Electricity Principles Applications Sixth Edition Richard J Fowler
















- Slides: 38

Electricity Principles & Applications Sixth Edition Richard J. Fowler Chapter 1 Basic Concepts © 2003 Glencoe/Mc. Graw-Hill

INTRODUCTION • Work and Energy • Atoms • Ions • Charge

Work Requires Energy

Work (W) consists of a force moving through a distance. Energy (W) is the capacity to do work. The joule (J) is the base unit for both energy and work. The amount of work done equals the amount of energy used (converted). Fifty joules of energy are required to do fifty joules of work.

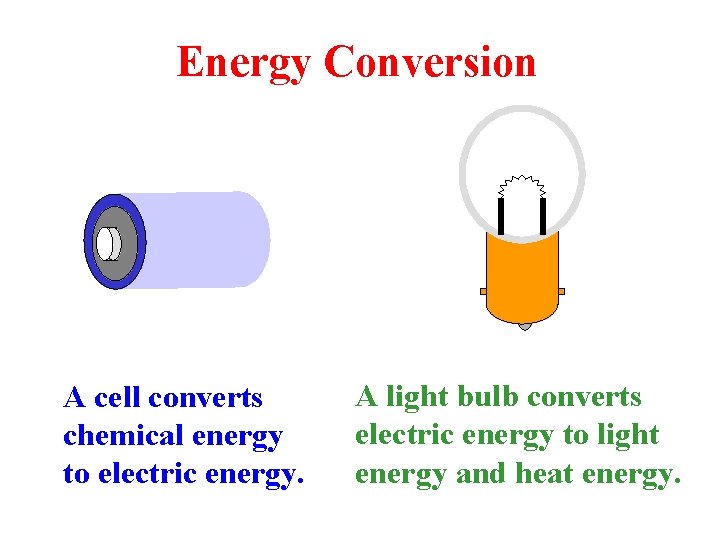
Energy Conversion A cell converts chemical energy to electric energy. A light bulb converts electric energy to light energy and heat energy.

Energy Quiz The _____ is the base unit of energy. joule The symbol for the base unit of work is the letter _____. J The italicized upper-case _____ is the symbol for energy. W Doing forty joules of work requires _____ joules of energy. forty A light bulb converts _____ energy into _____ energy and _____ energy. electric light, heat

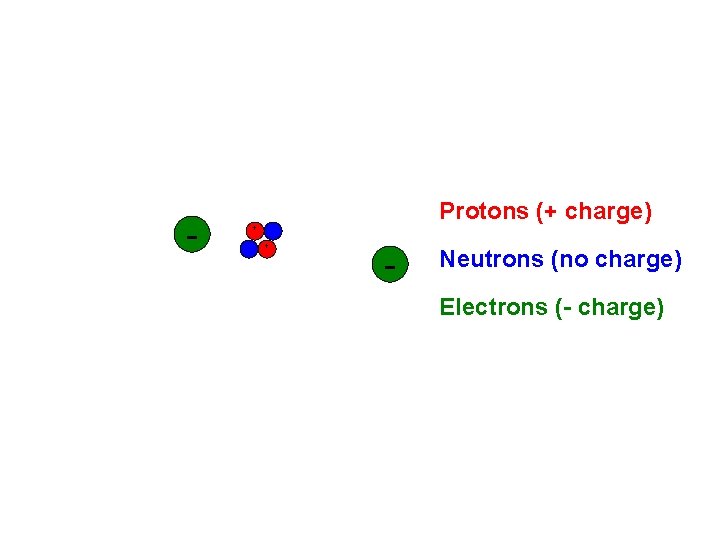
Particles of the Helium Atom

+ Protons (+ charge) +

+ Protons (+ charge) + Neutrons (no charge)

- + Protons (+ charge) + - Neutrons (no charge) Electrons (- charge)

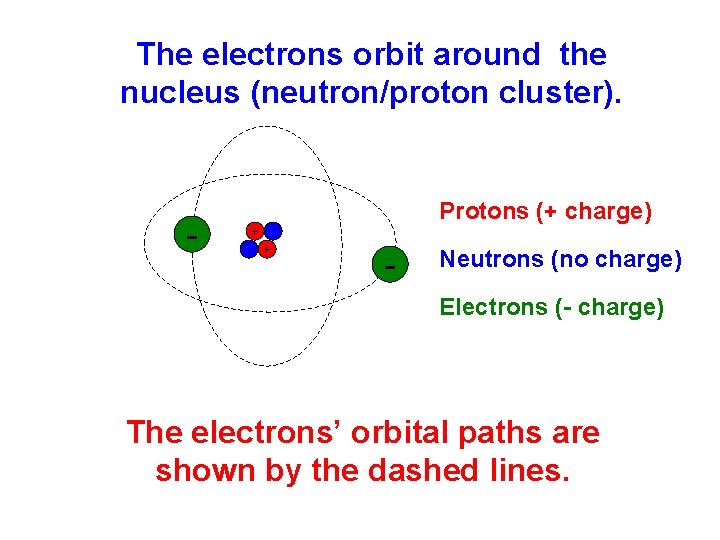
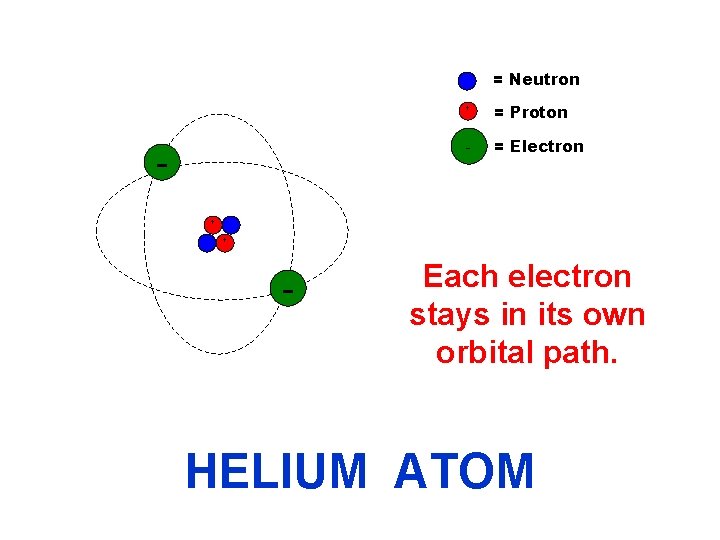
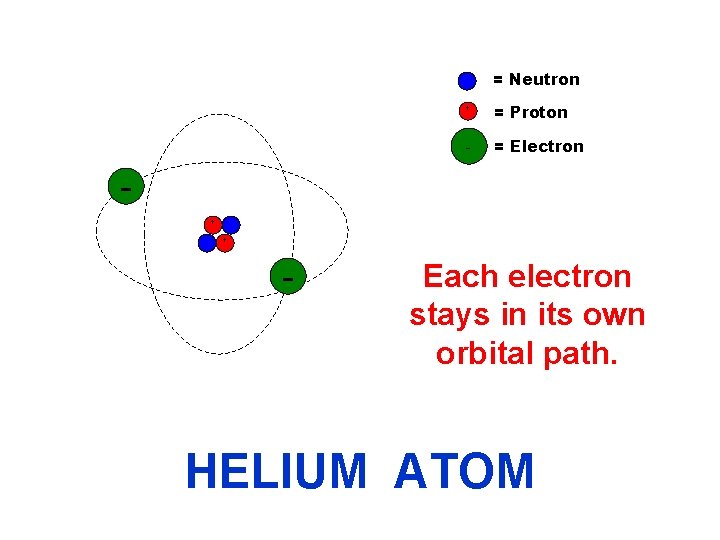
The electrons orbit around the nucleus (neutron/proton cluster). - Protons (+ charge) + + - Neutrons (no charge) Electrons (- charge) The electrons’ orbital paths are shown by the dashed lines.

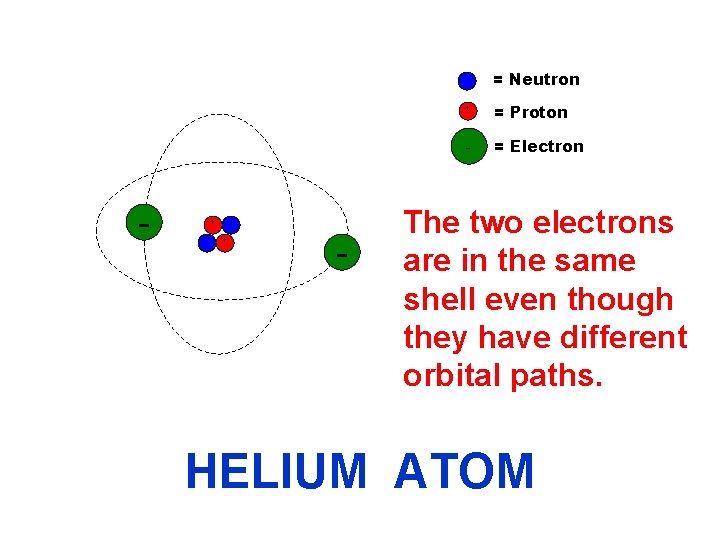
= Neutron - + + - + = Proton - = Electron The two electrons are in the same shell even though they have different orbital paths. HELIUM ATOM

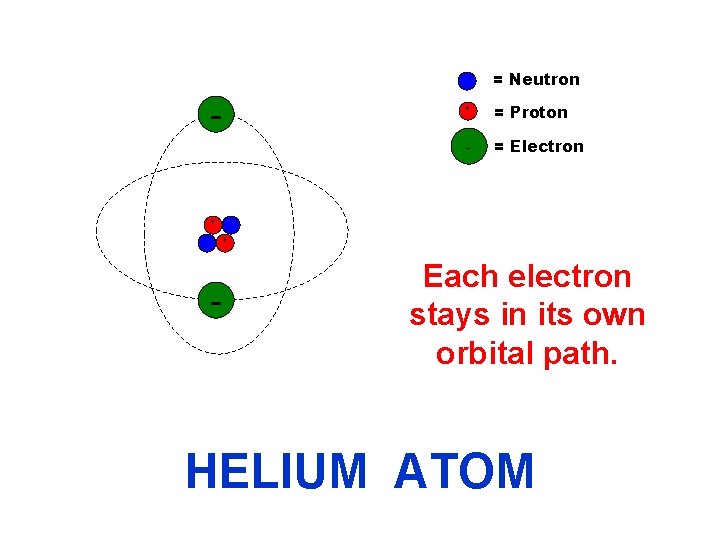
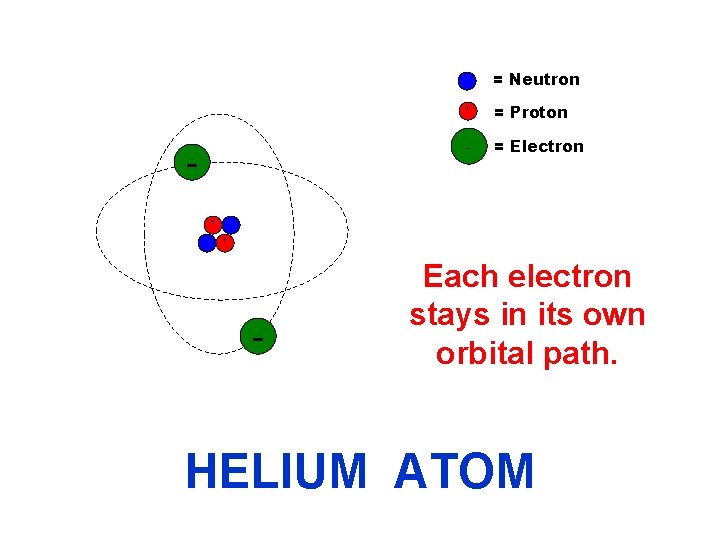
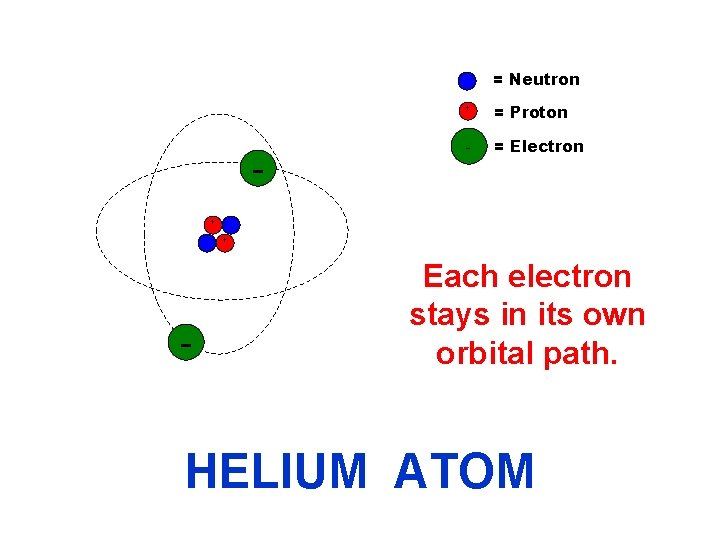
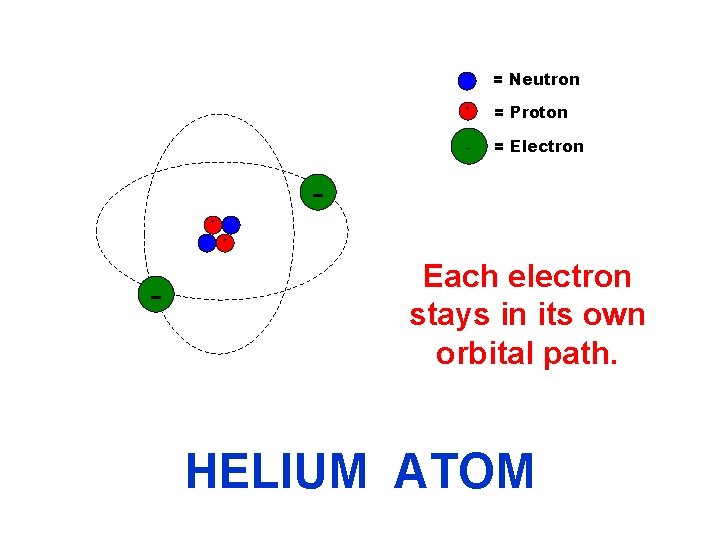
= Neutron + + = Proton - = Electron + - Each electron stays in its own orbital path. HELIUM ATOM

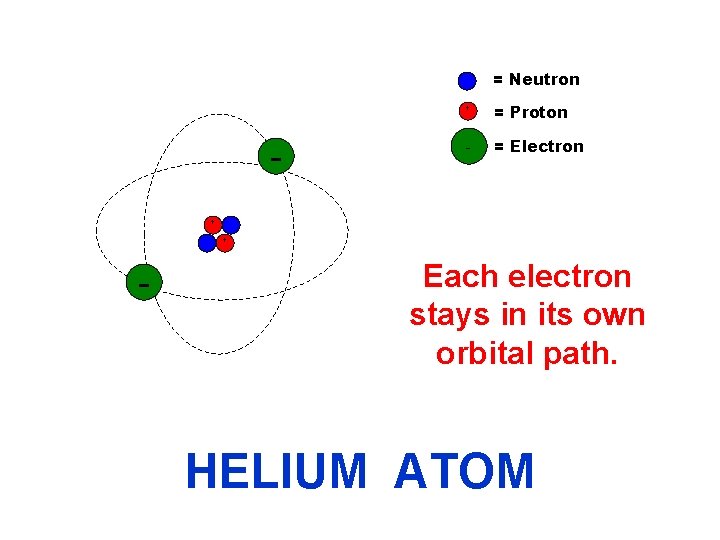
= Neutron + - + = Proton - = Electron + Each electron stays in its own orbital path. HELIUM ATOM

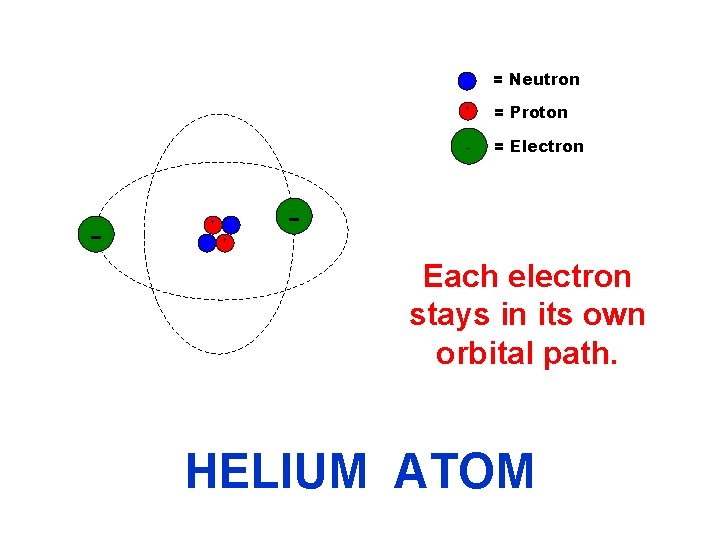
= Neutron + - + = Proton - = Electron + Each electron stays in its own orbital path. HELIUM ATOM

= Neutron - + + + = Proton - = Electron Each electron stays in its own orbital path. HELIUM ATOM

= Neutron + = Proton - = Electron + + - Each electron stays in its own orbital path. HELIUM ATOM

= Neutron + + = Proton - = Electron + - Each electron stays in its own orbital path. HELIUM ATOM

= Neutron + - + = Proton - = Electron + Each electron stays in its own orbital path. HELIUM ATOM

= Neutron + = Proton - = Electron + - + Each electron stays in its own orbital path. HELIUM ATOM

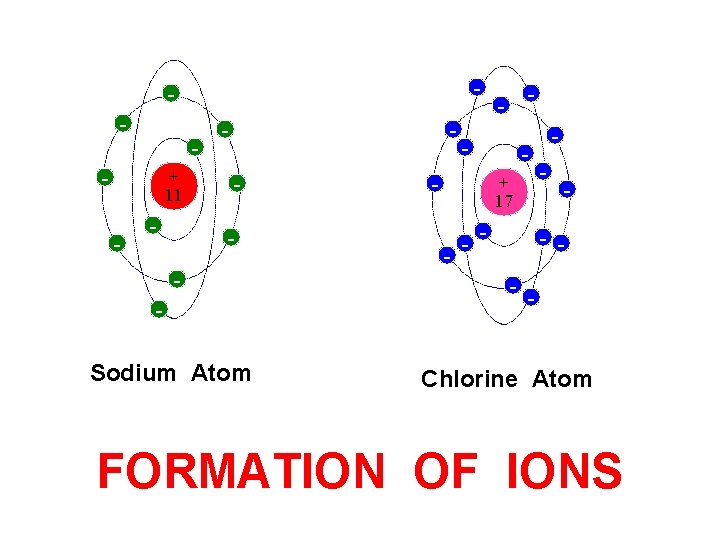
Formation of Ions from. Atoms

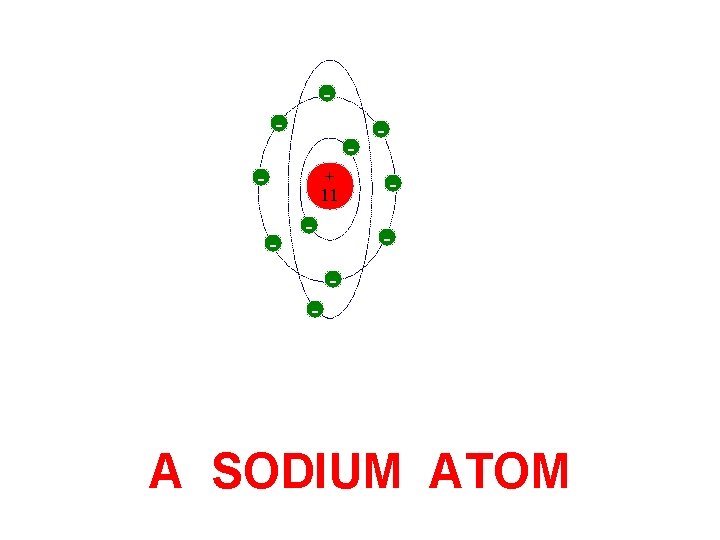
- - - + 11 - - A SODIUM ATOM

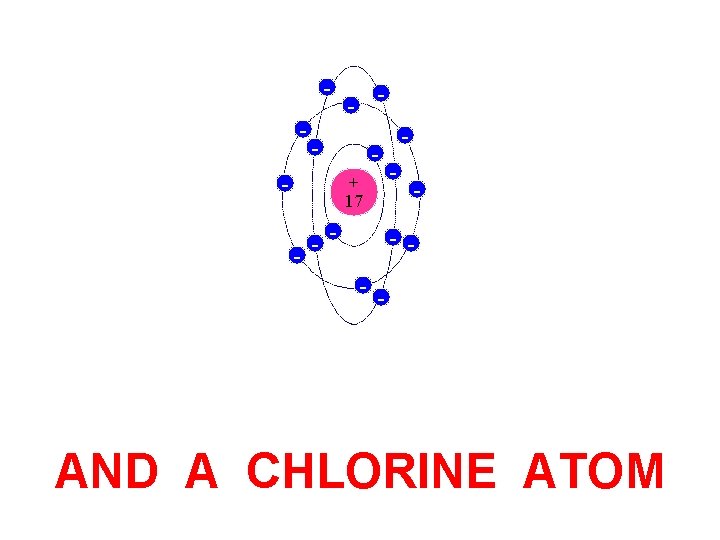
- - - + 17 - - - AND A CHLORINE ATOM

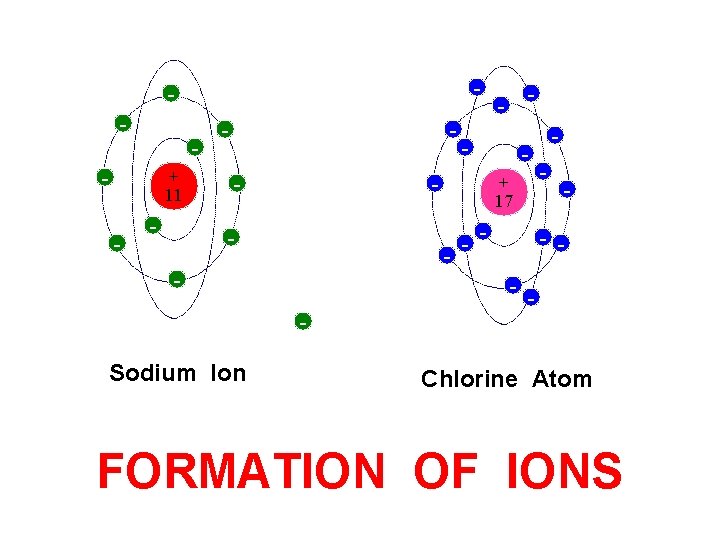
- - + 11 - - - Sodium Atom - - + 17 - - - Chlorine Atom FORMATION OF IONS

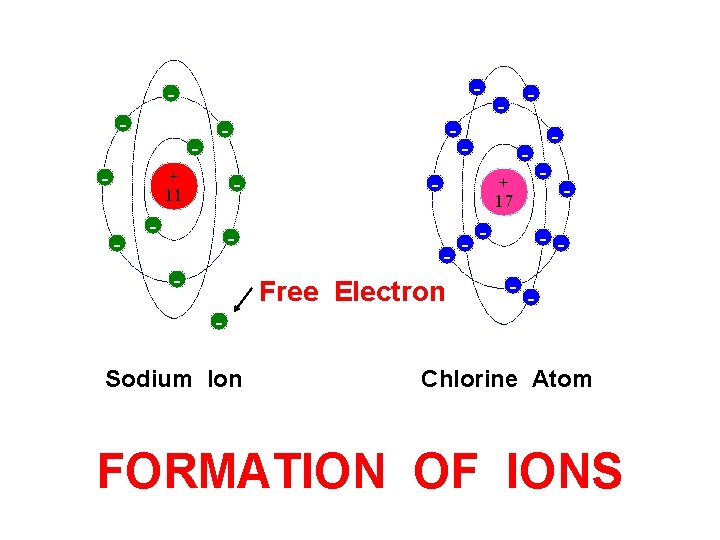
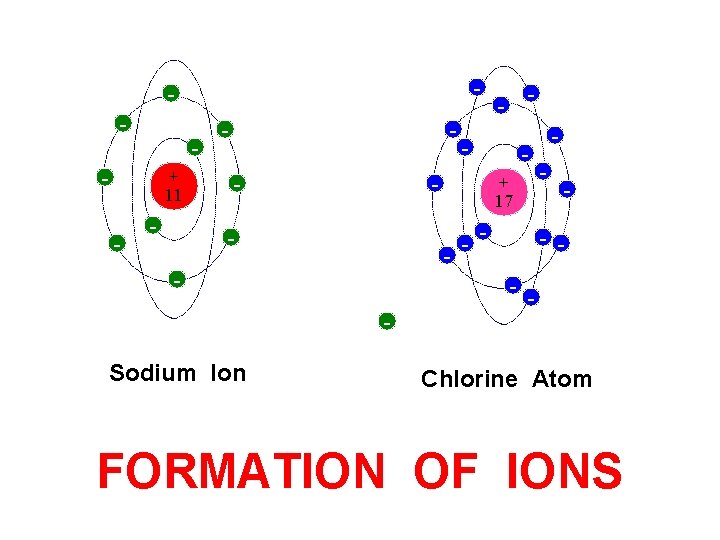
- - - + 11 - - Sodium Ion - - + 17 - Free Electron - - - - Chlorine Atom FORMATION OF IONS

- - + 11 - - - + 17 - - Sodium Ion - - - Chlorine Atom FORMATION OF IONS

- - + 11 - - - + 17 - - Sodium Ion - - - Chlorine Atom FORMATION OF IONS

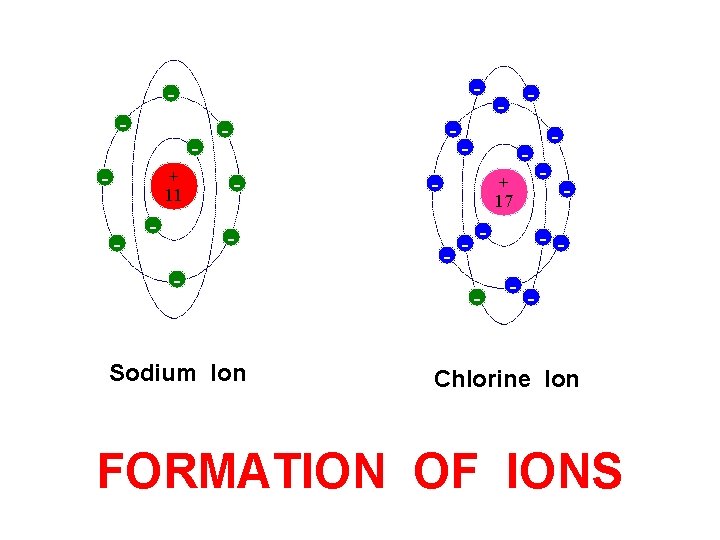
- - + 11 - - - Sodium Ion - - - + 17 - - - Chlorine Ion FORMATION OF IONS

Static Charges

Force Between Electric Charges _ charge _ charge Like charges produce a repelling force. When physically released, they repel each other.

Charged rod. (Many negative charges have been removed from the rod. ) +++ + + + + --- + +++

_ +_ +_ +_+_+_+ +_+_ Uncharged ball suspended on a thread. (Equal number of negative and positive charges uniformly distributed. )

Ball receives small induced charge as rod approaches. (Note clustering of like charges on ball’s surface. ) Weak force of attraction develops. +_ +_ _ +++_+_ _ + _+ _ +++ + + -+++ + + + + --- + +++

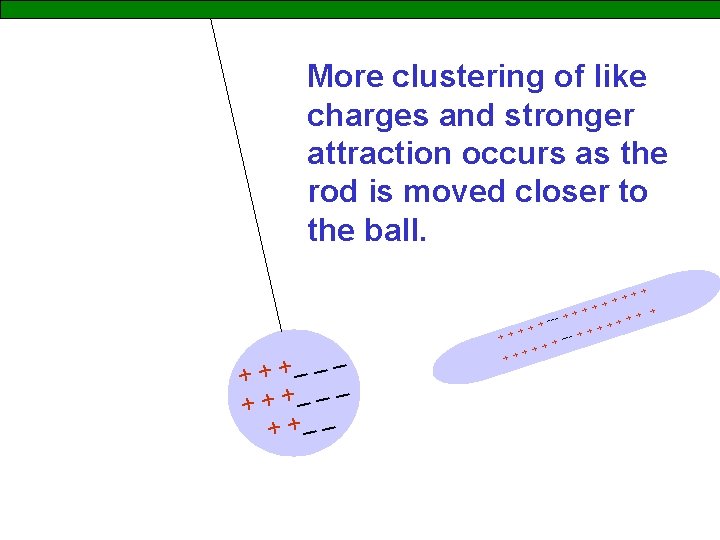
More clustering of like charges and stronger attraction occurs as the rod is moved closer to the ball. __ _ + + + _ _ _ + ++ + +_ _ +++ + ++ ++ + + --- + +++

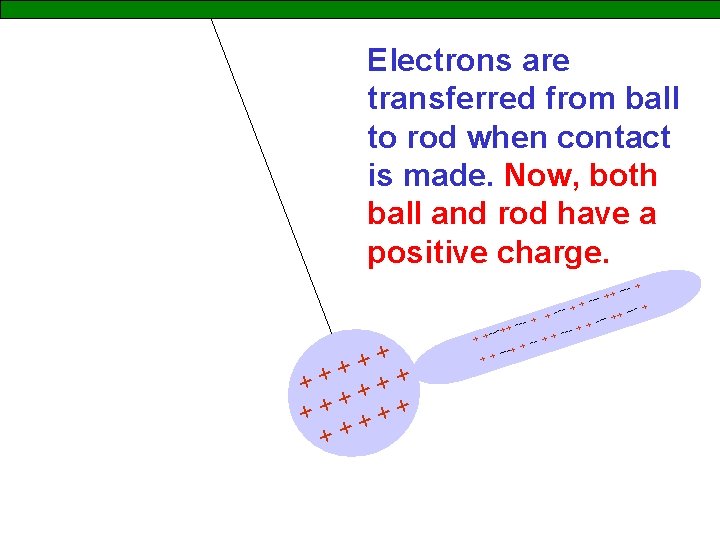
Electrons are transferred from ball to rod when contact is made. Now, both ball and rod have a positive charge. + ++ +++ + + + + -- + + + - ++ --- + + ---+ + + +-- + + + ---

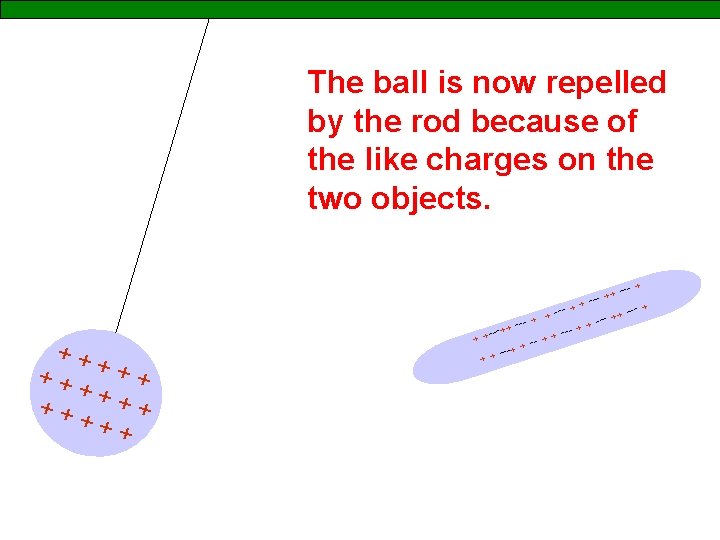
The ball is now repelled by the rod because of the like charges on the two objects. ++ ++ ++ + -- + + + - ++ --- + + ---+ + + +-- + + + ---

Charge Quiz Electrons have a _____ electric charge. negative Protons have a _____ electric charge. positive The _____ is electrically neutral. neutron The charge of a sodium ion is _____. positive A chlorine ion has a(n) _____ of electrons. excess Like charges _____ each other. repel

REVIEW • • Work and Energy Atoms Ions Charge