Electricity Lesson 10 Fruity Batteries Learning Intention By

Electricity Lesson 10 Fruity Batteries

Learning Intention By the end of this lesson I will Develop my knowledge of building batteries Success Criteria Explain how electricity can be produced when different metals are used as electrodes, with an electrolyte between them. Construct a battery using metal electrodes and a piece of fruit to investigate how electricity can be produced in a cell/battery. • Compare voltages produced from different combinations of metals in a simple chemical cell.

Starter Questions Decide if the following statements describe a parallel or a series circuit: 1. 2. 3. 4. 5. 6. Has one path for current to flow Voltage is the same at every point Has branches for additional components Current is the same at every point If one bulb breaks in this circuit, all others would go off Would be better suited to your hall lights if you wanted to be able to use 2 different switches to control a light

Starter Answers Decide if the following statements describe a parallel or a series circuit: 1. 2. 3. 4. 5. Has one path for current to flow SERIES Voltage is the same at every point PARALLEL Has branches for additional components PARALLEL Current is the same at every point SERIES If one bulb breaks in this circuit, all others would go off SERIES 6. Would be better suited to your hall lights if you wanted to be able to use 2 different switches to control a light PARALLEL

Introduction to Batteries • At the start of this topic we discussed how a power supply can come from a battery or mains electricity. • In our investigations so far we have used batteries to power our circuits. • What is an advantage of using batteries over mains electricity? • Can you think of a disadvantage?

Investigating Batteries • Batteries are a useful way to make electricity but can be expensive. They are used mainly to power portable electronic devices such as i. Pods and mobile phones. • There is a wide range of batteries of different sizes and types for different uses. • Some batteries such as mobile phone batteries can be recharged so that they work again. • Batteries produce only a few volts, while mains electricity produces 230 volts. Therefore, batteries operate at lower voltages and are safer than mains electricity.

Energy Stores • Batteries are filled with chemicals that store energy. • Batteries transform chemical energy into electrical energy • A battery makes an electric current until the chemicals inside it are used up. (This is when rechargeable ones would need plugged in!) • Most commercial batteries are made from two different bits of metal (called electrodes) immersed in a chemical solution called an electrolyte. • These are useful in making batteries as they are materials that allow electricity to pass through them.

Using Batteries • Batteries make devices portable and can be safer than mains electricity but are expensive. • Most batteries are made from two different bits of metal (called electrodes) immersed in a chemical solution called an electrolyte.

Activity 1: Fruity Batteries • We know we can make a battery from using 2 metal electrodes and an electrolyte. • An electrolyte is simply a solution that can conduct electricity. • Fruit juice contains an electrolyte and so you should be able to make a battery from a piece of fruit.

Activity 1: THINK • Consider the battery above. We want to try and produce the largest voltage. What could we change? What could we investigate?
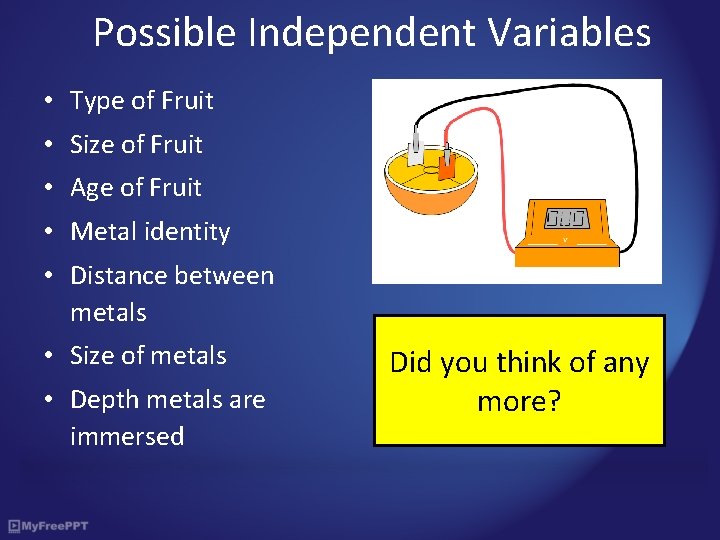
Possible Independent Variables • Type of Fruit • Size of Fruit • Age of Fruit • Metal identity • Distance between metals • Size of metals • Depth metals are immersed Did you think of any more?

Activity 1: Fruity Batteries What you need: A piece of fruit (citrus fruits work well), metal strips, two wires, two crocodile clips and a voltmeter What you do: • Push the two bits of metal foil into a piece of fruit. • Use the crocodile clips to connect the metal foils to different terminals of the voltmeter • Measure the voltage, V • Repeat the measurements with combinations of metals

Activity 1: Fruity Batteries Metal 1 Metal 2 Voltage (V) Magnesium Copper Zinc Copper Iron Copper Tin Copper From your results, can you tell which combination of metals makes the best battery?

Plenary Activity –Guess! Choose one of the words/terms from today’s lesson listed below. Explain the word to your partner. They must guess which key word you are explaining! Copper Expensive Safer Voltage Electrode Battery Electrolyte Voltmeter Magnesium Portable

Summary • Batteries are portable but expensive. They tend to be deemed safer than mains electricity. • Most batteries are made from 2 pieces of metal (electrodes) and a solution that can conduct electricity (electrolyte). • You can make a simple battery using two bits of metal and a piece of fruit • Different pairs of metals produce different voltages

Learning Intention By the end of this lesson I will Develop my knowledge of building batteries Success Criteria Explain how electricity can be produced when different metals are used as electrodes, with an electrolyte between them. Construct a battery using metal electrodes and a piece of fruit to investigate how electricity can be produced in a cell/battery. • Compare voltages produced from different combinations of metals in a simple chemical cell.
- Slides: 16