Ehab Mostafa Hassouna MD A Prof of Internal


Ehab Mostafa Hassouna, MD A. Prof. of Internal Medicine, Hepatobiliary Unit, Faculty of Medicine, Alexandria University, Egypt EASL, AASLD member
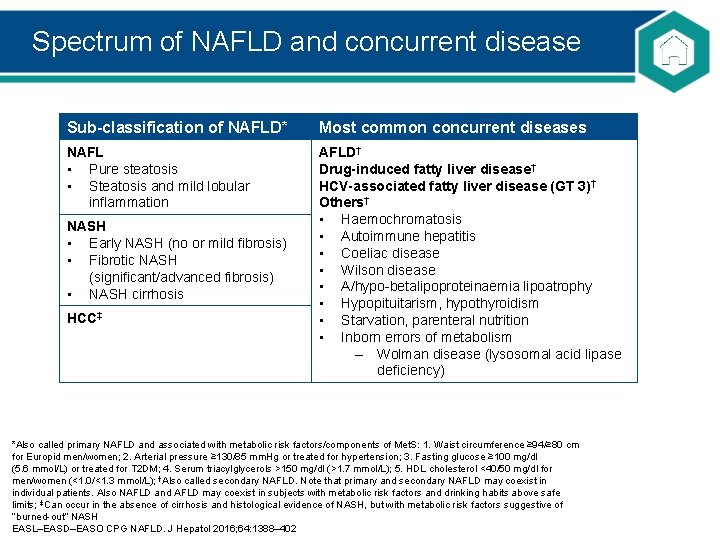
Spectrum of NAFLD and concurrent disease Sub-classification of NAFLD* Most common concurrent diseases NAFL • Pure steatosis • Steatosis and mild lobular inflammation AFLD† Drug-induced fatty liver disease† HCV-associated fatty liver disease (GT 3)† Others† • Haemochromatosis • Autoimmune hepatitis • Coeliac disease • Wilson disease • A/hypo-betalipoproteinaemia lipoatrophy • Hypopituitarism, hypothyroidism • Starvation, parenteral nutrition • Inborn errors of metabolism – Wolman disease (lysosomal acid lipase deficiency) NASH • Early NASH (no or mild fibrosis) • Fibrotic NASH (significant/advanced fibrosis) • NASH cirrhosis HCC‡ *Also called primary NAFLD and associated with metabolic risk factors/components of Met. S: 1. Waist circumference ≥ 94/≥ 80 cm for Europid men/women; 2. Arterial pressure ≥ 130/85 mm. Hg or treated for hypertension; 3. Fasting glucose ≥ 100 mg/dl (5. 6 mmol/L) or treated for T 2 DM; 4. Serum triacylglycerols >150 mg/dl (>1. 7 mmol/L); 5. HDL cholesterol <40/50 mg/dl for men/women (<1. 0/<1. 3 mmol/L); †Also called secondary NAFLD. Note that primary and secondary NAFLD may coexist in individual patients. Also NAFLD and AFLD may coexist in subjects with metabolic risk factors and drinking habits above safe limits; ‡Can occur in the absence of cirrhosis and histological evidence of NASH, but with metabolic risk factors suggestive of ‘‘burned-out” NASH EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Role of non-invasive assessments • Non-invasive markers should aim to: – Identify the risk of NAFLD among individuals with increased metabolic risk in primary care – Identify those with a worse prognosis in secondary and tertiary care • E. g. severe NASH – Monitor disease progression – Predict response to therapeutic interventions Achieving these aims could reduce the need for liver biopsy EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402
Non-Invasive Approaches
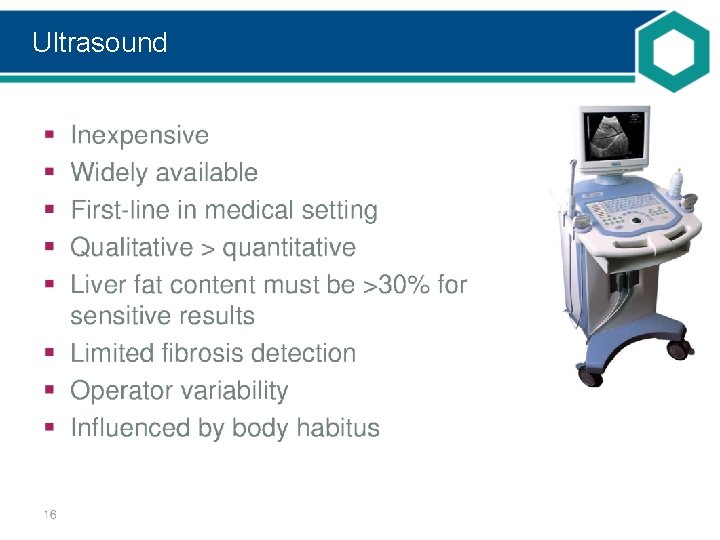
Ultrasound
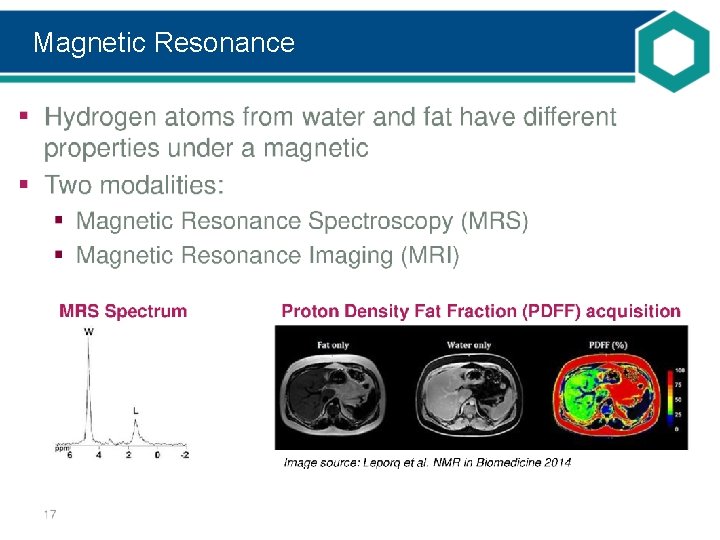
Magnetic Resonance
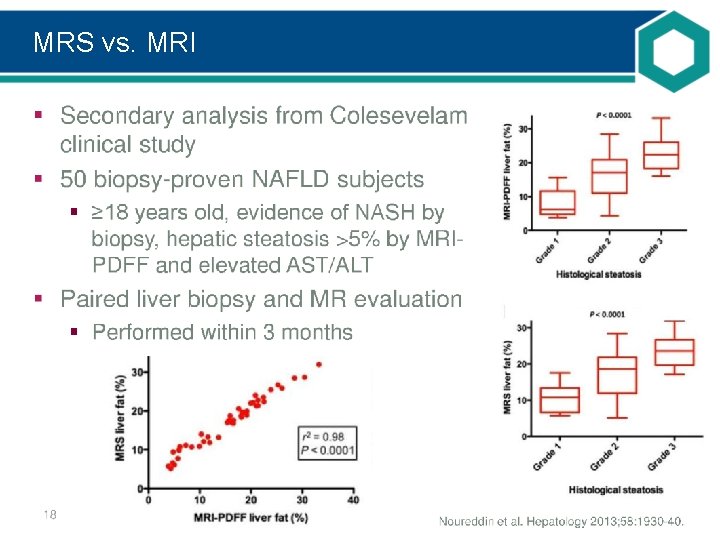
MRS vs. MRI
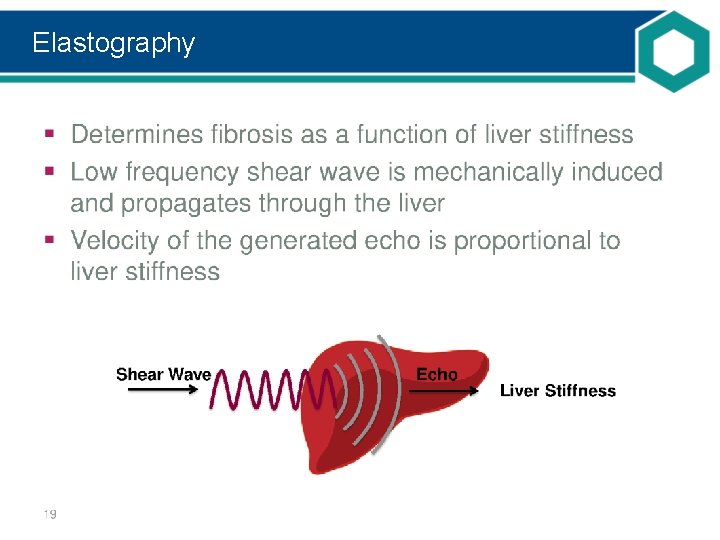
Elastography

Elastography Modalities
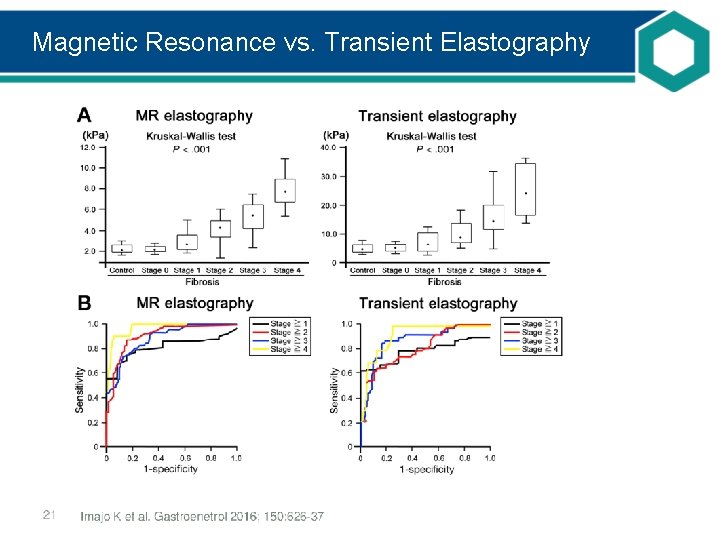
Magnetic Resonance vs. Transient Elastography
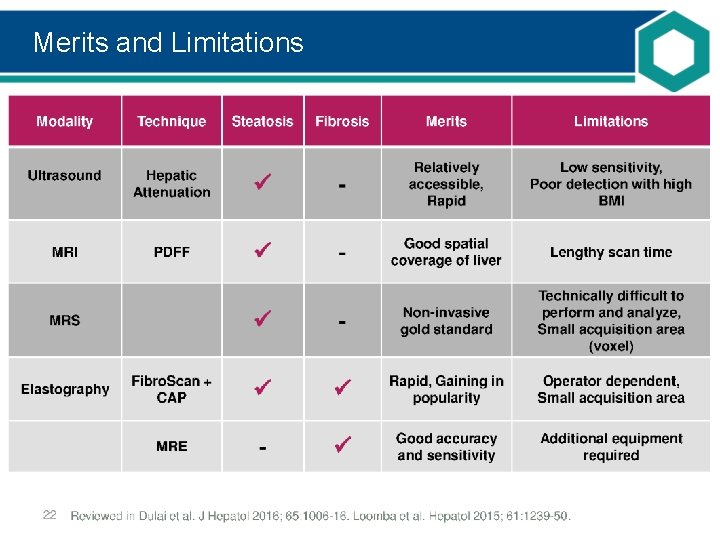
Merits and Limitations
Soluble Biomarkers
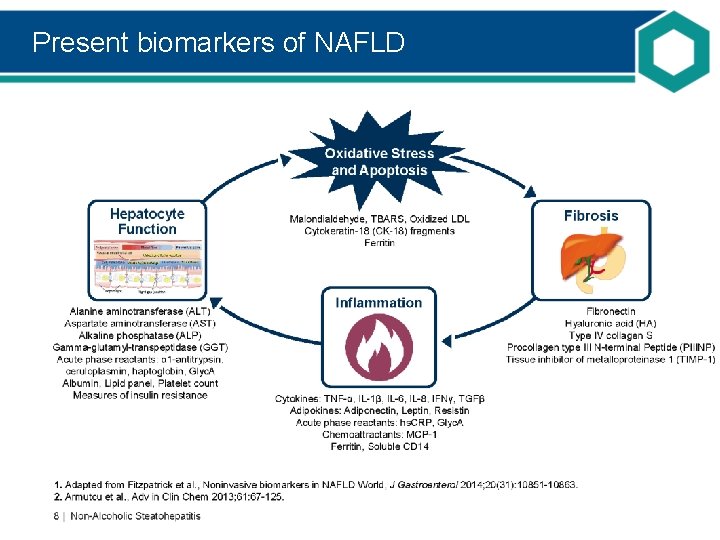
Present biomarkers of NAFLD
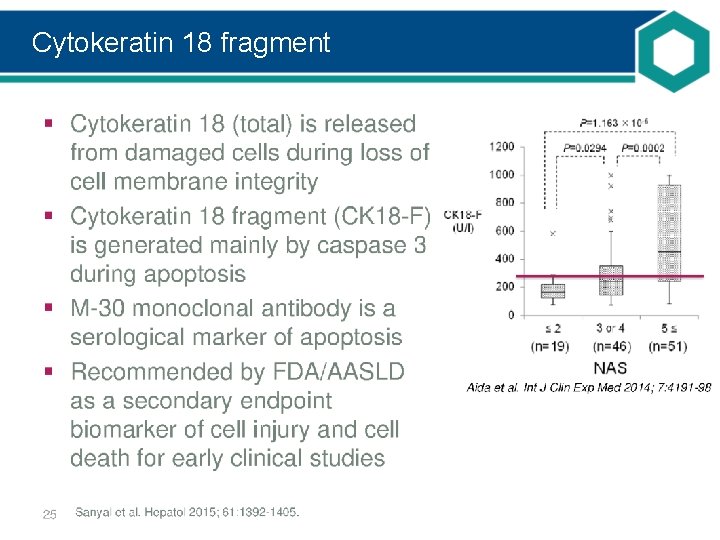
Cytokeratin 18 fragment
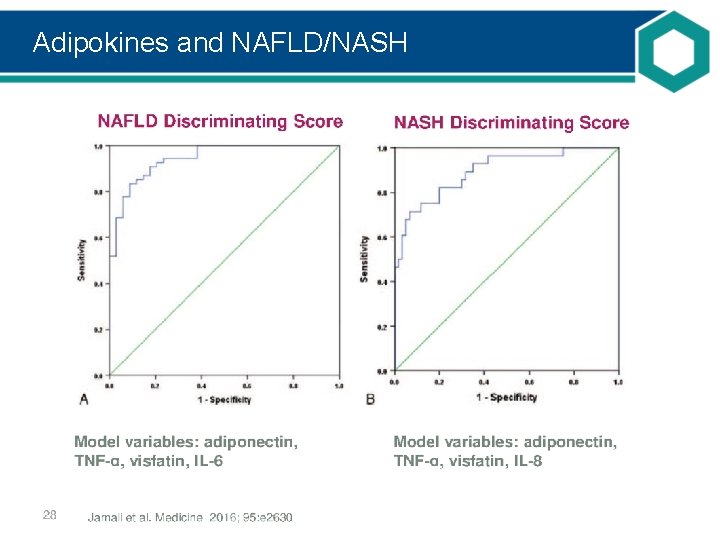
Adipokines and NAFLD/NASH

NASH Fibro. Sure Test
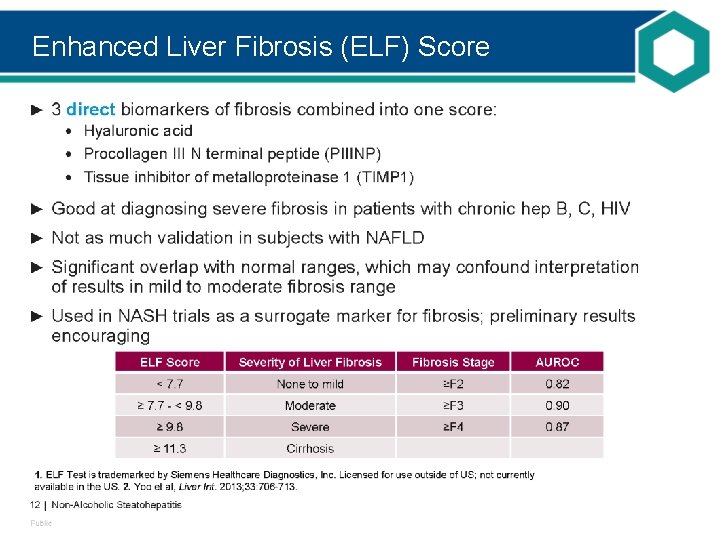
Enhanced Liver Fibrosis (ELF) Score
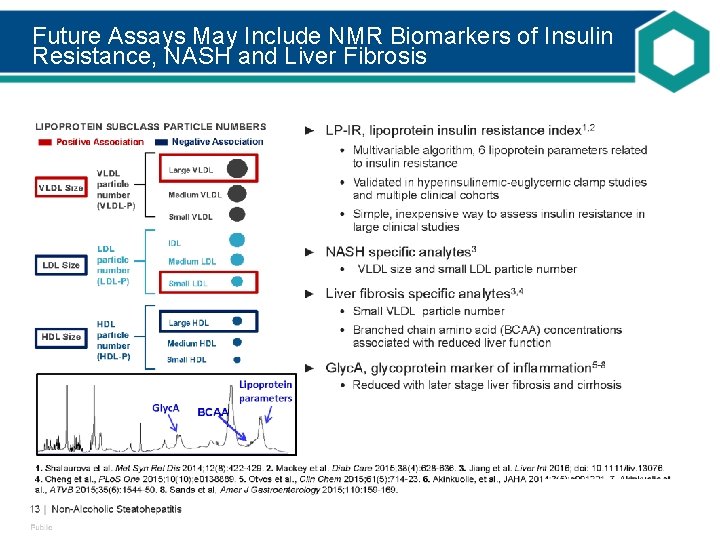
Future Assays May Include NMR Biomarkers of Insulin Resistance, NASH and Liver Fibrosis
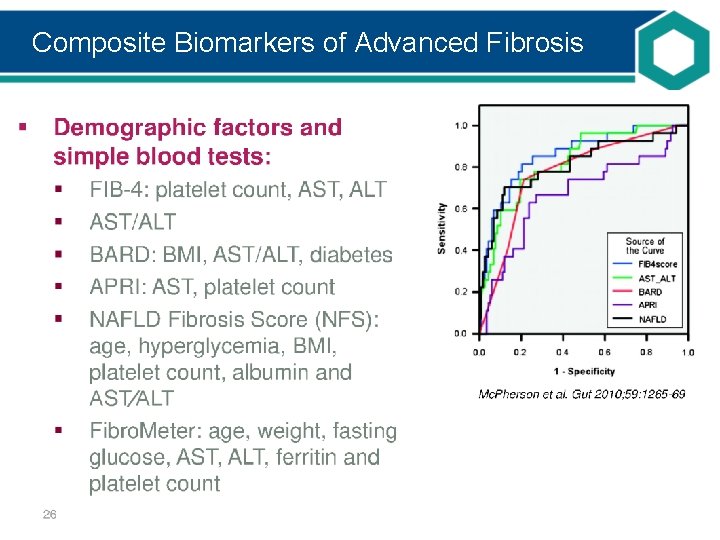
Composite Biomarkers of Advanced Fibrosis
NASH in a Genomic Context
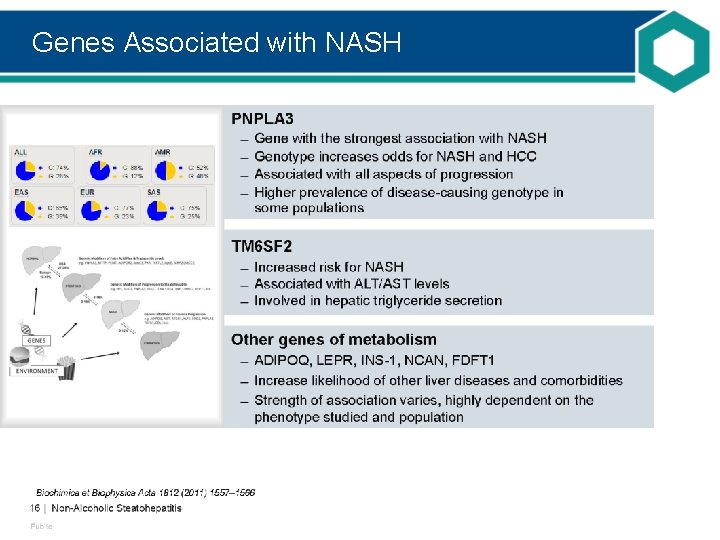
Genes Associated with NASH

miro. RNA Profiling

Non-invasive assessment of steatosis • Steatosis should be documented whenever NAFLD is suspected – Predicts future T 2 DM, cardiovascular events and arterial hypertension – Quantification of fat content is of limited clinical relevance • Except as a surrogate of treatment effectiveness Recommendations Grade of evidence Grade of recommendation US is the preferred first-line diagnostic procedure for imaging of NAFLD, as it provides additional diagnostic information A 1 Whenever imaging tools are not available or feasible serum biomarkers and scores are an acceptable alternative for the diagnosis of steatosis B 2 A quantitative estimation of liver fat can only be obtained by 1 H-MRS. This technique is of value in clinical trials and experimental studies, but is expensive and not recommended in the clinical setting A 1 EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402

Non-invasive assessment of fibrosis • Fibrosis is the most important prognostic factor in NAFLD – Correlates with liver-related outcomes and mortality – Advanced fibrosis indicates thorough investigation Recommendations Grade of evidence Grade of recommendation Biomarkers, fibrosis scores, and transient elastography, are acceptable non-invasive procedures to identify those at low risk of advanced fibrosis/cirrhosis A 2 Biomarkers/scores PLUS transient elastography might confer additional diagnostic accuracy and reduce need for liver biopsy B 2 Monitoring of fibrosis progression may rely on biomarkers/scores and transient elastography, although this strategy requires validation C 2 The identification of advanced fibrosis or cirrhosis by serum biomarkers/scores and/or elastography is less accurate and needs to be confirmed by liver biopsy, according to the clinical context B 2 In selected patients at high risk of liver disease progression, monitoring should include a repeat biopsy after 5 -year follow-up C 2 EASL–EASD–EASO CPG NAFLD. J Hepatol 2016; 64: 1388– 402
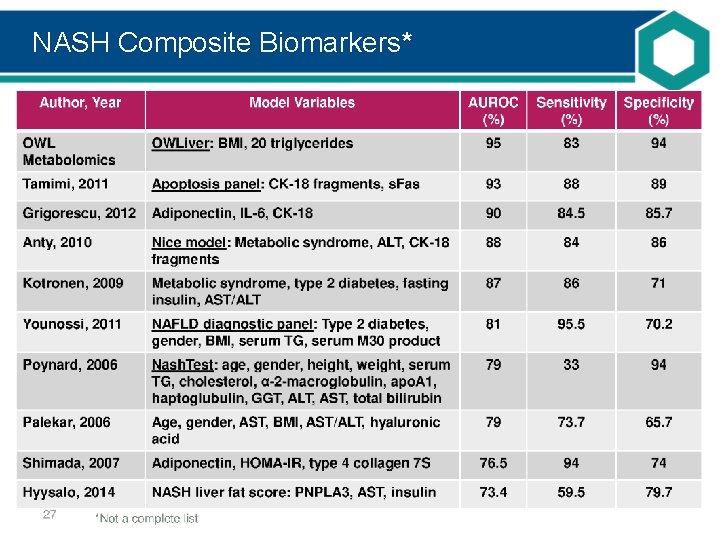
NASH Composite Biomarkers*
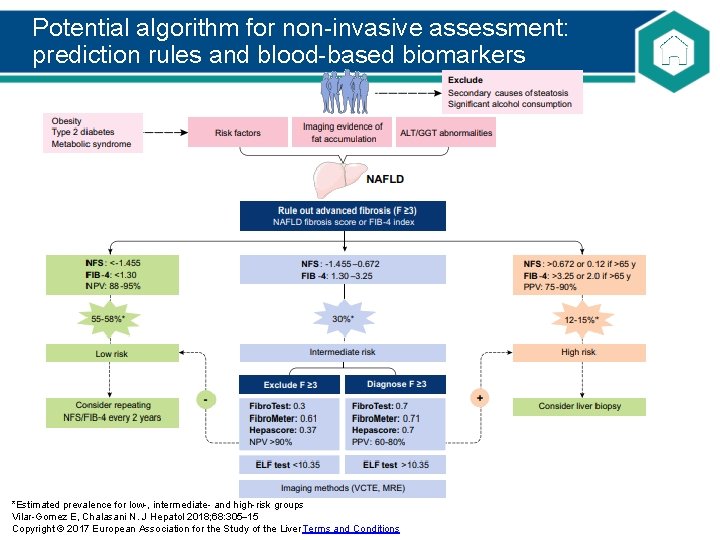
Potential algorithm for non-invasive assessment: prediction rules and blood-based biomarkers *Estimated prevalence for low-, intermediate- and high-risk groups Vilar-Gomez E, Chalasani N. J Hepatol 2018; 68: 305 15 Copyright © 2017 European Association for the Study of the Liver Terms and Conditions

Conclusion § In summary, no highly sensitive and specific tests are available to differentiate NASH from simple steatosis. However, diagnostic accuracy can be improved by combining blood biomarkers. Combination and complex models including clinical, routine blood based variables and markers that reflect the dynamic nature of the fibrogenic process appear to have higher diagnostic accuracy and predictive value in identifying advanced fibrosis , in patients with NAFLD.

NFS and FIB-4 are useful screening tools to be routinely applied in clinical practice, since they can accurately exclude patients with advanced fibrosis, are inexpensive and easy to obtain.

References 1. Chalasani N, Younossi Z, Lavine JE, Charlton M, Cusi K, Rinella M, et al. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology 2017, [In press]. 2. Younossi Z, Henry L. Contribution of alcoholic and nonalcoholic fatty liver disease to the burden of liver-related morbidity and mortality. Gastroenterology 2016; 150: 1778– 1785. 3. Younossi ZM, Blissett D, Blissett R, Henry L, Stepanova M, Younossi Y, et al. The economic and clinical burden of nonalcoholic fatty liver disease in the United States and Europe. Hepatology. 2016; 64: 1577– 1586. 4. Ulitsky A, Ananthakrishnan AN, Komorowski R, Wallace J, Surapaneni N, Franco J, et al. A Noninvasive Clinical Scoring Model Predicts Risk of Nonalcoholic Steatohepatitis in Morbidly Obese Patients. Obes Surg 2010; 20: 685– 691. 5. Kwok R, Tse YK, Wong GL, Ha Y, Lee AU, Ngu MC, et al. Systematic review with meta-analysis: non-invasive assessment of non-alcoholic fatty liver disease–the role of transient elastography and plasma cytokeratin-18 fragments. Aliment Pharmacol Ther 2014; 39: 254– 269. 6. Feldstein AE, Wieckowska A, Lopez AR, Liu YC, Zein NN, Mc. Cullough AJ. Cytokeratin-18 fragment levels as noninvasive biomarkers for nonalcoholic steatohepatitis: a multicenter validation study. Hepatology 2009; 50: 1072– 1078. 7. Leroy V, Sturm N, Faure P, Trocme C, Marlu A, Hilleret MN, et al. Prospective evaluation of Fibro. Test(R), Fibro. Meter(R), and Hepa. Score(R) for staging liver fibrosis in chronic hepatitis B: comparison with hepatitis C. J Hepatol 2014; 61: 28– 34. 8. Adams LA, George J, Bugianesi E, Rossi E, De Boer WB, van der Poorten D, et al. Complex non-invasive fibrosis models are more accurate than simple models in non-alcoholic fatty liver disease. J Gastroen Hepatol 2011; 26: 1536– 1543. 9. Hardy T, Zeybel M, Day CP, Dipper C, Masson S, Mc. Pherson S, et al. Plasma DNA methylation: a potential biomarker for stratification of liver fibrosis in non-alcoholic fatty liver disease. Gut 2017; 66: 1321– 1328. 10. Pirola CJ, Fernandez Gianotti T, Castano GO, Mallardi P, San Martino J, Ledesma Mora Gonzalez Lopez, et al. Circulating micro. RNA signature in non-alcoholic fatty liver disease: from serum non-coding RNAs to liver histology and disease pathogenesis. Gut 2015; 64: 800– 812. 11. Sookoian S, Pirola CJ. Cell-free DNA methylation as liquid biopsy for the assessment of fibrosis in patients with nonalcoholic steatohepatitis: a gap between innovation and implementation. Hepatol Surg Nutr 2017; 6: 117– 121. 12. Ratziu V, Massard J, Charlotte F, Messous D, Imbert-Bismut F, Bonyhay L, et al. Diagnostic value of biochemical markers (Fibro. Test-Fibro. SURE) for the prediction of liver fibrosis in patients with non-alcoholic fatty liver disease. BMC Gastroenterol 2006; 6. 13. Loaeza-del-Castillo A, Paz-Pineda F, Oviedo-Cardenas E, Sanchez-Avila F, Vargas-Vorackova F. AST to platelet ratio index (APRI) for the noninvasive evaluation of liver fibrosis. Ann Hepatol 2008; 7: 350– 357.

THANK YOU
- Slides: 31