Electric Forces and Fields Chapter 17 Section 17



















- Slides: 20

Electric Forces and Fields Chapter 17

Section 17 -1 Objectives n Understand charge the basic properties of electric n Differentiate insulators n Distinguish between conductors and between charging by contact, charging by induction and charging by polarization

Electric Charge What’s happening to this little girl?

Electric Charge n There are two kinds of electric charge – Positive and negative – Like charges repel – Unlike charges attract

Electric Charge n Electric charge is conserved!! – What else is conserved? ? § Mass, Energy, Momentum – Electric charge cannot be created or destroyed but it can be transferred – Example: Transferring electrons from someone’s hair to a balloon

Electric Charge n Electric Charge is quantized – One unit of charge : e= 1. 60219 x 10 -19 C § C stands for Coulomb, the unit of electric charge – A proton has a charge of +1. 60 x 10 -19 C – An electron has a charge of -1. 60 x 10 -19 C

Conductors & Insulators n Conductors: Materials in which electric charges move freely – Examples: most metals n Insulators: Materials in which electric charges do not move freely – Examples: Plastic, glass, silk, rubber

Charging by contact n The two objects are rubbed together and electrons are transferred from one to the other – electrons from the fur are transferred to the rod

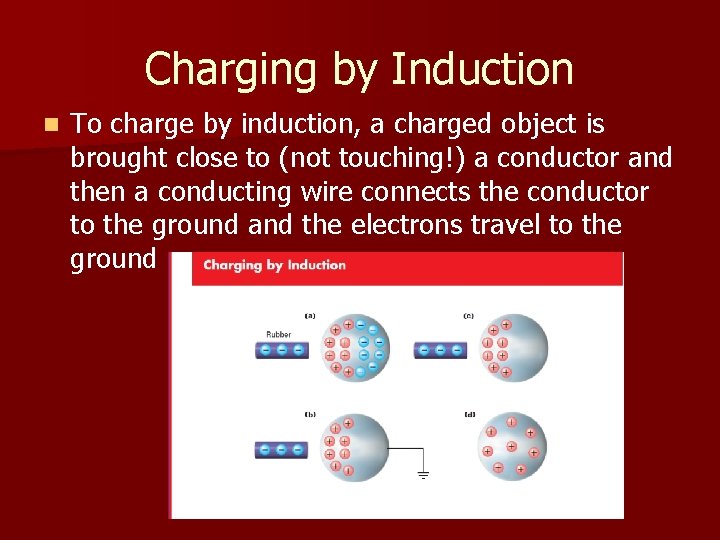
Charging by Induction n To charge by induction, a charged object is brought close to (not touching!) a conductor and then a conducting wire connects the conductor to the ground and the electrons travel to the ground

Charging by Polarization n Charging charge by polarization creates a surface – A charged object is brought close to an insulator and the electrons and protons realign themselves to create one side that is more positive and one that is more negative

Section 17. 2 Objectives n Calculate electric force using coulomb’s law n Compare electric force with gravitational force n Apply the superposition principle to find the resultant force on a charge and to find the position at which the net force on a charge is zero

Coulomb’s Law n Coulomb’s Law describes the mathematical relationship between electric force, distance and electric charge for two objects k. C= 8. 99 x 109 Nm 2 C 2 Electric force= Coulomb’s Constant x (charge 1)(charge 2) distance 2

Coulomb’s Law n The force between two charges is proportional to the magnitude of the charges n The force between two charges is inversely proportional to the distance 2 between them

Coulomb’s Law n Remember that force is a vector! – For problems involving two charges, the direction is either “attractive” or “repulsive” – i. e. the direction of the force between a positive charge and negative charge is attractive and the direction of the force between two negative charges is repulsive

Example Problem p. 636 #2 n Two identical conducting spheres are placed with their centers 0. 30 m apart. One is given a charge of +12 x 10 -9 C and the other is given a charge of -18 x 10 -9 C – A. Find the electric force exerted on one sphere by the other – B. The spheres are connected by a conducting wire. After equilibrium has occurred, find the electric force between the two spheres.

p. 636 #2 (part a) n Use Coulomb’s Law

p. 636 #2 (part b) n What does it mean “after equilibrium has occurred”? – The charge on each sphere is the same

Things to Remember n Electric charge is conserved n Conductors and insulators can be charged by contact n Conductors can be charged by induction n Insulators can have a surface charge due to polarization n Electric force is a vector!


Sources n Balloon Hair Pic: http: //heyheyheyparty 365. blogspot. com/2 011/01/static-electricity. html n http: //startswithabang. com/? p=1643 n http: //blogs. nature. com/news/2011/06/res earchers_studying_static_ge. html