cell is therefore called the voltaic cell. n

Primary Wet Cells A wet cell is made up of two pieces of metal that are placed in a liquid. n The metal plates, usually zinc and copper, are called electrodes. n The liquid in the cell is called the electrolyte. n An electrolyte is any liquid that conducts an electric current. n

Primary Wet Cells Electrons collect at the positive and negative terminals of the cell. n The cell will only discharge when connected to a closed circuit. n

Primary Dry Cells The dry cell is similar to the wet cell, but the electrolyte is a moist paste instead of a liquid. n When most of the negative electrode has been used up by the chemical reaction, the electrons stop flowing and the cell is discharged. n n A group of cells make up a battery.

Simple Primary Wet Cell · Consists of two different metal electrodes immersed in acidic liquid electrolyte. · In solution, the acid molecules separate into positive and negative ions. Figure 4 -1 A simple primary cell

Secondary Cells Unlike primary cells, a secondary cell can be discharged and recharged many hundreds of times. n Secondary cells are often referred to rechargeable batteries. n Secondary cells are so named since there are two chemical processes involved: n one to discharge the cell n one to charge the cell n n A car battery consists of a group of secondary cells.

Lemon Power http: //electronics. howstuffworks. com/framed. ht m? parent=battery. htm&url=http: //members. aol. c om/dswart/index. html

Battery Arrangements http: //electronics. howstuffworks. com/battery 8. htm

Modern Battery Chemicals Modern batteries use a variety of chemicals to power their reactions. Typical battery chemistries include: Zinc-carbon battery - Also known as a standard carbon battery, zinc-carbon chemistry is used in all inexpensive AA, C and D dry-cell batteries. The electrodes are zinc and carbon, with an acidic paste between them that serves as the electrolyte. Alkaline battery - Used in common Duracell and Energizer batteries, the electrodes are zinc and manganese-oxide, with an alkaline electrolyte. Lithium photo battery - Lithium, lithium-iodide and lead-iodide are used in cameras because of their ability to supply power surges. Lead-acid battery - Used in automobiles, the electrodes are made of lead and lead-oxide with a strong acidic electrolyte (rechargeable). Nickel-cadmium battery - The electrodes are nickel-hydroxide and cadmium, with potassium-hydroxide as the electrolyte (rechargeable). Nickel-metal hydride battery - This battery is rapidly replacing nickel-cadmium because it does not suffer from the memory effect that nickel-cadmiums do (rechargeable). Lithium-ion battery - With a very good power-to-weight ratio, this is often found in high-end laptop computers and cell phones (rechargeable). Zinc-air battery - This battery is lightweight and rechargeable. Zinc-mercury oxide battery - This is often used in hearing-aids. Silver-zinc battery - This is used in aeronautical applications because the power-to-weight ratio is good. Metal-chloride battery - This is used in electric vehicles. Modern Battery Chemicals http: //electronics. howstuffworks. com/battery 7. htm

Battery Basics http: //electronics. howstuffworks. com/battery 1. htm

If you arrange four of these 1. 25 -volt, 500 milliamphour batteries in a serial arrangement, you get 5 volts (1. 25 x 4) at 500 milliamp-hours. you arrange Milliamp-Hour & 9 Volt If. Cutaway them in parallel, you get 1. 25 volts at 2, 000 (500 x 4) milliamp-hours. Have you ever looked inside a 9 -volt battery? It contains six, very small batteries producing 1. 5 volts each in a serial arrangement!
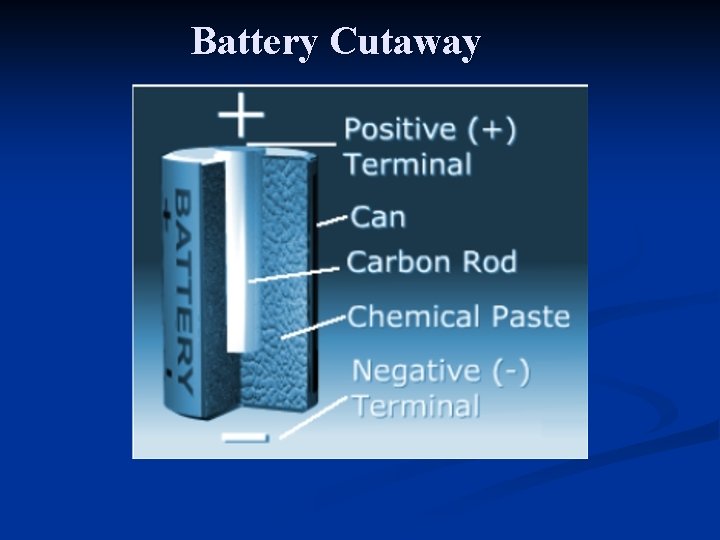
Battery Cutaway
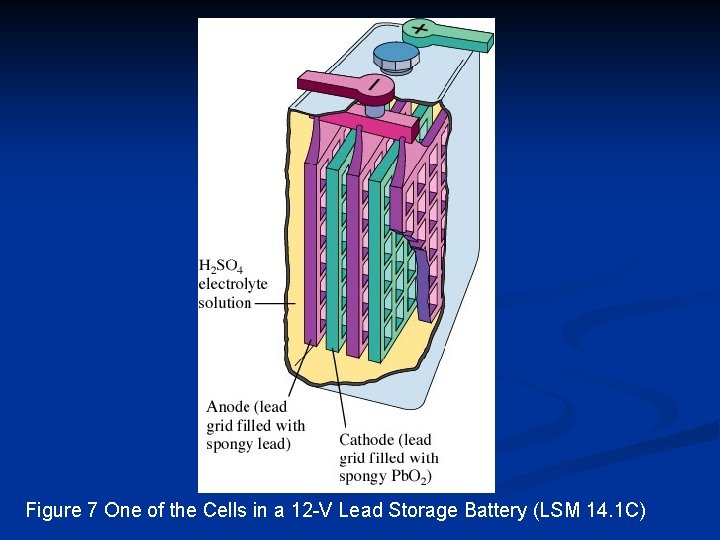
Figure 7 One of the Cells in a 12 -V Lead Storage Battery (LSM 14. 1 C)
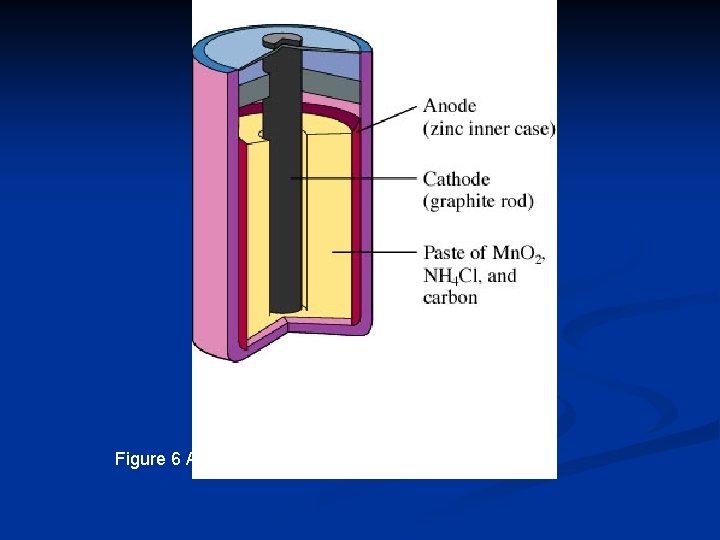
Figure 6 A Common Dry Cell Battery (LSM 14. 1 C)
- Slides: 16