Electric Charges and Fields The Amber Effect Ancient

Electric Charges and Fields

The Amber Effect Ancient Greeks discovered that when amber (fossilized tree sap – like a plastic) is rubbed, it will attract various substances like cloth fibers, fine wood shavings, paper, debris or lint. Amber’s attracting of these materials (when rubbed) was called the “amber effect”.

Elecktron The Greek word for amber is elecktron, the root of the English words, electron and electricity. William Gilbert in the 1600 s used the term electrics to refer to substances (like amber and glass) that when rubbed, would attract bits of paper and such. Substances that when rubbed (like metals) did not attract paper bits were called nonelectrics.

Static Electricity: Insulators and Conductors Today we call the amber effect static electricity. We call substances insulators (Gilbert’s electrics) if they will attract paper bits when rubbed. Substances that do not attract paper bits when rubbed are called conductors (Gilbert’s nonelectrics). Insulators Conductors

Static Charge When insulators are rubbed, picking up paper bits, they are said to possess a static charge (A charge that is stationary on the insulator, not readily moving out of the insulator).

Electroscopes An electroscope is a device used to detect static electric charges on an object. There are two main kinds of electroscope – the pith ball electroscope and the leaf electroscope.
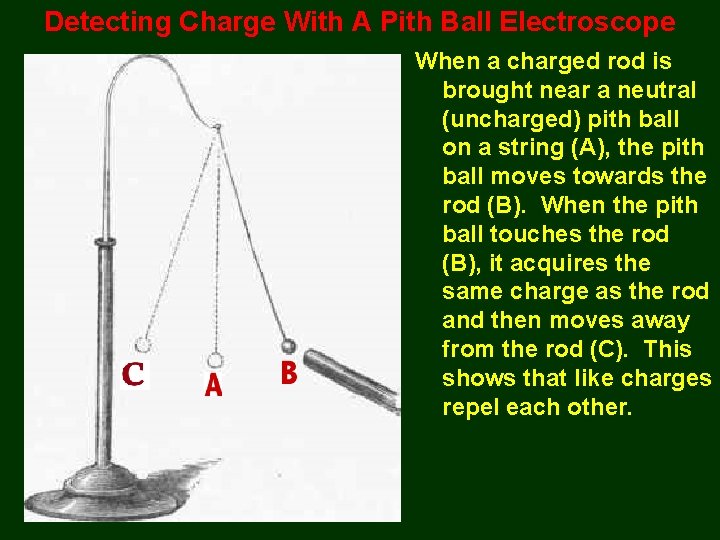
Detecting Charge With A Pith Ball Electroscope When a charged rod is brought near a neutral (uncharged) pith ball on a string (A), the pith ball moves towards the rod (B). When the pith ball touches the rod (B), it acquires the same charge as the rod and then moves away from the rod (C). This shows that like charges repel each other.
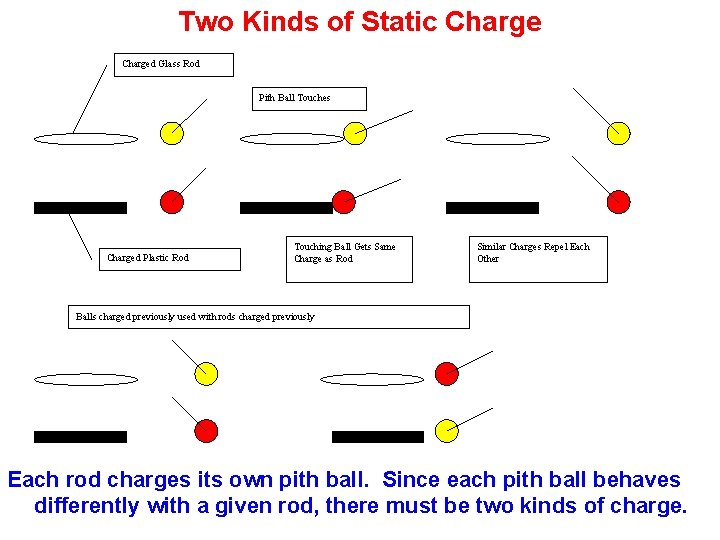
Two Kinds of Static Charged Glass Rod Pith Ball Touches Charged Plastic Rod Touching Ball Gets Same Charge as Rod Similar Charges Repel Each Other Balls charged previously used with rods charged previously Each rod charges its own pith ball. Since each pith ball behaves differently with a given rod, there must be two kinds of charge.

Positive and Negative Charges The words positive and negative refer to the two kinds of electric charge. A glass rod when rubbed with a silk cloth becomes positively charged while a plastic rod rubbed with a wool cloth becomes negatively charged. Glass Rod Plastic Rod Wool Silk
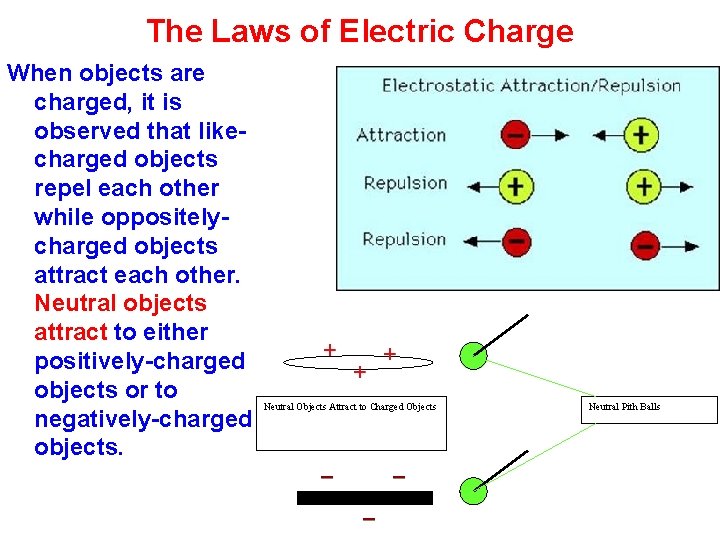
The Laws of Electric Charge When objects are charged, it is observed that likecharged objects repel each other while oppositelycharged objects attract each other. Neutral objects attract to either positively-charged objects or to negatively-charged objects. Neutral Objects Attract to Charged Objects Neutral Pith Balls
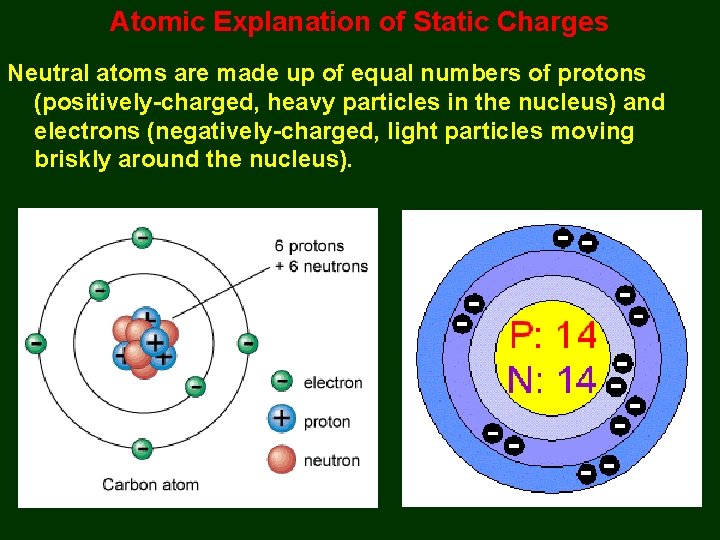
Atomic Explanation of Static Charges Neutral atoms are made up of equal numbers of protons (positively-charged, heavy particles in the nucleus) and electrons (negatively-charged, light particles moving briskly around the nucleus).

Atomic Explanation of Static Charges In solids, protons (positive charges) are fixed and do not move while electrons (negative charges) can move (to one side of the atom or from one atom to another atom, depending on the substance). Atom holding its electrons around itself. Atom allowing its electrons to wander to neighboring atoms.

How Insulators and Conductors Differ Atomically Atoms in insulators hold their electrons around their nuclei with no free electron movement from atom to atom while atoms in conductors electrons are shared and move freely from atom to atom. Charged objects brought close to insulators cause their electrons to move towards one side of their atoms but charged objects brought close to conductors cause their electrons to move in mass to one side of the object, giving rise to a large charge separation which makes one side oppositely charged to the other side.

Different Atoms Exert Different Attractions For Electrons Atoms that have electrons closer to their nuclei or that have more protons in their nuclei tend to attract electrons more strongly than those atoms having electrons in higher energy levels or having fewer protons in their nuclei. The nitrogen atom (left) has less electron pull than the oxygen atom (right) since the oxygen atom has the same number of energy levels but has one more proton and electron which provide more attraction. The phosphorus atom (right) has less electron pull since its outer electrons are farther from the nucleus (one energy level farther out).

Close Contact of Different Substances (Rubbing) When different solid objects come into close contact as in rubbing them against each other, electrons from one substance commonly move into the other substance. This happens because the different atoms of each substance attract electrons either more strongly or weakly. In the example below, the atoms in the silk cloth attract electrons more strongly than the atoms in the glass rod. This uneven attraction for electrons leaves the rod overall positive and the silk cloth overall negative.
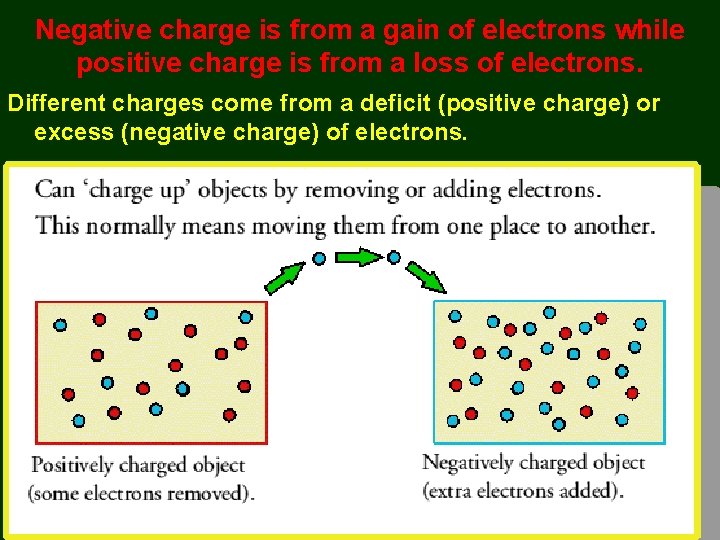
Negative charge is from a gain of electrons while positive charge is from a loss of electrons. Different charges come from a deficit (positive charge) or excess (negative charge) of electrons.

Law of Conservation of Electric Charge When neutral objects are charged by rubbing, the net charge produced on both objects together is zero. In other words, the amount of positive charge produced on one object is equal to the amount of negative charge produced on the other object. Their sum is zero due to their opposite signs.
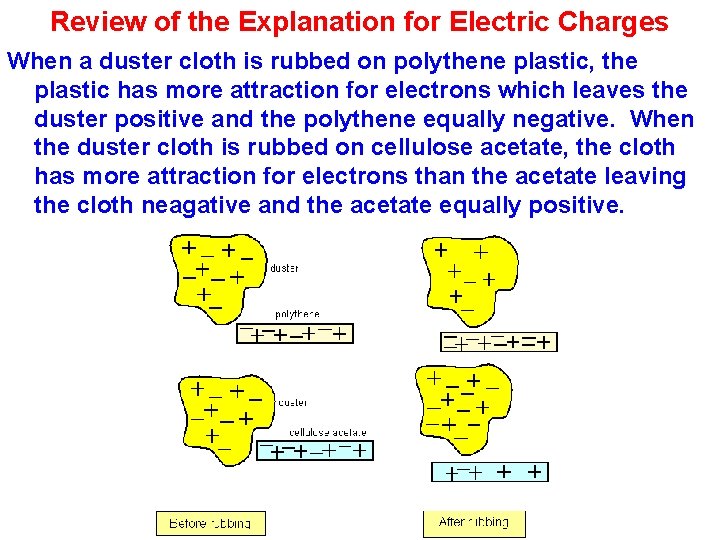
Review of the Explanation for Electric Charges When a duster cloth is rubbed on polythene plastic, the plastic has more attraction for electrons which leaves the duster positive and the polythene equally negative. When the duster cloth is rubbed on cellulose acetate, the cloth has more attraction for electrons than the acetate leaving the cloth neagative and the acetate equally positive.
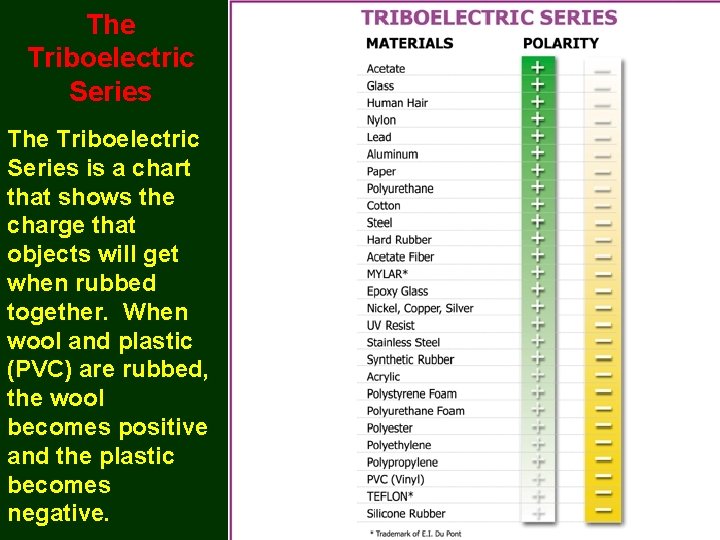
The Triboelectric Series is a chart that shows the charge that objects will get when rubbed together. When wool and plastic (PVC) are rubbed, the wool becomes positive and the plastic becomes negative.
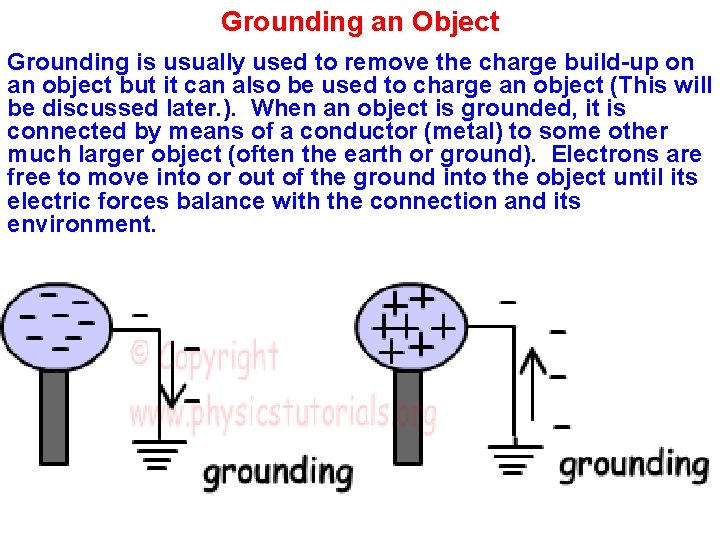
Grounding an Object Grounding is usually used to remove the charge build-up on an object but it can also be used to charge an object (This will be discussed later. ). When an object is grounded, it is connected by means of a conductor (metal) to some other much larger object (often the earth or ground). Electrons are free to move into or out of the ground into the object until its electric forces balance with the connection and its environment.

Grounding an Object Grounding removes a charge as long as the grounding object is larger than the discharging object. For example, a tiny pith ball is neutralized when a person touches it (the person’s body is huge compared to the tiny pith ball. )
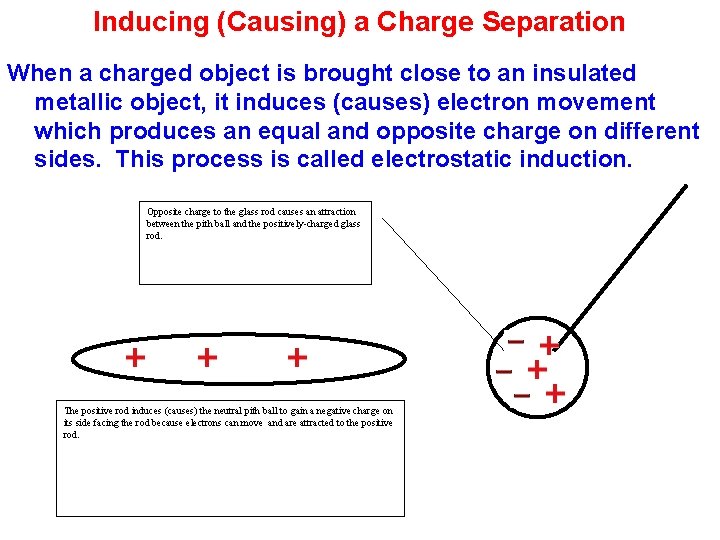
Inducing (Causing) a Charge Separation When a charged object is brought close to an insulated metallic object, it induces (causes) electron movement which produces an equal and opposite charge on different sides. This process is called electrostatic induction. Opposite charge to the glass rod causes an attraction between the pith ball and the positively-charged glass rod. The positive rod induces (causes) the neutral pith ball to gain a negative charge on its side facing the rod because electrons can move and are attracted to the positive rod.

How a Charged Comb Picks Up Paper Bits The negatively-charged comb induces neutral paper bits to get positive ends facing the comb (negative electrons move away from or are repelled by the negative comb). The positive paper ends are then attracted to the (–) comb.
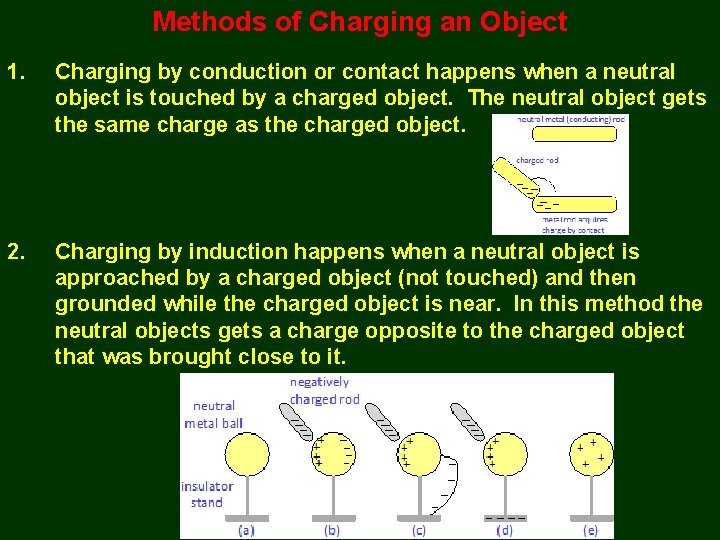
Methods of Charging an Object 1. Charging by conduction or contact happens when a neutral object is touched by a charged object. The neutral object gets the same charge as the charged object. 2. Charging by induction happens when a neutral object is approached by a charged object (not touched) and then grounded while the charged object is near. In this method the neutral objects gets a charge opposite to the charged object that was brought close to it.

Bringing a Charged Object to a Leaf Electroscope A rod is brought close to the end knob of a leaf electroscope. As it gets close to the electroscope, the rod repels electrons down (if it is negative) or attracts electrons up (if it is positive). Since the bottom leaves are both negative (if the rod is negative) or both positive (if the rod is positive), they repel each other and separate.

Removing a Charge by Grounding A leaf electroscope is charged negatively. A person touches the electroscope, gounding it. Electrons leave through the hand until the electroscope is neutral and the leaves drop.

Charging a Leaf Electroscope by Induction The negative balloon is brought close to the knob of the electroscope. This repels electrons from the knob. When the electroscope is grounded, the electrons are repelled by the balloon out of the electroscope (leaving it overall positive). When the ground is broken and then the balloon removed, the electroscope remains positively-charged (now with the leaves spread), opposite to the charge of the balloon.

Measuring Electrical Charge The unit measuring electrical charge is called the coulomb (C), named in honor of Charles Coulomb who investigated the laws of electrical charge. The symbol used in formulas for charge is Q or q. One coulomb is the amount of charge on 6. 2415 x 10 18 electrons (for a negative coulomb) or on 6. 2415 x 10 18 protons (for a positive coulomb) etc … … etc

The Elementary Charge A charge on a single proton or a single electron is referred to as the elementary charge. The value of an electron’s charge is – 1. 6022 x 10 -19 C and the value of a proton’s charge is + 1. 6022 x 10 -19 C. The magnitude or size of the charge on an electron and proton is the same even though their sizes and masses differ greatly. electron
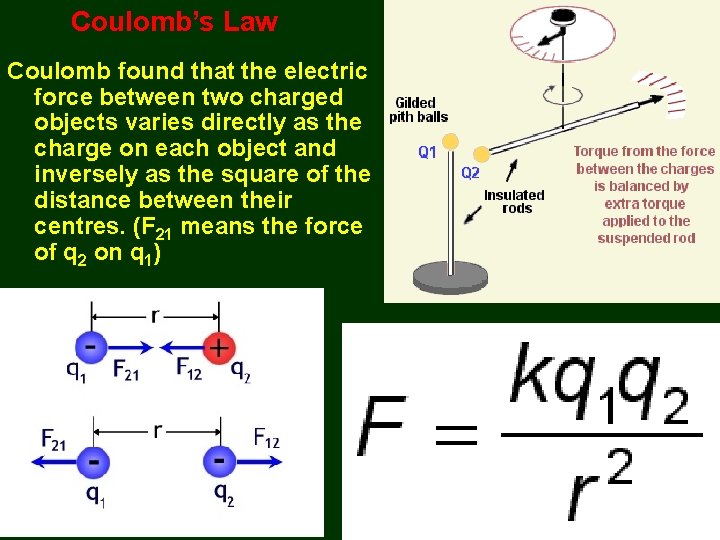
Coulomb’s Law Coulomb found that the electric force between two charged objects varies directly as the charge on each object and inversely as the square of the distance between their centres. (F 21 means the force of q 2 on q 1)

Similar Form: Coulomb’s Law and Gravitation Law Coulomb’s Law for charges has the same form as Newton’s Law of Gravitation. The k constant of Coulomb’s Law is 8. 988 x 109 Nm 2/C 2 The G constant of Newton’s Law is 6. 674 x 10 -11 Nm 2/Kg 2 For everyday objects and charges that we handle, gravity is typically a much weaker force than charge forces.

Getting a Sense of Charge Size and Force Size A charge of +1 C and – 1 C placed 1 m apart will produce an attractive force of 8. 998 x 109 N. This is a force of about a million tons! We normally do not encounter charges as large as a coulomb. Rubbing a comb typically produces a charge of about 1 microcoulomb (10 -6 C).

Problem 1 Determining Electric Force Determine the electric force on an electron in a hydrogen atom (has one proton) if the average orbit radius is 0. 53 x 10 -10 m. F = (9. 0 x 109 Nm 2/C 2)(1. 6 x 10 -19 C) (0. 53 x 10 -10 M)2 = 8. 2 x 10 -8 N (in a direction toward the centre)

Problem 2 Determining Electric Forces Q 1 , a charge of 50 µC is 1 m from Q 2 , a charge of 1 µC Which is larger in magnitude, the force that Q 1 exerts on Q 2, or the force that Q 2 exerts on Q 1? Q 1 ------1 m------- F 21 = k(50 µC)(1 µC) (1 m)2 Q 2 F 12 = k(1 µC)(50 µC) (1 m) 2 The forces are equal as would be expected from Newton’s 3 rd Law

3: Determining Force with three linear charges Given three colinear charges, Q 1 (-8. 0 µC), Q 2 (+3. 0 µC), Q 3 (-4. 0 µC) with the distances as shown, determine the net electrostatic force on Q 3. Q 1 (-8. 0 µC) <--0. 30 m-- Q 2 (+3. 0 µC) <-0. 20 m Q 3 (-4. 0 µC) The net force on Q 3, FNet is F 31 + F 32 F 31 = (9. 0 x 109 Nm 2/C 2)(8. 0 µC)(4. 0 µC) = 1. 2 N to the right (repulsion) F 32 (0. 50 m)2 = (9. 0 x 109 Nm 2/C 2)(3. 0 µC)(4. 0 µC) = 2. 7 N to the left (attraction) (0. 20 m)2 2. 7 N 1. 2 N Q 3 F = 1. 2 N – 2. 7 N = -1. 5 N (Where – is left and + is right) = 1. 5 N left
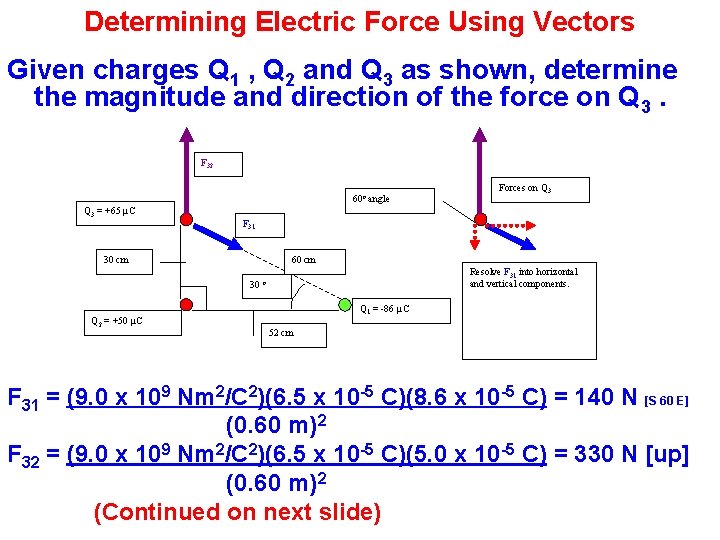
Determining Electric Force Using Vectors Given charges Q 1 , Q 2 and Q 3 as shown, determine the magnitude and direction of the force on Q 3. F 32 60 o angle Q 3 = +65 µC F 31 30 cm 60 cm Resolve F 31 into horizontal and vertical components. 30 o Q 2 = +50 µC Forces on Q 3 Q 1 = -86 µC 52 cm F 31 = (9. 0 x 109 Nm 2/C 2)(6. 5 x 10 -5 C)(8. 6 x 10 -5 C) = 140 N [S 60 E] (0. 60 m)2 F 32 = (9. 0 x 109 Nm 2/C 2)(6. 5 x 10 -5 C)(5. 0 x 10 -5 C) = 330 N [up] (0. 60 m)2 (Continued on next slide)
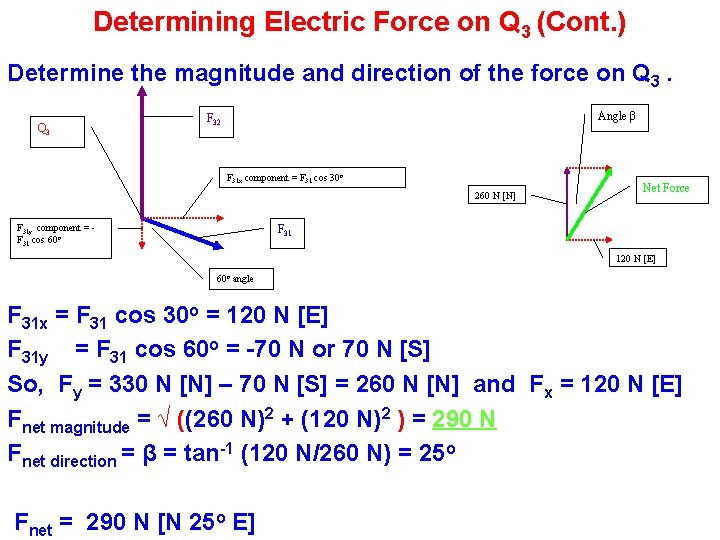
Determining Electric Force on Q 3 (Cont. ) Determine the magnitude and direction of the force on Q 3 Angle β F 32 F 31 x component = F 31 cos 30 o 260 N [N] F 31 y component = F 31 cos 60 o Net Force F 31 120 N [E] 60 o angle F 31 x = F 31 cos 30 o = 120 N [E] F 31 y = F 31 cos 60 o = -70 N or 70 N [S] So, Fy = 330 N [N] – 70 N [S] = 260 N [N] and Fx = 120 N [E] Fnet magnitude = √ ((260 N)2 + (120 N)2 ) = 290 N Fnet direction = β = tan-1 (120 N/260 N) = 25 o Fnet = 290 N [N 25 o E]

Experiments With Charges Reveal Invisible Force Fields When powder from an insulator is spread over the surface of a light oil and point charges are inserted into the oil, a force pattern around the charged point source is revealed. This pattern makes visible an invisible force field that exists around a charged object(s).

Electric Fields An electric field is a sphere of influence around a charged object. Electric field exert a force on another charged object. An electric field is a vector quantity and its direction is determined by the direction a positive point charge would move in the field. Between positive and negative charges, the field runs from positive to negative.
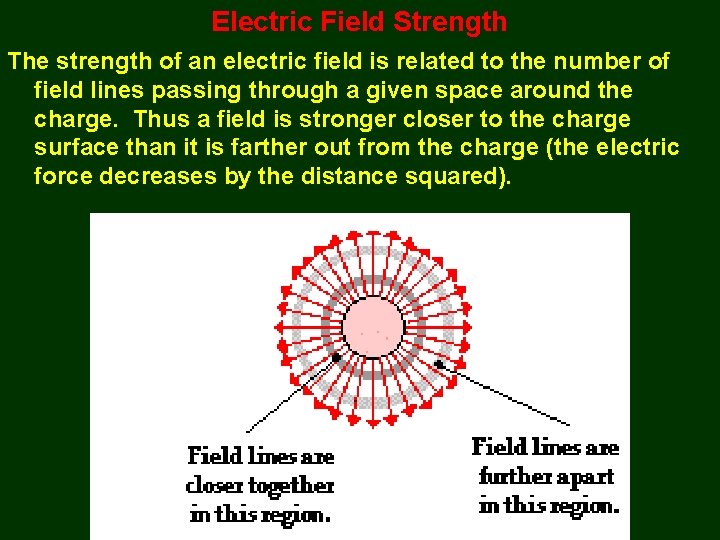
Electric Field Strength The strength of an electric field is related to the number of field lines passing through a given space around the charge. Thus a field is stronger closer to the charge surface than it is farther out from the charge (the electric force decreases by the distance squared).
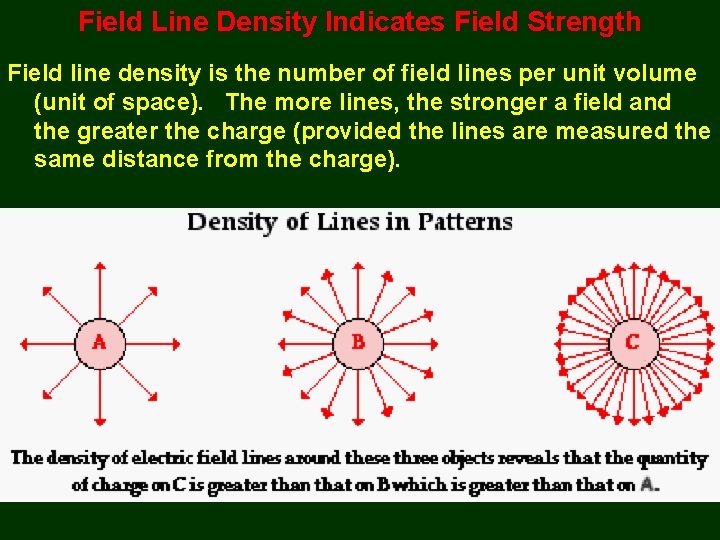
Field Line Density Indicates Field Strength Field line density is the number of field lines per unit volume (unit of space). The more lines, the stronger a field and the greater the charge (provided the lines are measured the same distance from the charge).
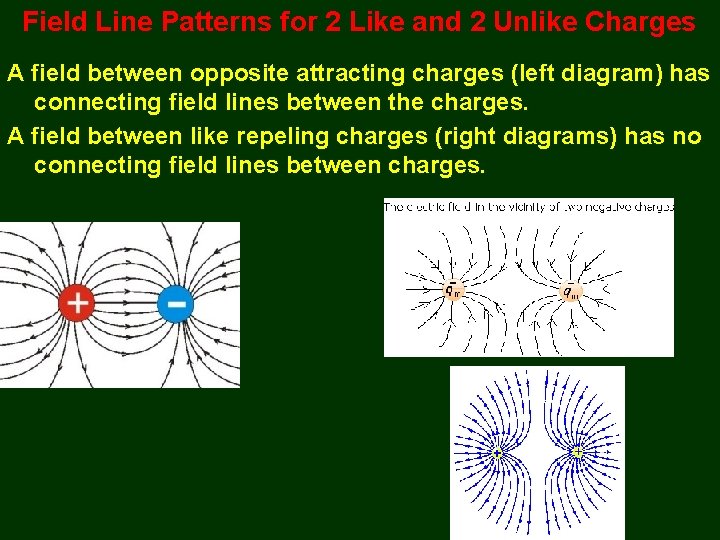
Field Line Patterns for 2 Like and 2 Unlike Charges A field between opposite attracting charges (left diagram) has connecting field lines between the charges. A field between like repeling charges (right diagrams) has no connecting field lines between charges.

The Vector Nature of an Electric Field Adding the vectors of two point charges at various points in space produces the field. Vector addition explains the shape of the field made by point charges.

Faraday’s Ice Pail Experiment Michael Faraday investigated many aspects of static and current electricity. In his “ice pail experiment” he discovered that the static charge on an object is distributed over the outside of an object, not on the inside.

Faraday’s Butterfly Bag Faraday found that if an insulated, cone-shaped conductor cloth is charged, the inside of the cone is neutral while the outside of the bag is charged. When strings are used to turn the bag inside out, the former inside (now the outside) becomes charged while the former outside (now inside) is neutral. This confirms that static electric charge resides on the outside of an object.

Electrostatic Shielding: Faraday’s Cage Faraday’s experiments suggested that an object could be shielded from electrostatic charge (electric fields) if a wire mesh cage or a metal container were to surround an object. Electronic components that needed to be shielded from electric fields are protected with such cages or metal containers.
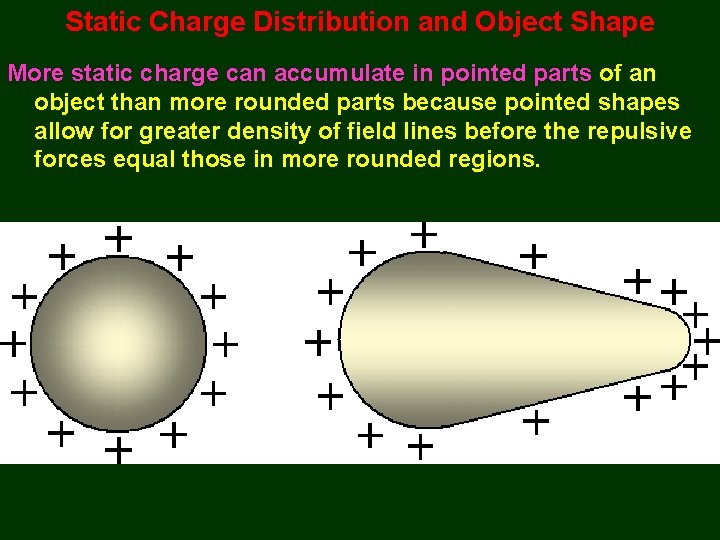
Static Charge Distribution and Object Shape More static charge can accumulate in pointed parts of an object than more rounded parts because pointed shapes allow for greater density of field lines before the repulsive forces equal those in more rounded regions.

Measuring Electric Field Strength -> Electric field strength (|E| or E) is defined as the force per unit charge at a given point in the field (Ex: A or B). Fe is the force in N on the charge, q. From this relation it can be seen that Fe = |E|q. This first formula for electric field strength is defined in terms of a test object in the field.

A Second Formula for Electric Field Strength Given object Q exerting a force on charge q (qt). The force between Q and q is given by F = k. Qq. r 2 Combining the formula from the previous slide with the formula above we obtain |E| = F or |E| = k. Qq = k. Q q ( r )2 r 2 q This new formula gives field strength in terms of the fieldproducing object. |E| = k. Q r 2

Permittivity (ε) is a measure of the resistance of a medium (called the dielectric) to an electric field being set up in the medium. It measures how readily a medium “permits” an electric field being set up in it. The relative permittivity (εr) of a substance is a comparison of its permittivity to the permittivity of a vacuum.
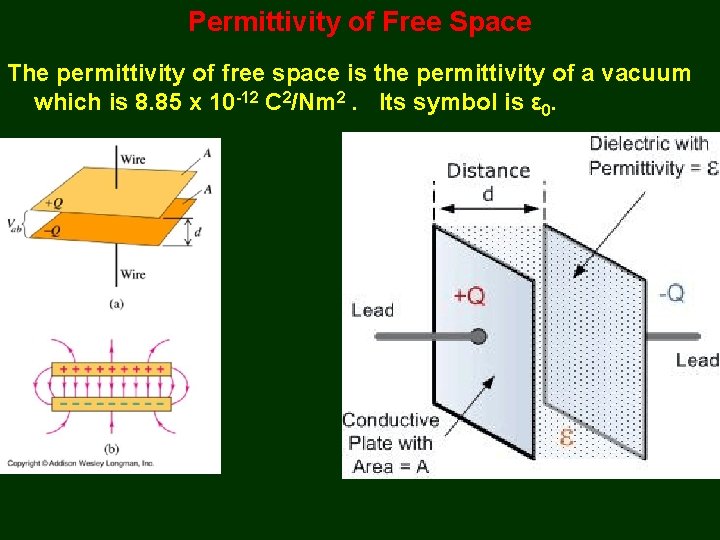
Permittivity of Free Space The permittivity of free space is the permittivity of a vacuum which is 8. 85 x 10 -12 C 2/Nm 2. Its symbol is ε 0.

Coulomb’s Constant and ε 0 Coulomb’s Law can be written with a constant that replaces k with ε 0. Knowing the value of k allows the calculation of ε 0. Thus, ε 0 = 1 4πk = 8. 85 x 10 -12 C 2/Nm 2.

Electric Field Problem

Electric Field Problem

Electric Field Problem A
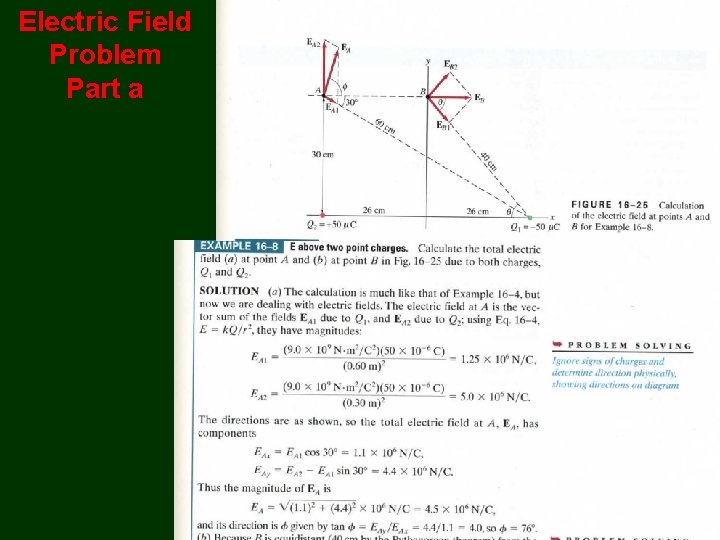
Electric Field Problem Part a
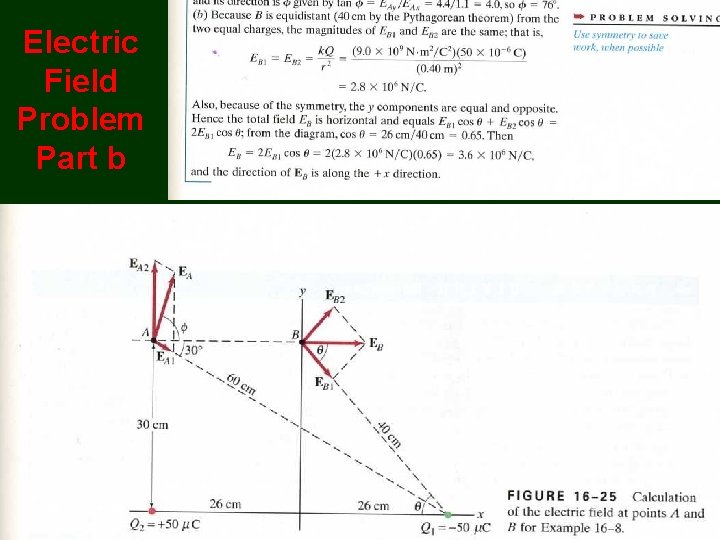
Electric Field Problem Part b

A A

A A

A A
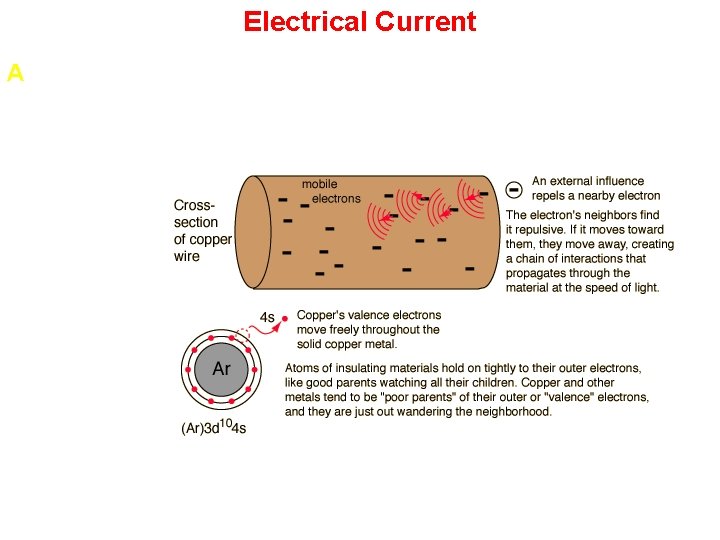
Electrical Current A
- Slides: 61