Einstein A coefficients for vibrationalrotational transitions of NO

Einstein A coefficients for vibrational-rotational transitions of NO Mauricio Gutiérrez 1 and John Ogilvie 2 1 Georgia Institute of Technology, Atlanta GA 2 Universidad de Costa Rica, San José, Costa Rica 68 th International Symposium on Molecular Spectroscopy June 20 th, 2013

Why Einstein A coefficients of NO? • Einstein A coefficient: proportionality factor between the intensity of spectral lines and the relative populations. • There is considerable uncertainty in vibrational distributions of NO products in several reactions. • Our method has been applied to other molecules, but not to NO. P. Houston et al, J. Phys. Chem. A 114, 11292 (2010)

Einstein A coefficients J. F. Ogilvie, The vibrational and rotational spectrometry of diatomic molecules (Academic Press, 1998) G. Herzberg, Spectra of diatomic molecules (D. Van Nostrand Company, 1950)

Einstein A coefficients J. F. Ogilvie, The vibrational and rotational spectrometry of diatomic molecules (Academic Press, 1998) G. Herzberg, Spectra of diatomic molecules (D. Van Nostrand Company, 1950)

Einstein A coefficients Radial dipole moment D. M. Dennison, Phys. Rev. 28, 318 (1926) G. Herzberg, Spectra of diatomic molecules (D. Van Nostrand Company, 1950)

Einstein A coefficients Radial dipole moment Angular dipole moment D. M. Dennison, Phys. Rev. 28, 318 (1926) G. Herzberg, Spectra of diatomic molecules (D. Van Nostrand Company, 1950)

Einstein A coefficients Radial dipole moment Angular dipole moment D. M. Dennison, Phys. Rev. 28, 318 (1926) G. Herzberg, Spectra of diatomic molecules (D. Van Nostrand Company, 1950)

TDM II: radial contribution Herman-Wallis approach: • Apply perturbation theory. • Re-express the matrix elements in terms of a purely vibrational part and a vibrational-rotational interaction. R. Herman and R. F. Wallis, J. Chem. Phys. 23, 4 (1955) R. H. Tipping and J. F. Ogilvie, J. Mol. Spec. 96, 442 (1982)

TDM II: radial contribution Herman-Wallis approach: • Apply perturbation theory. • Re-express the matrix elements in terms of a purely vibrational part and a vibrational-rotational interaction. R. Herman and R. F. Wallis, J. Chem. Phys. 23, 4 (1955) R. H. Tipping and J. F. Ogilvie, J. Mol. Spec. 96, 442 (1982)
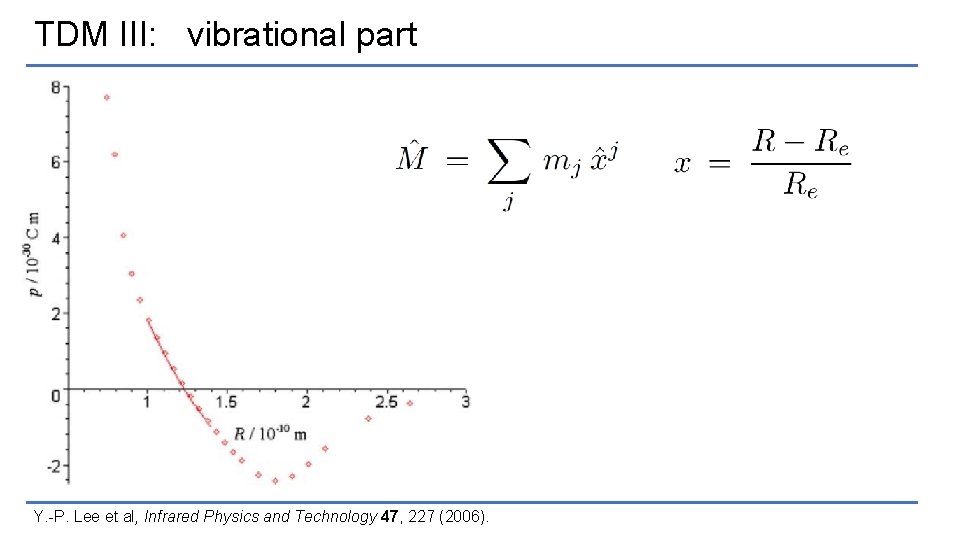
TDM III: vibrational part Y. -P. Lee et al, Infrared Physics and Technology 47, 227 (2006).

TDM III: vibrational part Y. -P. Lee et al, Infrared Physics and Technology 47, 227 (2006).

TDM IV: vibrational matrix elements Dunham’s potential Dunham’s method: • Use the harmonic oscillator eigenfunctions as a basis and apply perturbation theory with Dunham’s potential. • Obtain symbolic expressions for the matrix elements. J. F. Ogilvie, The vibrational and rotational spectrometry of diatomic molecules (Academic Press, 1998)
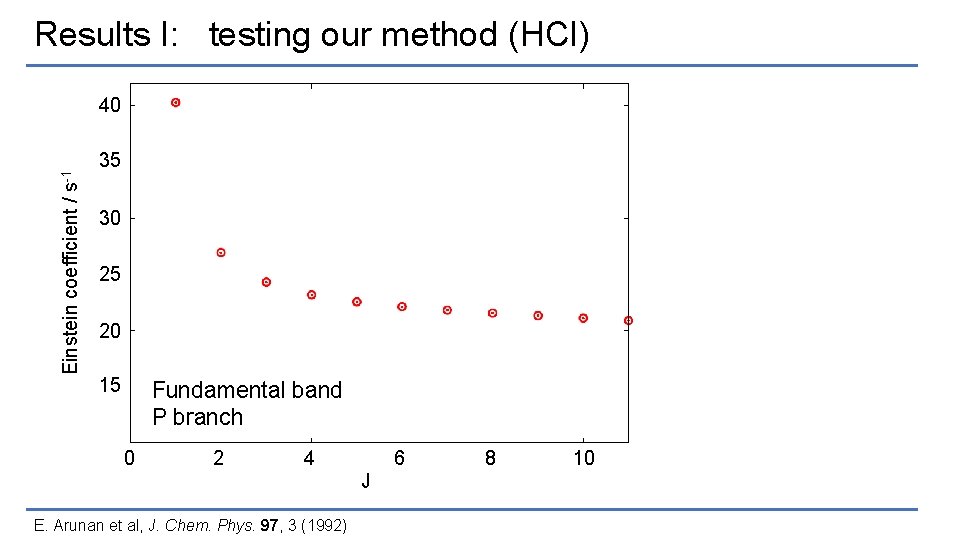
Results I: testing our method (HCl) Einstein coefficient / s-1 40 35 30 25 20 15 Fundamental band P branch 0 2 4 6 J E. Arunan et al, J. Chem. Phys. 97, 3 (1992) 8 10
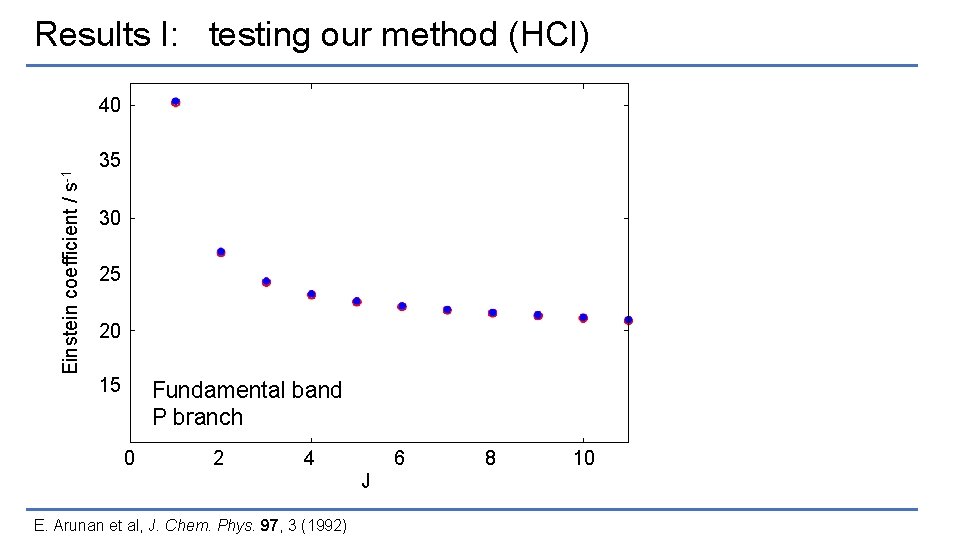
Results I: testing our method (HCl) Einstein coefficient / s-1 40 35 30 25 20 15 Fundamental band P branch 0 2 4 6 J E. Arunan et al, J. Chem. Phys. 97, 3 (1992) 8 10
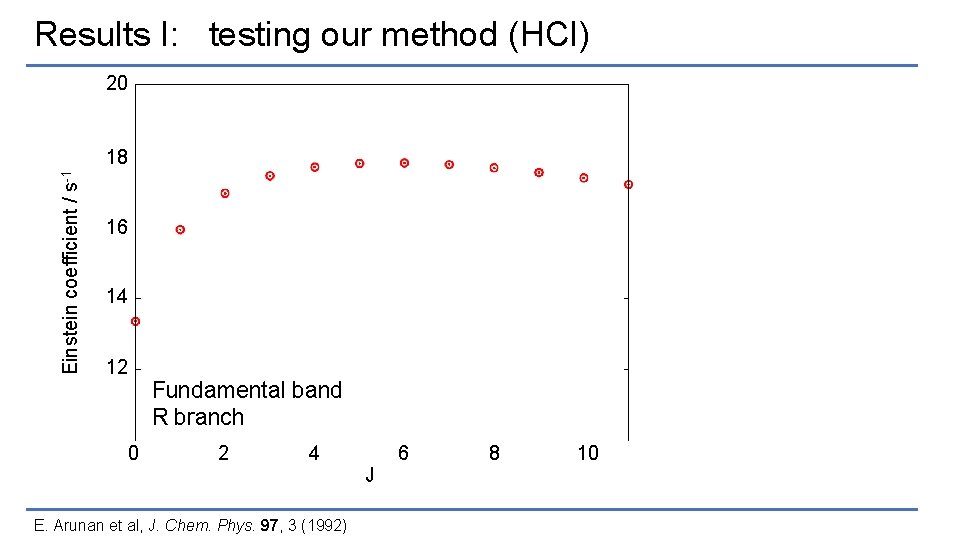
Results I: testing our method (HCl) 20 Einstein coefficient / s-1 18 16 14 12 Fundamental band R branch 0 2 4 6 J E. Arunan et al, J. Chem. Phys. 97, 3 (1992) 8 10
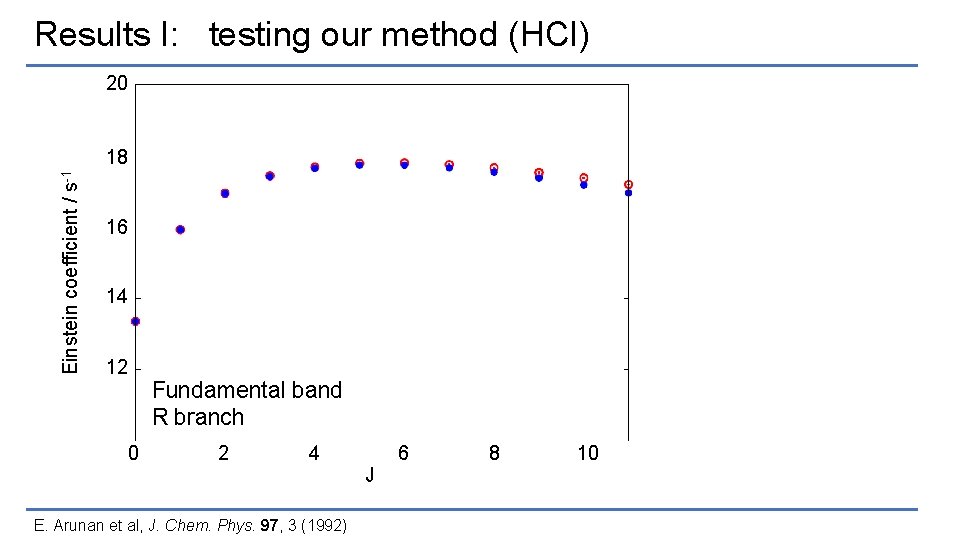
Results I: testing our method (HCl) 20 Einstein coefficient / s-1 18 16 14 12 Fundamental band R branch 0 2 4 6 J E. Arunan et al, J. Chem. Phys. 97, 3 (1992) 8 10
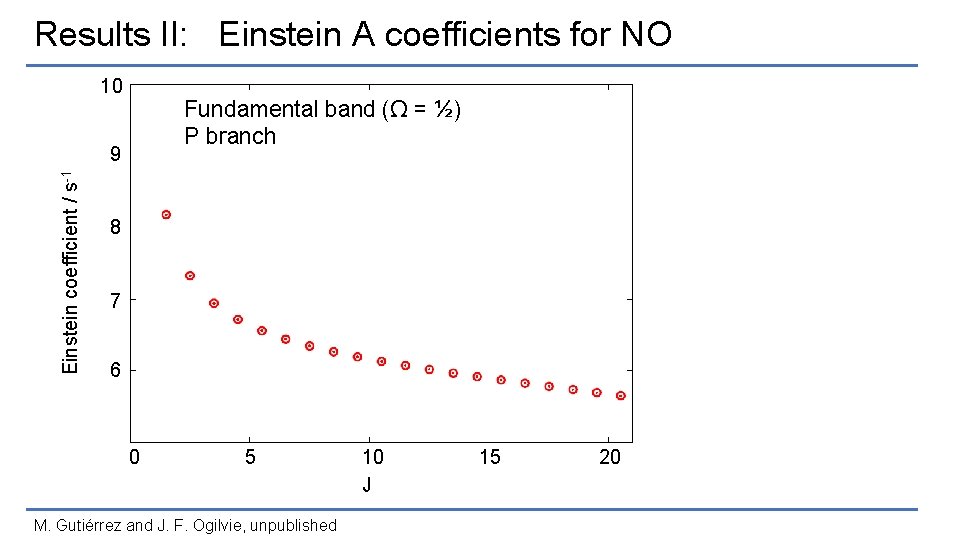
Results II: Einstein A coefficients for NO 10 Fundamental band (Ω = ½) P branch Einstein coefficient / s-1 9 8 7 6 0 5 M. Gutiérrez and J. F. Ogilvie, unpublished 10 J 15 20
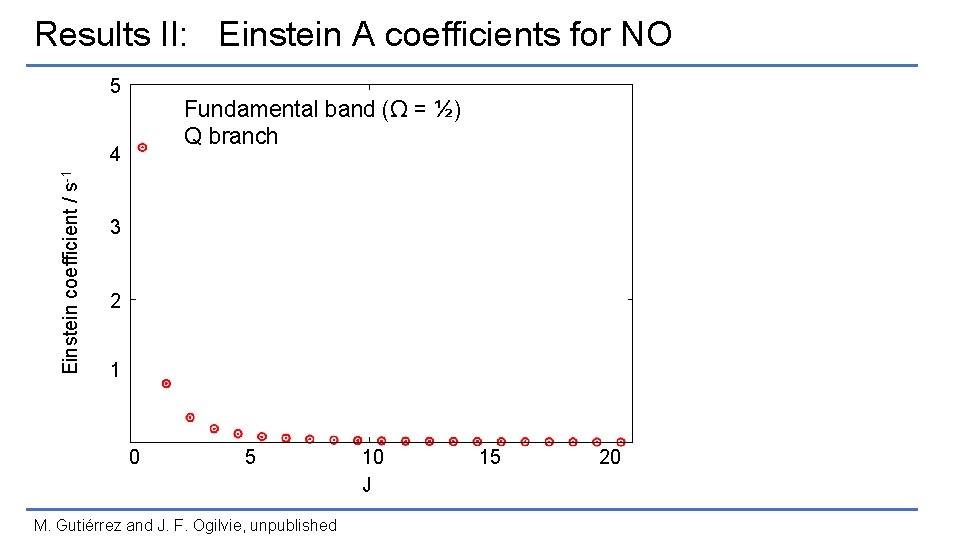
Results II: Einstein A coefficients for NO 5 Fundamental band (Ω = ½) Q branch Einstein coefficient / s-1 4 3 2 1 0 5 M. Gutiérrez and J. F. Ogilvie, unpublished 10 J 15 20
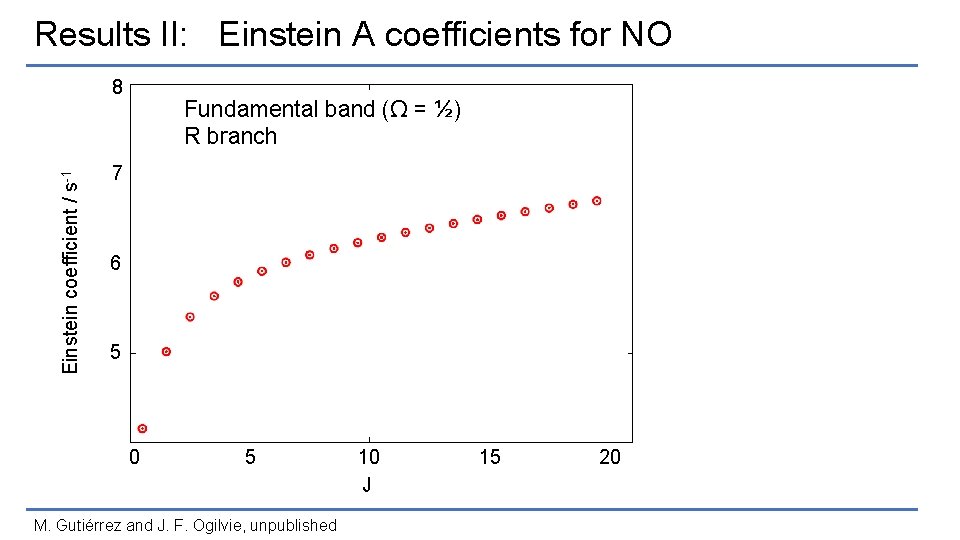
Results II: Einstein A coefficients for NO Einstein coefficient / s-1 8 Fundamental band (Ω = ½) R branch 7 6 5 0 5 M. Gutiérrez and J. F. Ogilvie, unpublished 10 J 15 20
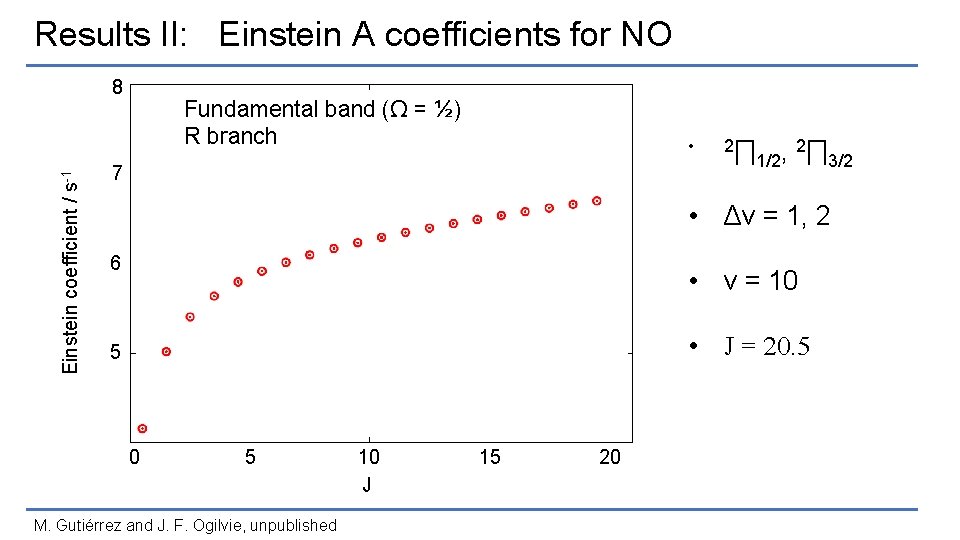
Results II: Einstein A coefficients for NO Einstein coefficient / s-1 8 Fundamental band (Ω = ½) R branch • 7 2∏ 1/2, 2∏ 3/2 • Δv = 1, 2 6 • v = 10 • J = 20. 5 5 0 5 M. Gutiérrez and J. F. Ogilvie, unpublished 10 J 15 20

Conclusions • We have calculated the spontaneous emission coefficients for vibration-rotational transitions with Δv = 1, 2 up to v = 10 for NO in its electronic ground state. • Using the same method, we calculated coefficients for HCl and they agree with previous results. • Future work: comparison with results from ab initio methods.

Acknowledgements • John Ogilvie • Ken Brown’s group
- Slides: 22