El mtodo cientfico cmo elaborar un proyecto de

El método científico: cómo elaborar un proyecto de investigación Alejandro Piris Giménez, Ph. D Scientific Research Manager Instituto de Oncología Vall d’Hebron (VHIO)

¿Do I have what I need to propose research? - Novelty (Idea – Proof of Concept) Capability (CV, team, management, facilities) Additional resources (co-financing) Expected impacts (in Health)

Principal Investigator/ Partner © Vall d'Hebron Institute of Oncology (VHIO) Evaluator / Panels
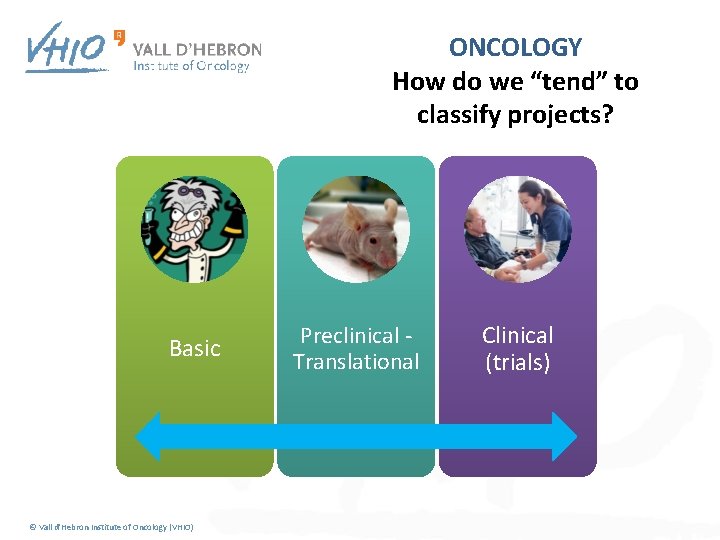
ONCOLOGY How do we “tend” to classify projects? Basic © Vall d'Hebron Institute of Oncology (VHIO) Preclinical - Translational Clinical (trials)

¿What do we need to design a good proposal? A good idea - Scientific Excellence / high potential of innovation - The Idea must adjust to the elegibility of the Call and local/ international policies A good writting, to: - Sell the idea/ Convince evaluators Collaborative projects: - A balanced consortium/ good collaborators

Realistic Vision– Utility Self-critic Vision– Competitors… Solidary Vision– Equal share of tasks among partners Well-dimensioned Vision– CV, timelines, resources Methodological Vision- Statistics Social Vision– Real need Market Vision– Novelty/ cost-efficacy © Vall d'Hebron Institute of Oncology (VHIO)
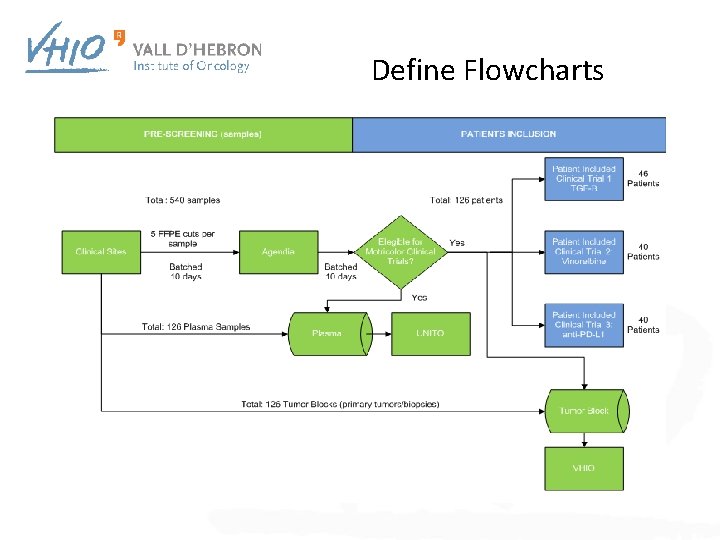
Define Flowcharts
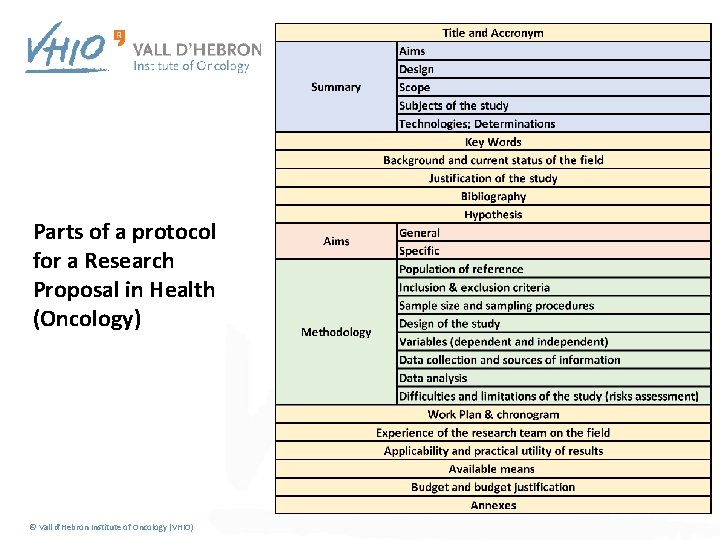
Parts of a protocol for a Research Proposal in Health (Oncology) © Vall d'Hebron Institute of Oncology (VHIO)

Example – Spanish Grant Número de Becas: 1 Dotación: 40. 000€ Beca FSEOM 2017 para Proyectos de Investigación Traslacional en Inmuno-oncología Requisitos de los solicitantes: Médicos Especialistas en Oncología Médica, con ejercicio profesional en España. Socio de SEOM y al corriente en el pago de la cuota de socio. Condiciones del proyecto: ü Proyecto de investigación original de 2 años de duración ü Área de investigación: temas de investigación clínico-traslacional relacionados con la inmunoterapia de tumores sólidos ü No se financiaran proyectos donde más del 50% del trabajo sea de carácter básico ü Proyectos de investigación coordinados o dirigidos en Unidades de Oncología Médica en España ü Debe indicar si el proyecto cuenta con financiación adicional y adjuntar documentación que lo acredite © Vall d'Hebron Institute of Oncology (VHIO)

Beca FSEOM 2017 para Proyectos de Investigación Traslacional en Inmuno-oncología Formato de la solicitud: Debe presentarse una Memoria del Proyecto de investigación, que no debe exceder de 12 páginas, y debe tener los siguientes apartados: Título Resumen (máximo 250 palabras) Introducción (máximo 1000 palabras) Indicar duración del Proyecto: máximo 2 años CV del Investigador principal y CV individual de todo el Equipo Investigador siguiendo plantilla SEOM o estructura de modelos normalizados Hipótesis y objetivos Plan de trabajo, protocolo y metodología Medios disponibles Adecuación a la prioridad de la convocatoria. Cronograma Presupuesto con justificación detallada Referencias bibliográficas © Vall d'Hebron Institute of Oncology (VHIO)
Example – EU Grant © Vall d'Hebron Institute of Oncology (VHIO)
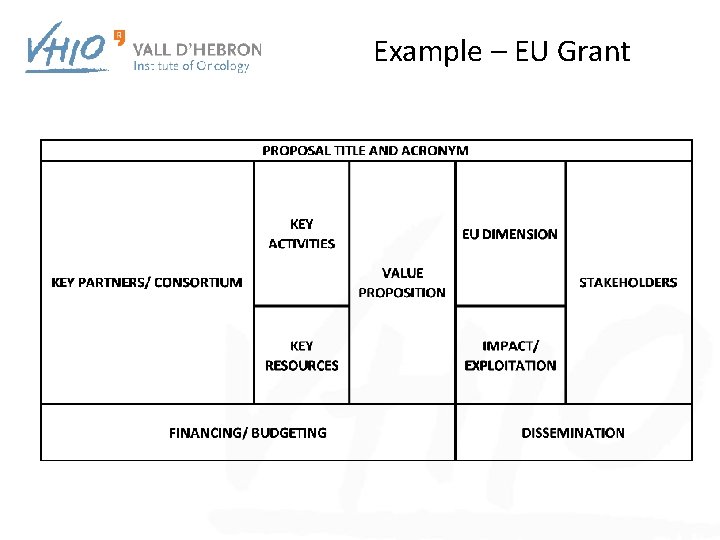
Example – EU Grant

Example of Clinical – Translational Work FIRST CLINICAL NEEDS Mo. Tri. Color © Vall d'Hebron Institute of Oncology (VHIO)
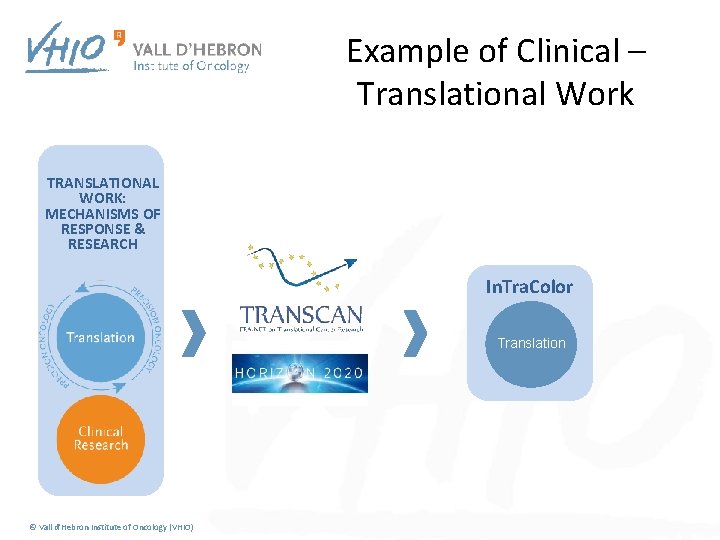
Example of Clinical – Translational Work First idea TRANSLATIONAL WORK: MECHANISMS OF RESPONSE & RESEARCH In. Tra. Color Translation © Vall d'Hebron Institute of Oncology (VHIO)
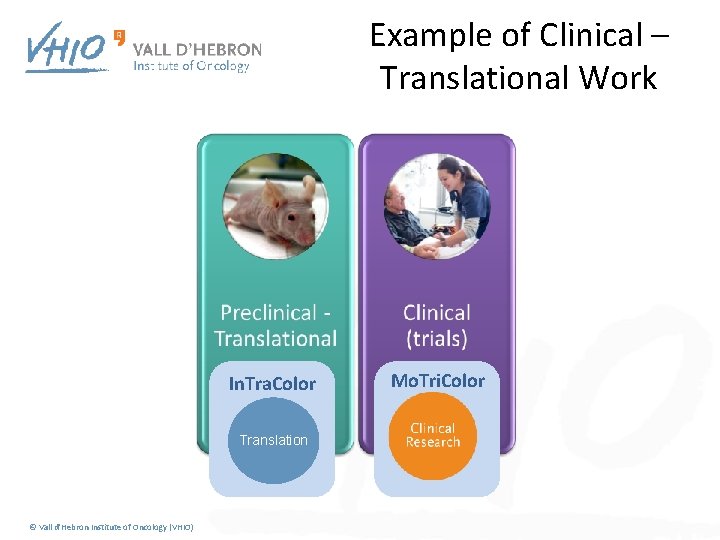
Example of Clinical – Translational Work In. Tra. Color Translation © Vall d'Hebron Institute of Oncology (VHIO) Mo. Tri. Color

Molecularly guided Trials with treatment strategies in patients with advances newly molecular defined subtypes od Colorectal cancer
Consortium
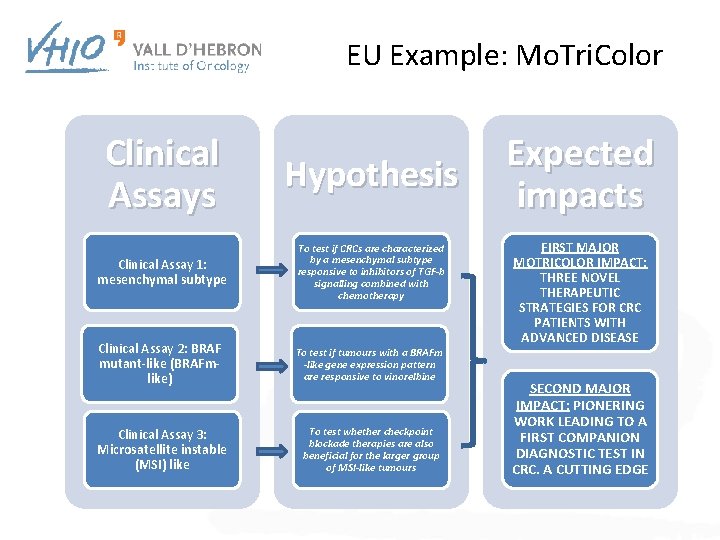
EU Example: Mo. Tri. Color Clinical Assays Hypothesis Clinical Assay 1: mesenchymal subtype To test if CRCs are characterized by a mesenchymal subtype responsive to inhibitors of TGF-b signalling combined with chemotherapy Clinical Assay 2: BRAF mutant-like (BRAFmlike) To test if tumours with a BRAFm -like gene expression pattern are responsive to vinorelbine Clinical Assay 3: Microsatellite instable (MSI) like To test whether checkpoint blockade therapies are also beneficial for the larger group of MSI-like tumours Expected impacts FIRST MAJOR MOTRICOLOR IMPACT: THREE NOVEL THERAPEUTIC STRATEGIES FOR CRC PATIENTS WITH ADVANCED DISEASE SECOND MAJOR IMPACT: PIONERING WORK LEADING TO A FIRST COMPANION DIAGNOSTIC TEST IN CRC. A CUTTING EDGE
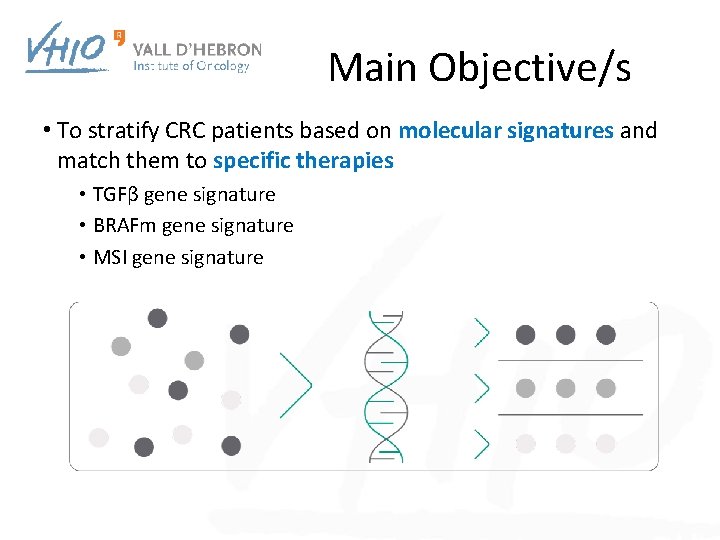
Main Objective/s • To stratify CRC patients based on molecular signatures and match them to specific therapies • TGFβ gene signature • BRAFm gene signature • MSI gene signature

Specific objectives • To perform 3 two-stage single arm multi center open-label phase II studies in molecularly selected patients: • To determine anti-tumour activity • To characterize the safety and tolerability • To determine other anti-tumour activity parameters (PFS, CB, OS) • To explore mechanism of response and resistance to the treatments • To monitor circulating DNA in blood

Impact • Three novel therapeutic strategies for CRC patients with advance disease – Mo. Tri. Color data: ground-breaking for further studies in CRC – Further clinical development in Europe • Pioneering work leading to a first companion diagnostic (predictive) test in CRC: a cutting edge – Agendia: worldwide positioning in cancer diagnostics • Extended impacts – New innovative and cost effective therapeutic approach for CRC patients – Pioneering work in identifying patients to most sensitive therapies and stratify them – Additional biomarkers of response prediction for CRC (Intra. Color)
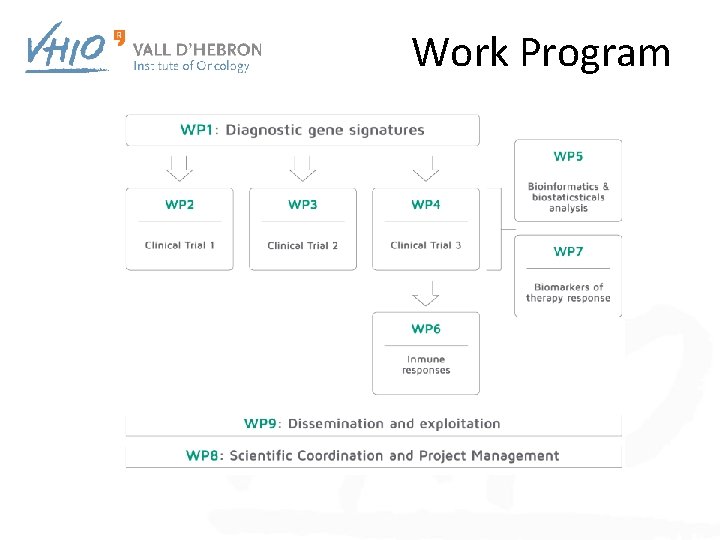
Work Program
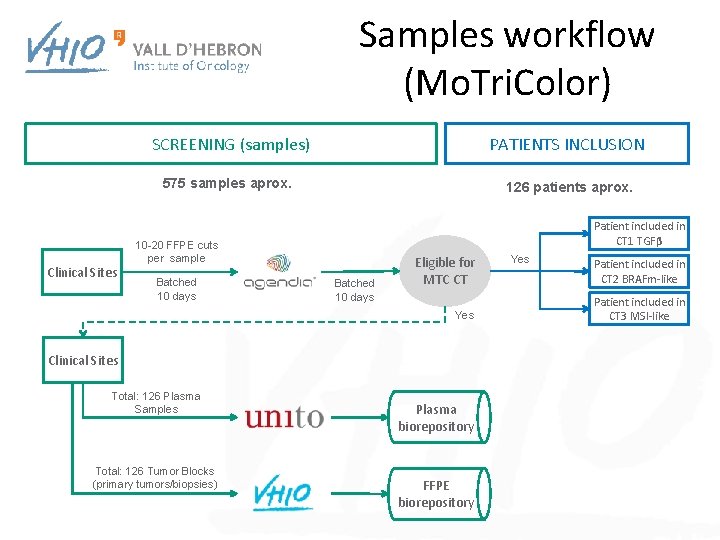
Samples workflow (Mo. Tri. Color) Clinical Sites SCREENING (samples) PATIENTS INCLUSION 575 samples aprox. 126 patients aprox. Patient included in CT 1 TGFβ 10 -20 FFPE cuts per sample Batched 10 days Eligible for MTC CT Yes Clinical Sites Total: 126 Plasma Samples Total: 126 Tumor Blocks (primary tumors/biopsies) Plasma biorepository FFPE biorepository Yes Patient included in CT 2 BRAFm-like Patient included in CT 3 MSI-like
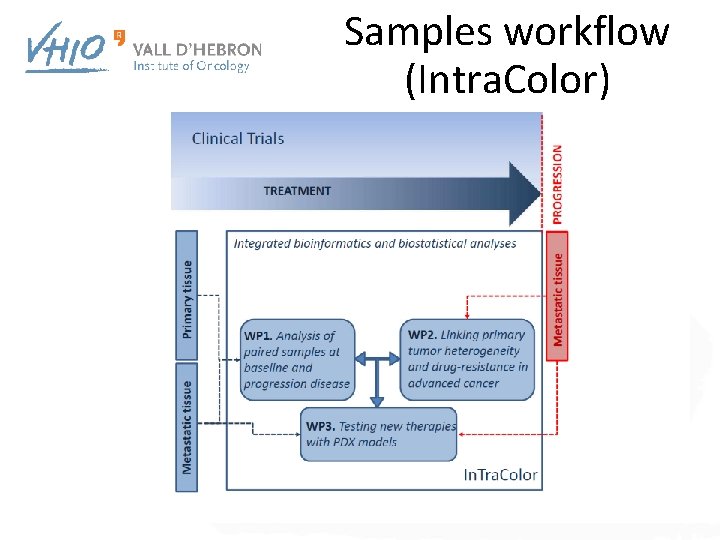
Samples workflow (Intra. Color)
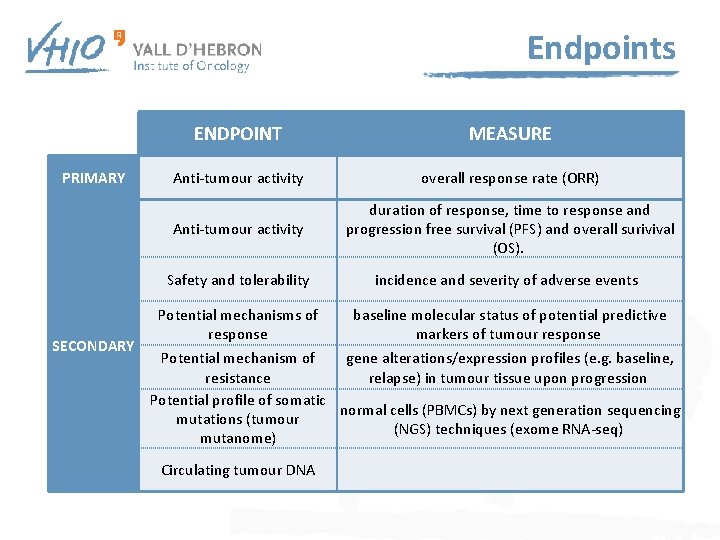
Endpoints ENDPOINT MEASURE PRIMARY Anti-tumour activity overall response rate (ORR) Anti-tumour activity duration of response, time to response and progression free survival (PFS) and overall surivival (OS). Safety and tolerability incidence and severity of adverse events Potential mechanisms of response baseline molecular status of potential predictive markers of tumour response SECONDARY Potential mechanism of gene alterations/expression profiles (e. g. baseline, resistance relapse) in tumour tissue upon progression Potential profile of somatic normal cells (PBMCs) by next generation sequencing mutations (tumour (NGS) techniques (exome RNA-seq) mutanome) Circulating tumour DNA

WP Tasks and deliverables • • Task 4. 1 Obtaining ethics (IRB) approval of clinical protocol Task 4. 2 Development and implement e. CRF (electronic case record form) Task 4. 3 Patient recruitment phase II trial and data evaluation Task 4. 4 Reporting follow-up and publication # TYP DISS DATE Clinical Trial 3 protocol R Clinical Trial 3 EUDRA CT R Operational e. CRF for data collection of the phase II trial CT 3 R IRB approved clinical protocol encompassing the phase II trial CT 3 R Assessment of the activity of MPDL 3280 A in patients with advanced CRC D 4. 3 R with MSI and MSI-like genotypic features Submission of scientific paper for publication and thereby bringing trial DE D 4. 4 results into the public domain CT 3 C CO PU CO CO 3 3 4 5 CO 32 PU 48 D 4. 5 D 4. 6 D 4. 2 D 4. 1 DELIVERABLE NAME

NATIONAL CALLS PROJECTS ONCOLOGY FONDO DE INVESTIGACIÓN SANITARIA - ISCIII AECC (300 k – 1, 2 M – seed) Plan Nacional BBVA/ LA CAIXA Grants ROCHE/MERCK SERONO CIBER - Ciber. Onc Foundations: FERO, Sandra Ibarra , FOT, GEIS, SOLTI, GEICO © Vall d'Hebron Institute of Oncology (VHIO)

INTERNATIONAL CALLS PROJECTS ONCOLOGY H 2020 – PHC – Big Collaborative Eranets – TRANSCAN – Medium Collaborative ASCO (YIA-CDA)/ ESMO/ AACR(SU 2 C) SUSAN KOMEN/ BCRF/ LCRF IMI - Industry CRUK – Grand Challenge / Accelerator AICR Grant for Oncology Innovation – GOI/ Merck NCI/NIH – US Calls ERC – European Research Council © Vall d'Hebron Institute of Oncology (VHIO)

Thank you Mo. Tri. Color Kick-off meeting 29
- Slides: 29