Electric Charge Static Electricity Static Electricity a stationary

















- Slides: 18

Electric Charge

Static Electricity ● Static Electricity : a stationary electrical charge typically produced by friction ● Works best on a dry day because excessive moisture can provide a pathway for charge to leak off a charged object.

Electric Charge ● Two Types of Electric Charge (distinguished by Benjamin Franklin) 1. Positive 2. Negative ● Like Charges Repel & Unlike Charges Attract

The Atom Every Atom is made up of 3 Subatomic Particles 1. Protons (positively charged) 2. Neutrons (uncharged) 3. Electrons (negatively charged)

Ion Formation ● When # protons = # electrons, the atom has no net charge ● Because electrons are outside the nucleus, they are easily transferred from one atom to another. ● When one or more electrons are transferred the atoms are no longer neutron (ions)

Conservation of Electric Charge ● Electric charge is not created or destroyed ● Most objects are electrically neutral (positive and negative charges are balanced) ● Friction makes these charges unbalanced. ○ Ballon gains electrons (-) ○ Hair loses electrons (+) ● This build up of charge can be released through a circuit.

Electric Charge is Quantized ● The SI unit of electric charge is the coulomb (C) ● Electric charge is quantized. This means that charge occurs as integer multiples of e in nature. (e= 1. 6 x 10^-19 C) Particle Charge (C) Mass (kg) Proton +1. 6 x 10^-19 1. 67 x 10^-27 Electron -1. 6 x 10^-19 9. 11 x 10^-31 Neutron 0 1. 67 x 10^-27

Materials are classified based on their ability to transfer electric charge ● Conductors : materials in which electric charges can move freely ○ Ex: copper, aluminum, human body ● Insulators : materials in which charges cannot move freely ○ Ex: glass, rubber, silk, plastic ● Semiconductors : materials that share properties of both conductors and insulators ○ Ex: Silicon, Germanium

Transfer of Electric Charge **demos here**--transfer charge by touching Electric Charge can be transferred from one object to another 3 ways: 1. Contact 2. Induction 3. Polarization

Transfer of Electric Charge by Contact Conductors contact. and Insulators can become charged by

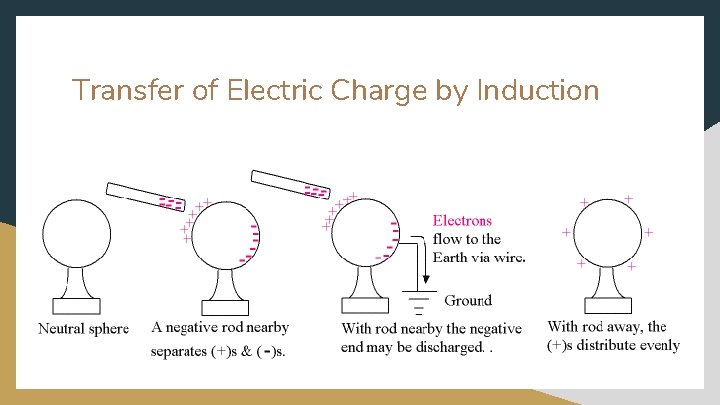
Transfer of Electric Charge by Induction Step 1 : a charged object is brought near ( conductor. without touching Step 2 : The presence of the charge object will force (or i the neutral conductor to move. Step 3 : Ground the conductor to ) a neutral nduce ) electrons within discharge electrons Step 4 : Remove charged object and grounding wire. The conductor is left with a (+) charge. ● ● Grounded-when a conductor is connected to Earth by a conducting wire Earth is an infinite reservoir for electrons

Transfer of Electric Charge by Induction

Transfer of Electric Charge by Polarization: the realignment of charge within individual molecules creating an i nduced charge on the surface of an insulator. Both objects still have a net charge of zero Ex: “Static Cling”

Electric Force ● Field Force: a force that is exerted by one object on another even though there is no physical contact between the two objects. (Ex. Electric Force, Gravitational Force) ● Two charged objects near one another may experience acceleration either toward or away from each other because each object exerts an Electric Force on the other object. ● Qualitatively determined by the type of electric charge ○ Like charges repel ○ Unlike charges attract What determines the quantitative value (magnitude) of this force?

Coulomb’s Law The magnitude of the Electric Force between objects is affected by… 1. The distance between the two objects 2. The amount of charge on the objects ● F=Electric Force [N] ● k= Coulomb’s Law Constant (k=9. 0 x 10^9 Nm^2/C^2) ● q 1=charge 1 [C] ● q 2=charge 2 [C] ● r=distance [m]

Coulomb’s Law Practice #1 The electron and proton of a hydrogen atom are separated, on average, by a distance of about 5. 3 x 10^-11 m. Find the magnitudes of the electric force and the gravitational force that each particle exerts on the other.

Coulomb’s Law Practice #2 Consider three point charges at the corners of a triangle, as shown, where q 1=6 x 10^-9 C, q 2=-2 x 10^-9 C, and q 3=5 x 10^-9 C. Find the magnitude and direction of the resultant force on q 3.

Coulomb’s Law Practice #3 When charged objects are said to be in equilibrium, the forces are equal in magnitude, but opposite in direction. Essentially the electric forces cancel each other out and the Net Electric Force is zero. Three charges lie along the x-axis. One positive charge, q 1=15 µC, is at x=2. 0 m, and another positive charge, q 2=6. 0 µC, is at the origin. At what point on the x-axis must a negative charge, q 3, be placed so that the resultant force on it is zero?