Electricity Static and Dynamic Magnetism Electricity Electricity refers

















- Slides: 24

Electricity Static and Dynamic Magnetism

Electricity • Electricity refers to all phenomena caused by electric charges (positive and negative)

When is it positive? When is it negative? • A negatively charged body has more electrons than protons • A Positively charged body less electrons than protons

Electric Charges • Elementary electric charge is the charge of an electron or protons (1, 602 x 10 -19 C) – C refers to Coulomb

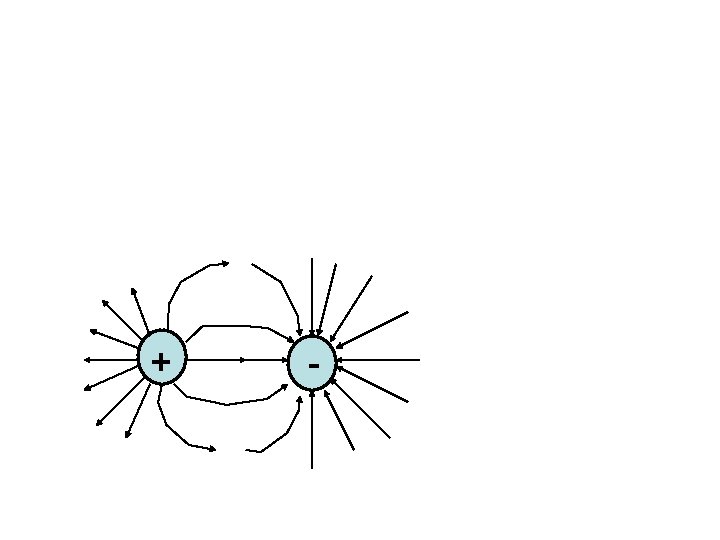
The Basics: Forces of attraction and repulsion 1. Like electric charges repulse each other 2. Opposite electric charges attract each other 3. The force that pushes charges together or apart is called the electric force 4. The “Law of conservation of charges” states that no charge is created or destroyed only transferred

How do we give charge? • How a material reacts when given a charge will determine in which category they will be placed 1. Conductors 2. Insulators 3. Semi-conductors

Link to the periodic Table • Which elements do you believe would be considered conductors? • Which elements do you believe would be considered insulators? • Which elements do you believe would be considered semi-conductors?

Conductors • Substances that allow the free flow of electrons • Usually metals (from the periodic table) because they are willing to give up their valence electrons • Can also be an electrolytic solution – electrolyte is any substance containing free ions that make the substance electrically conductive

What is an ion? • An ion is an atom that as become electrically charged by losing or gaining one or more electrons

Periodic Table and ions

Insulator • Substance that inhibits (prevents) the free flow of electrons, because they want to hold on to their valence electrons. • Once charged, the charges will stay in a specific location • Often non-metals as well as wood, plastic, glass, ceramics, rubber, silk and air

Semi-conductors • Substance in which the conductivity depends on the conditions • Metalloids and carbon are examples of semi-conductors

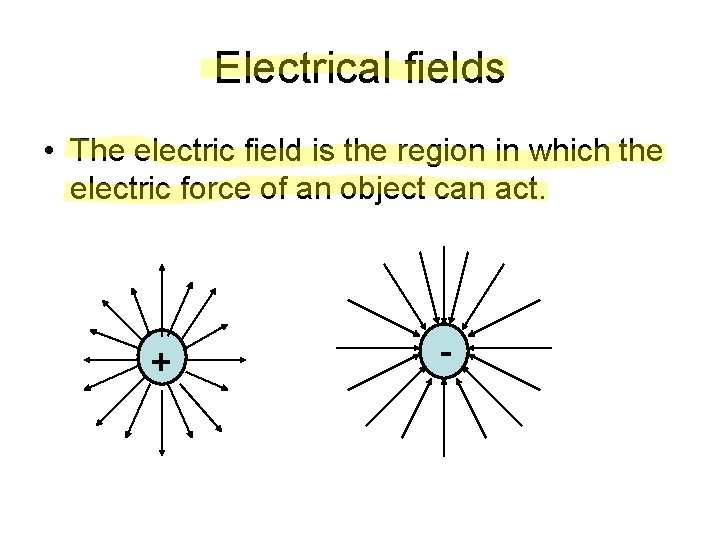
Electrical fields • The electric field is the region in which the electric force of an object can act. + -



Static Electricity • All phenomena related to electric charges at rest • Over time an electrically charged body can loose its charge gradually • In other cases, an object can recover its neutral state quickly through an electrostatic discharge.

Hey Look!!! A new carpet



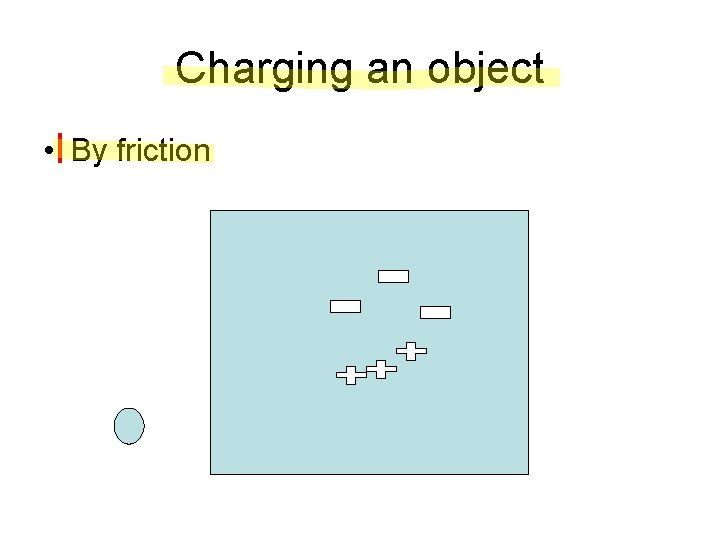
Charging an object • By friction

Charging an object • By friction – Start with two neutral objects – When rubbing the two objects together, electrons from one object will be ripped off by the atoms of the second object – End two objects with opposite charge

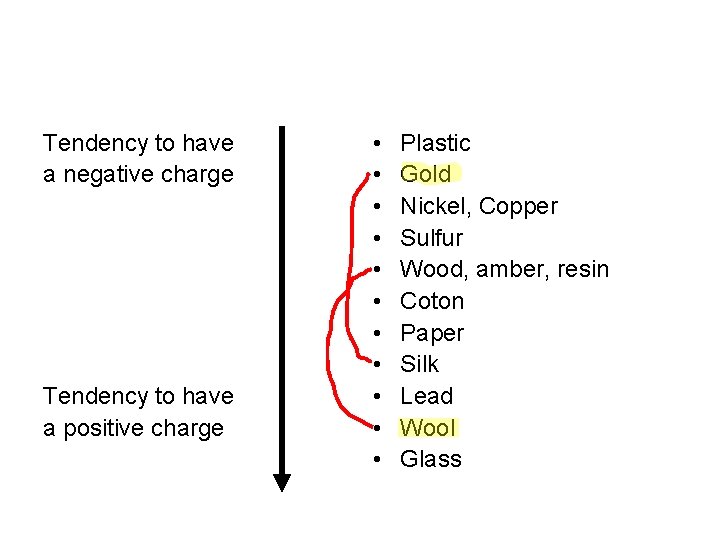
Tendency to have a negative charge Tendency to have a positive charge • • • Plastic Gold Nickel, Copper Sulfur Wood, amber, resin Coton Paper Silk Lead Wool Glass

Charging an object • Conduction – One charged object touches a neutral object – The Charged object shares its charge with the neutral object – We are left with two objects of the same charge

Charging an object • Induction – One neutral, one charged – Without touching the objects together, the charges in the neutral object will separate – One charged, one partially charged