EGF 2042 ORGANIC CHEMISTRY UNIT 2 HYDROCARBONS PART

EGF 2042 ORGANIC CHEMISTRY UNIT 2: HYDROCARBONS (PART 1)

OUTLINE (UNIT 2) 2. 1 Introduction in process industry 2. 2 Natural gas and petroleum resources 2. 3 Alkanes 2. 4 Alkenes, addition polymerization and the chemistry of rubber 2. 5 Alkynes 2. 6 Chlorinated alkanes and geometrical isomers 2. 7 Aromatic hydrocarbon

TOPIC LEARNING OUTCOMES At the end of the lesson, students should be able to: 1. Understand the process related organic chemistry occurred in industry. 2. Explain the sources of hydrocarbons from natural gas and petroleum. 3. Interpret the chemical names, general formulae and displayed formulae of alkanes, alkenes, alkynes and aromatics. 4. Describe the reactions and synthesis of alkanes, alkenes, alkynes, and aromatic and polymerization process. 5. Suggest the repeat unit of the polymer from a given monomer. 6. Understand the geometrical isomers and chlorinated alkanes.

2. 1 INTRODUCTION TO PROCESS INDUSTRY

WHAT IS PETROLEUM? Petroleum is a fossilized mass that has accumulated below the earth’s surface from time immemorial. Raw petroleum is known as crude (petroleum) oil or mineral oil. It is a mixture of various organic substances and is the source of hydrocarbons, such as methane, propane, butane, pentane, and various other paraffinic, naphthenic, and aromatic hydrocarbons, the building blocks of today’s organic industry.

PRODUCTS FROM PETROLEUM • Various petroleum products, such as gaseous and liquid fuels, lubricating oil, solvents, asphalts, waxes, and coke, are derived from refining crude oil. • The hydrocarbon processing industry is basically divided into three distinct activities—petroleum production, petroleum refining, and petrochemical manufacture.

PRODUCTS FROM PETROLEUM Refineries produce cooking gas (liquefied petroleum gas or LPG), motor spirit (also known as petrol or gasoline), naphtha, kerosene, aviation turbine fuel (ATF), high speed diesel (HSD), lubricating base oils, wax, coke, bitumen (or asphalt), etc. , which are mostly a mixture of various hydrocarbons.


PETROCHEMICALS • In a petrochemical plant (where one or more petrochemicals are produced) or in a petrochemical complex (where many petrochemical products are produced), pure hydrocarbons or other organic chemicals with a definite number and type of constituent element or compound are produced from the products in refineries. Thus, petrochemicals are derived from petroleum products obtained from refineries.

PETROCHEMICALS • Products from a petrochemical complex are plastics, rubbers, synthetic fibers, raw materials for soap and detergents, alcohols, paints, pharmaceuticals, etc. Since petroleum is the mixture of hundreds of thousands of hydrocarbon compounds, there is a possibility of synthesizing many new compounds. In fact, due to the advancement of new technology, new petrochemicals are being invented and will continue to be added to this industry in the near future. Hence, the petrochemical industry is still a growing industry. The manufacture of valuable petrochemicals from low-valued petroleum products has been the main attractive option for the refining industry investing in the petrochemical industry. Thus, modern refineries are, in fact, refinery cum petrochemical complexes.

2. 2 NATURAL GAS AND PETROLEUM RESOURCES

NATURAL GAS AND PETROLEUM RESOURCES • Vast underground deposits of liquid hydrocarbons, mainly alkanes, are fund in many regions of the earth. These are often locked under the dome-shaped impervious rock. When drilled out of the mine, the oily mixture of hydrocarbons in its crude form is called Petroleum (in Latin, Petra=rock, oleum=oil) or Crude oil. Accompanying the reservoirs of petroleum is Natural Gas, consisting of gaseous hydrocarbons, largely methane.
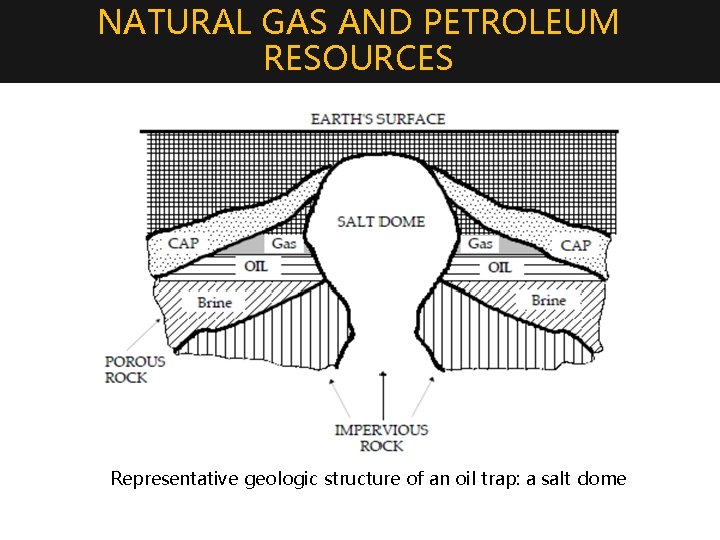
NATURAL GAS AND PETROLEUM RESOURCES Representative geologic structure of an oil trap: a salt dome

NATURAL GAS AND PETROLEUM RESOURCES • The origin of petroleum lies in plants and animals which lived on earth and in sea many millions of years ago. These organisms died and their remains became buried under the earth. Due to the bacterial decomposition and under the action of earth’s heat and pressure, these remains were converted to liquid hydrocarbons called as Petroleum. The gaseous hydrocarbons that were produced constituted the natural gas.

NATURAL GAS AND PETROLEUM RESOURCES • Today petroleum and natural gas rank as the main sources of our fuel and the bulk of organic chemicals used in industry. With the rapid development of automobile and aircraft industry, and the petrochemicals that have shot up into prominence in 1960 s, the rate of consumption of petroleum has become fast indeed. According to the present estimates, 80% of the world’s petroleum will be exhausted by the year 2025. • The gas found above the petroleum deposits in mines or by itself, is referred to as Natural Gas. It is a mixture of low molecular weight alkanes. It is composed primarily of methane (80%), ethane (13%), propane (3%), butane (1%), C 5 to C 8 alkanes (0. 5%) and nitrogen (1. 3%).
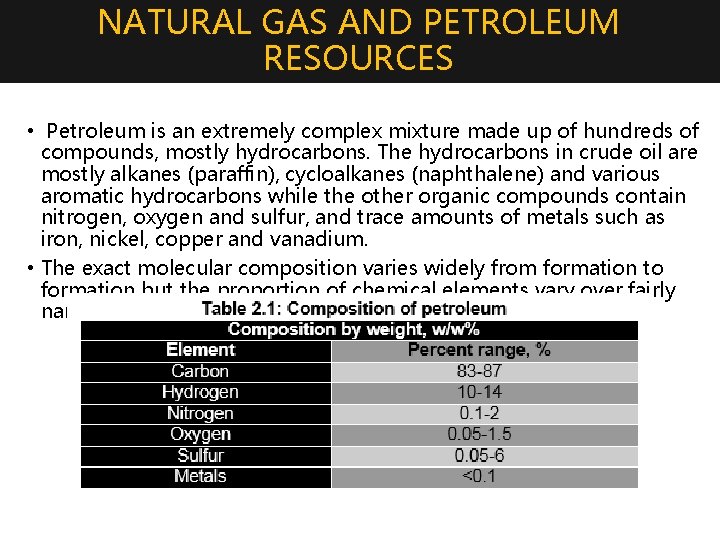
NATURAL GAS AND PETROLEUM RESOURCES • Petroleum is an extremely complex mixture made up of hundreds of compounds, mostly hydrocarbons. The hydrocarbons in crude oil are mostly alkanes (paraffin), cycloalkanes (naphthalene) and various aromatic hydrocarbons while the other organic compounds contain nitrogen, oxygen and sulfur, and trace amounts of metals such as iron, nickel, copper and vanadium. • The exact molecular composition varies widely from formation to formation but the proportion of chemical elements vary over fairly narrow limits as follows:

2. 3 ALKANES

WHAT ARE ALKANES? • Most simple classification of organic compounds • Saturated hydrocarbons • C and H connected by single bonds (C-C an d C-H) • Where can be found? – natural gas, gasoline, plastic • Chemical formula: Cn. H 2 n+2
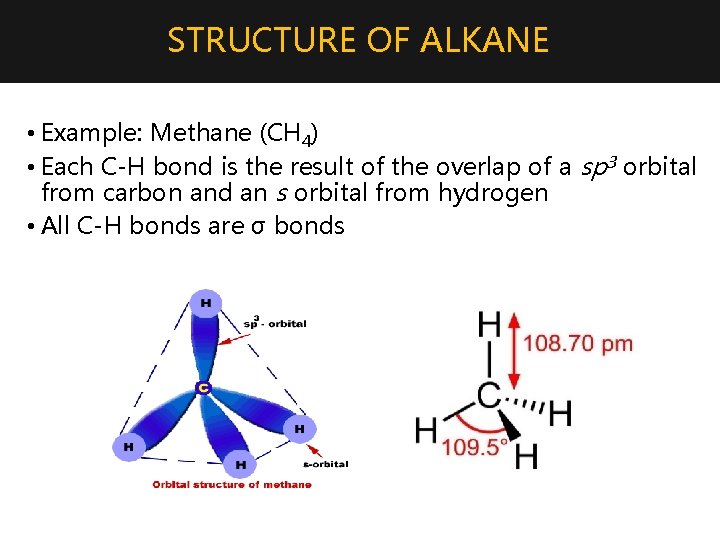
STRUCTURE OF ALKANE • Example: Methane (CH 4) • Each C-H bond is the result of the overlap of a sp 3 orbital from carbon and an s orbital from hydrogen • All C-H bonds are σ bonds
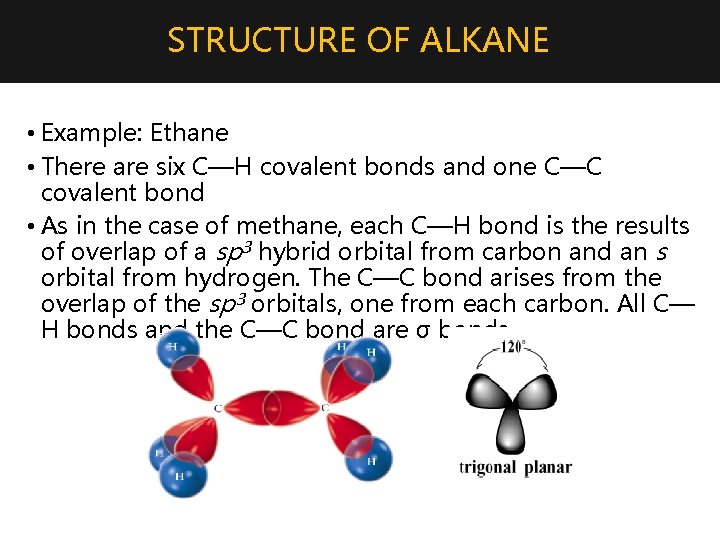
STRUCTURE OF ALKANE • Example: Ethane • There are six C—H covalent bonds and one C—C covalent bond • As in the case of methane, each C—H bond is the results of overlap of a sp 3 hybrid orbital from carbon and an s orbital from hydrogen. The C—C bond arises from the overlap of the sp 3 orbitals, one from each carbon. All C— H bonds and the C—C bond are σ bonds.

TYPES OF CARBON • Primary carbon – attached to only one other carbon • Secondary carbon – attached to two other carbons • Tertiary carbon – attached to three other carbons *Note: This definition implies that methane cannot have any such carbons, since it consists of only one carbon atom
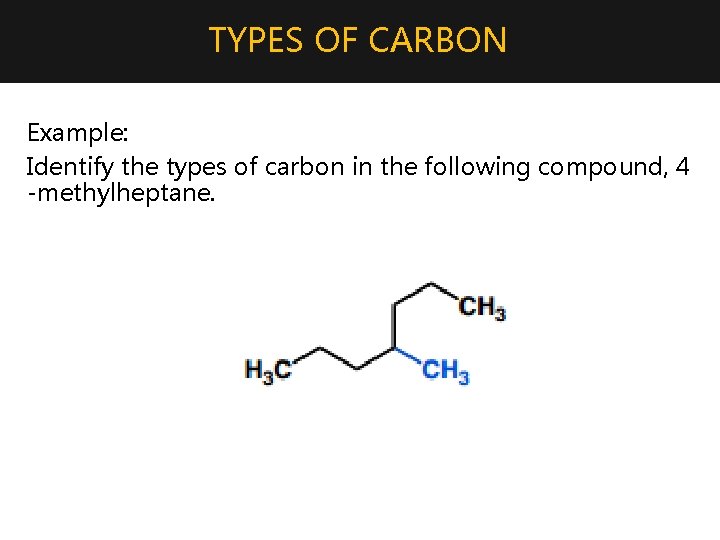
TYPES OF CARBON Example: Identify the types of carbon in the following compound, 4 -methylheptane.

BRANCHED ALKANES AND ALKYL GROUPS • When naming branched alkanes, • First, identify and name the longest continuous carbon chain • Then identify the branch, or branches. The branches are called alkyl groups. *Alky groups never exist by themselves. In alkanes they are always attached to a higher priority chain and are therefore sometime called substituents.

BRANCHED ALKANES AND ALKYL GROUPS • A lot more

HALOGEN SUBTITUENTS • Halogen substituents are easily accommodated, using the names: fluoro (F-), chloro (Cl-), bromo (Br-) and iodo (I-). • For example, CH 3 Br l l CH 3–CH–CH 2 1 -bromo-3 -methylbutane
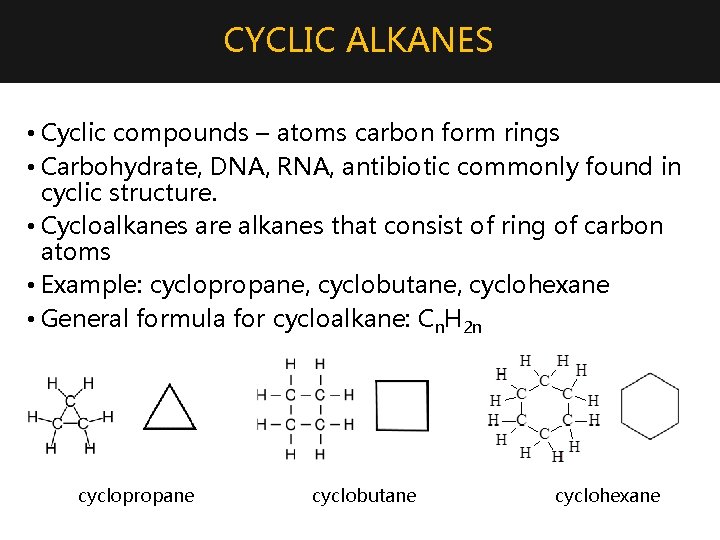
CYCLIC ALKANES • Cyclic compounds – atoms carbon form rings • Carbohydrate, DNA, RNA, antibiotic commonly found in cyclic structure. • Cycloalkanes are alkanes that consist of ring of carbon atoms • Example: cyclopropane, cyclobutane, cyclohexane • General formula for cycloalkane: Cn. H 2 n cyclopropane cyclobutane cyclohexane

NOMENCLATURE OF ORGANIC COMPOUNDS • Nomenclature is the scientific term for naming compounds. The governing body is “The International Union of Pure and Applied Chemistry”, or IUPAC for short. • Naming of organic structures, unlike biological classification, follows a rigid set of rules. • The IUPAC, came up with a set of rules that follows the same standards worldwide, and is accepted among all chemists. However, common names of compounds, or names that have historical roots, are still used today for many compounds.

THE IUPAC RULES • Prefix –where the substitutions, or other interesting things (anything but hydrogen is interesting to organic chemists!) is/are located. The number associated with the interesting “thing” or side group will always receive the lowest number possible in the alkane chain. • Parent / Root –how many carbons are in the longest chain in the molecule (the longest chain might twist and turn. Longest chain represents carbon atoms that are bonded to each other. • Suffix –family – type of functional group (alkane, alkene, alcohol, ester, etc. . . )

HOW TO NAME ALKANES? prefix – root – ane • Root words are named for its number of carbons: # of carbons root 1 meth- 2 eth- 3 prop- 4 but- 5 pent- 6 hex- 7 hept- 8 oct- 9 non- 10 dec-

HOMOLOGOUS SERIES OF ALKANES A Homologous Series is a group of organic chemical compounds, usually listed in order of increasing size, that have a similar structure (and hence also similar properties) and whose structures differ only by the number of CH 2 units in the main carbon chain. The simplest example of a homologous series in organic chemistry is that of alkanes. Alkanes consist of carbon and hydrogen atoms only, in proportions according to the general formula: Cn. H 2 n+2 Where the letter n represents the number of carbon atoms in each molecule of the compound.

IUPAC RULES FOR ALKANE NOMENCLATURE Find and name the longest continuous carbon chain. Identify and name groups attached to this chain. Number the chain consecutively, starting at the end nearest a substituent group. Designate the location of each substituent group by an appropriate number and name. Assemble the name, listing groups in alphabetical order using the full name (e. g. cyclopropyl before isobutyl). The prefixes di, tri, tetra etc. , used to designate several groups of the same kind, are not considered when alphabetizing.
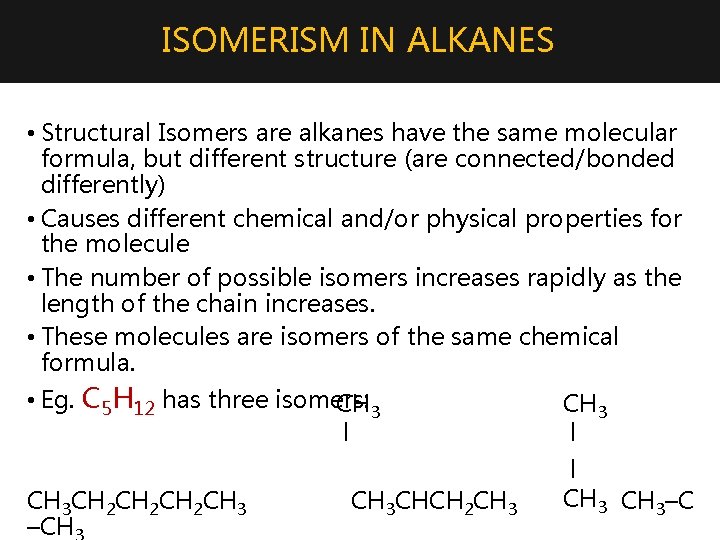
ISOMERISM IN ALKANES • Structural Isomers are alkanes have the same molecular formula, but different structure (are connected/bonded differently) • Causes different chemical and/or physical properties for the molecule • The number of possible isomers increases rapidly as the length of the chain increases. • These molecules are isomers of the same chemical formula. • Eg. C 5 H 12 has three isomers: CH 3 l CH 3 CH 2 CH 2 CH 3 –CH CH 3 CHCH 2 CH 3 l l CH 3–C
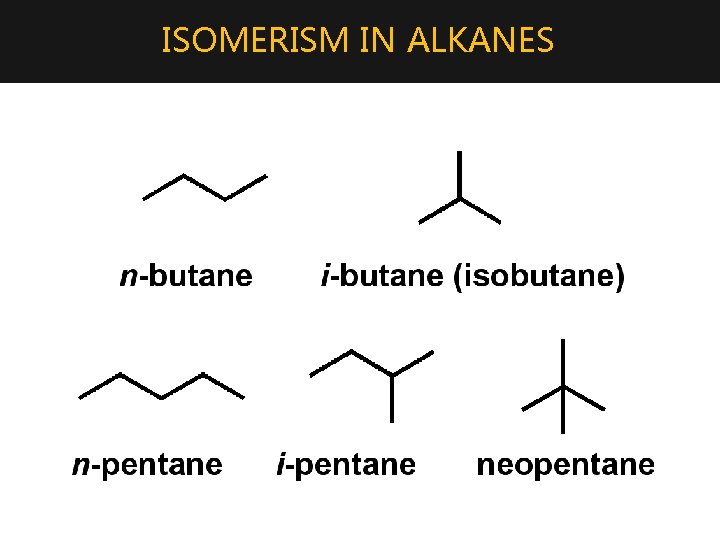
ISOMERISM IN ALKANES

LET’S PRACTICE - NAME THESE ALKANES! Try Exercise Sheet ES 03 ES 04
SOURCES OF ALKANES Distillation of crude oil • Crude oil is the main supplier to produce alkanes

SOURCES OF ALKANES Distillation of crude oil • Crude oil is normally heated in a tower, this heating produces vapours, the lighter compounds to rise higher in the tower, while the heavier compounds rise very little. Collection equipment is stationed at various levels ready to remove hydrocarbons of various masses. • Alkanes tend to be light and very non-polar, thus they travel to the top of the tower. Natural gas is the name given to the lightest of the alkanes, this gas is a combination of methane, ethane and propane. Natural gas pockets are found above large deposits of crude oil. (See picture of refinement tower below).

SOURCES OF ALKANES • Alkanes can also be made these synthetically in a lab, like Professor Wohler, in 1828 made urea. But his process is time consuming and expensive. Verify this yourself by pricing synthetic motor oil verse regular motor oil. Synthetic can easily be 15 times more expensive.

METHODS OF PREPARATION OF ALKANES Apart from the natural sources which serve as the prime producers of hydrocarbon, number of other METHODS OF SYNTHESIS of the alkanes has been designed, for examples: 1. Hydrogenation of alkenes 2. Reduction of alkyl halides A. Grignard reagent hydrolysis B. Reduction by metal and acid
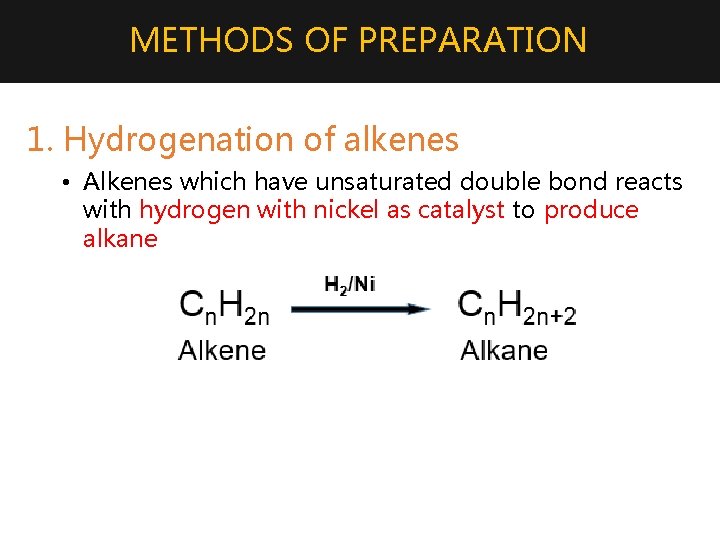
METHODS OF PREPARATION 1. Hydrogenation of alkenes • Alkenes which have unsaturated double bond reacts with hydrogen with nickel as catalyst to produce alkane

METHODS OF PREPARATION 2. Reduction of alkyl halides can be done in two ways: A. Grignard reagent hydrolysis • Grignard reagent prepared from the reaction of alkyl halide with magnesium in ether • Reaction of Grignard reagent with water produces alkane, very reactive • For example,

METHODS OF PREPARATION 2. Reduction of alkyl halides can be done in two ways: B. Reduction by metal and acid • Zinc in acetic acid is used as reagent to reduce alkyl halide to alkane • For example,

METHODS OF PREPARATION 2. Reduction of alkyl halides can be done in two ways: B. Reduction by metal and acid • Lithium aluminium hydride can also be used • For example,

PHYSICAL PROPERTIES OF ALKANES • Alkanes exist as gas, liquid or solid at room temperature: • C 1 to C 4 are gas, C 5 to C 17 mostly liquid, C 18 and above are solid • Density: 0. 6 to 0. 7 g/cm 3 • Non-polar: • Soluble in carbon tetrachloride, CCl 4 and benzene, C 6 H 6 • Insoluble in water • Melting point: • Increases as the molecule mass (no of carbon) increases

PHYSICAL PROPERTIES OF ALKANES • Boiling point: • Increases as the no of carbon increases (increase energy to break the bond) • Isomer with straight chain have higher b. p. than isomer with branches More surface area, an increase in dispersion forces, and a higher boiling point Smaller surface area, a decrease in dispersion forces, and a lower boiling point (B. P. = 50°C) (B. P. = 69°C)
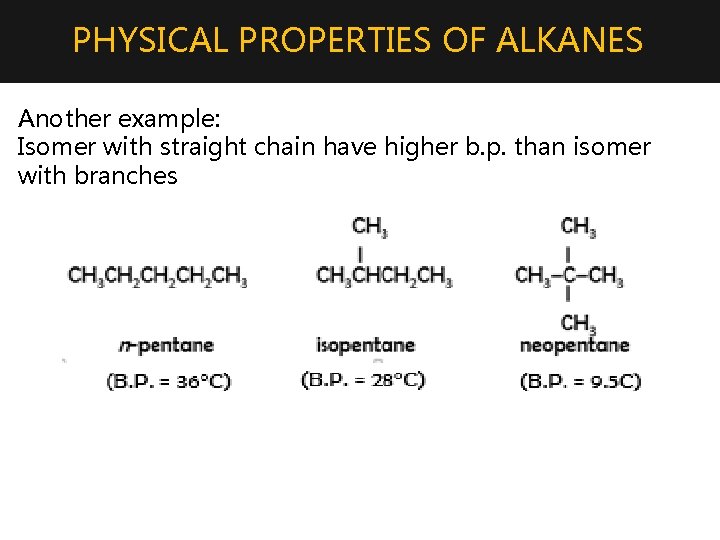
PHYSICAL PROPERTIES OF ALKANES Another example: Isomer with straight chain have higher b. p. than isomer with branches

IMPORTANT REACTIONS OF ALKANES Some important CHEMICAL REACTIONS of alkanes: 1. Combustion 2. Halogenation

IMPORTANT REACTIONS OF ALKANES 1. Combustion • All alkanes react with oxygen in a combustion reaction • Become increasingly difficult to ignite as the number of carbon increases • The general equation for complete combustion is: • When alkanes burned in excess oxygen, carbon dioxide is released with water and heat • For example, combustion of propane:

IMPORTANT REACTIONS OF ALKANES 2. Halogenation • Alkanes are very unreactive, but can be brominated or chlorinated in the presence of UV light or heat • Halogenation : replacement of H with halogen (eg. Chlorine, bromine, Iodine) to produce alkyl chloride or alkyl bromide • Takes place by free radical chain mechanism • Reaction is initiated by heat and light (which is required to break the H bond) Alkane Halogen Alkyl halide X = halogen (chlorine, bromine) What type of reaction is this?

IMPORTANT REACTIONS OF ALKANES 2. Halogenation (cont. ) • A substitution reaction in which a hydrogen is removed from the alkane and a halogen ( Br or Cl ) takes its place. The halogen atom can substitute at any site on the molecule. • The halogenated products can react further to give disubstituted or tri-substituted products (and so on). Reaction is not selective, mixture of products are formed.

2. 4 ALKENES, ADDITION POLYMERIZATION AND THE CHEMISTRY OF RUBBER
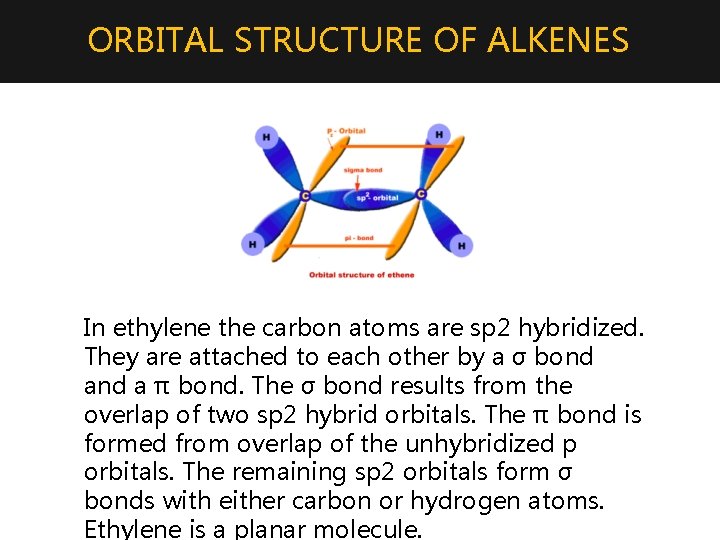
ORBITAL STRUCTURE OF ALKENES In ethylene the carbon atoms are sp 2 hybridized. They are attached to each other by a σ bond a π bond. The σ bond results from the overlap of two sp 2 hybrid orbitals. The π bond is formed from overlap of the unhybridized p orbitals. The remaining sp 2 orbitals form σ bonds with either carbon or hydrogen atoms. Ethylene is a planar molecule.

NOMENCLATURE OF ALKENES • Very similar to alkanes – naming system is very similar prefix – root – ene • Also made up of C and H (that’s why it is also classified as hydrocarbons) but it is unsaturated ! • Except that it contains DOUBLE BOND in the longest carbon chain • General formula for alkenes: • The first two are: • Ethene C 2 H 4 (or Ethylene) • Propene C 3 H 6 Cn. H 2 n
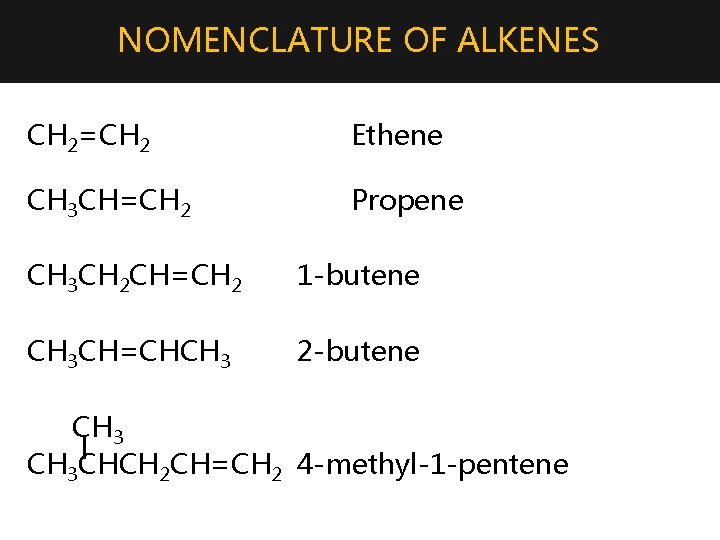
NOMENCLATURE OF ALKENES CH 2=CH 2 Ethene CH 3 CH=CH 2 Propene CH 3 CH 2 CH=CH 2 1 -butene CH 3 CH=CHCH 3 2 -butene CH 3 l CH 3 CHCH 2 CH=CH 2 4 -methyl-1 -pentene
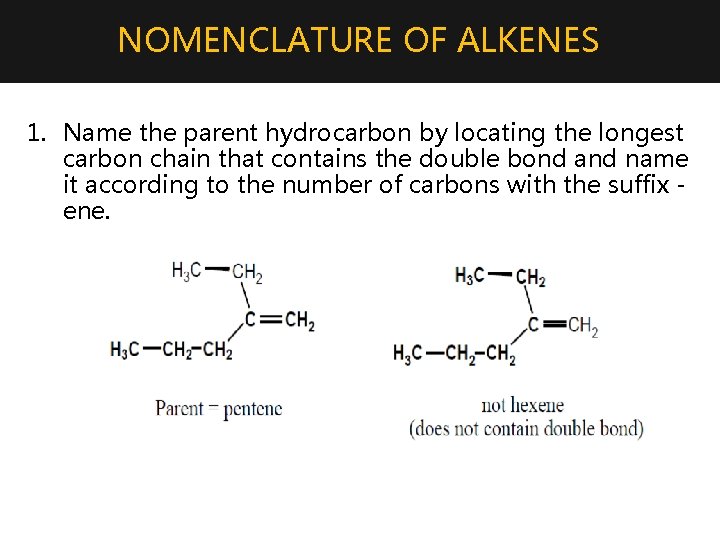
NOMENCLATURE OF ALKENES 1. Name the parent hydrocarbon by locating the longest carbon chain that contains the double bond and name it according to the number of carbons with the suffix ene.
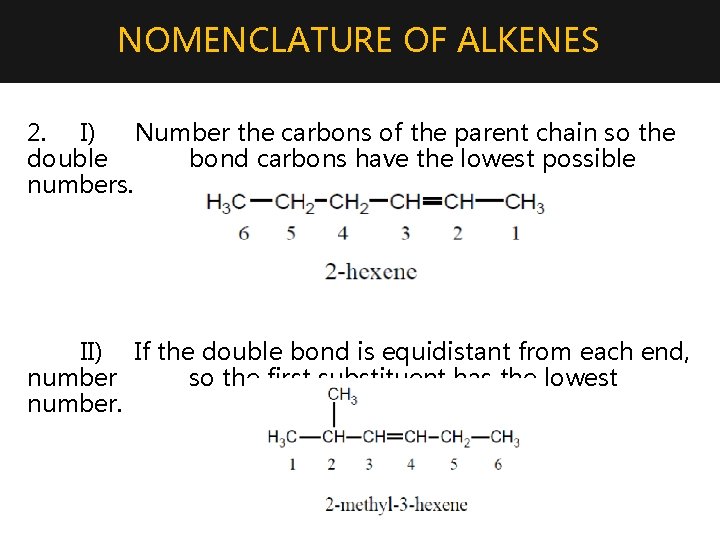
NOMENCLATURE OF ALKENES 2. I) Number the carbons of the parent chain so the double bond carbons have the lowest possible numbers. II) If the double bond is equidistant from each end, number so the first substituent has the lowest number.

NOMENCLATURE OF ALKENES 3. Write out the full name, numbering the substituents according to their position in the chain and list them in alphabetical order. 4. Indicate the double bond by the number of the first alkene carbon. 5. If more than one double bond is present, indicate their position by using the number of the first carbon of each double bond and use the suffix -diene (for 2 double bonds), -triene (for 3 double bonds), -tetraene (for 4 double bonds), etc.
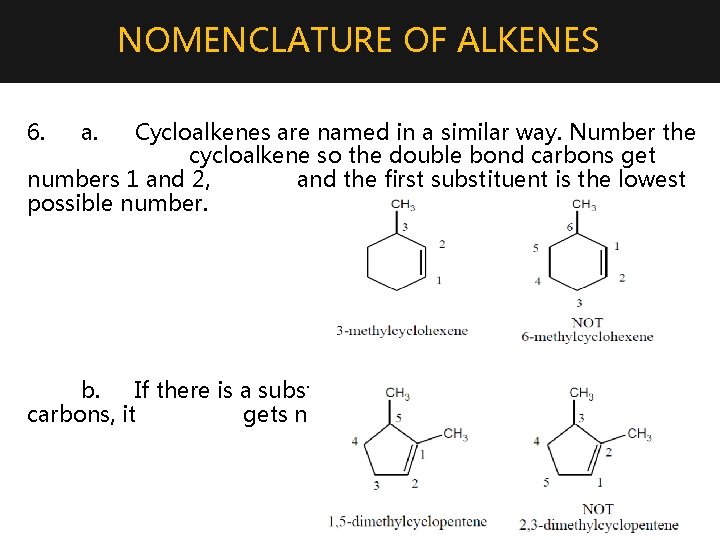
NOMENCLATURE OF ALKENES 6. a. Cycloalkenes are named in a similar way. Number the cycloalkene so the double bond carbons get numbers 1 and 2, and the first substituent is the lowest possible number. b. If there is a substituent on one of the double bond carbons, it gets number 1.
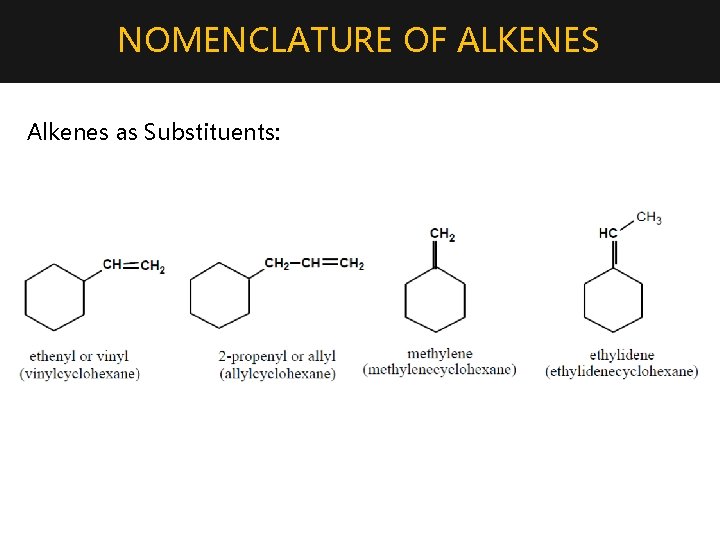
NOMENCLATURE OF ALKENES Alkenes as Substituents:

LET’S PRACTICE – NAME THESE ALKENES! Complete Exercise Sheet ES 05, ES 06
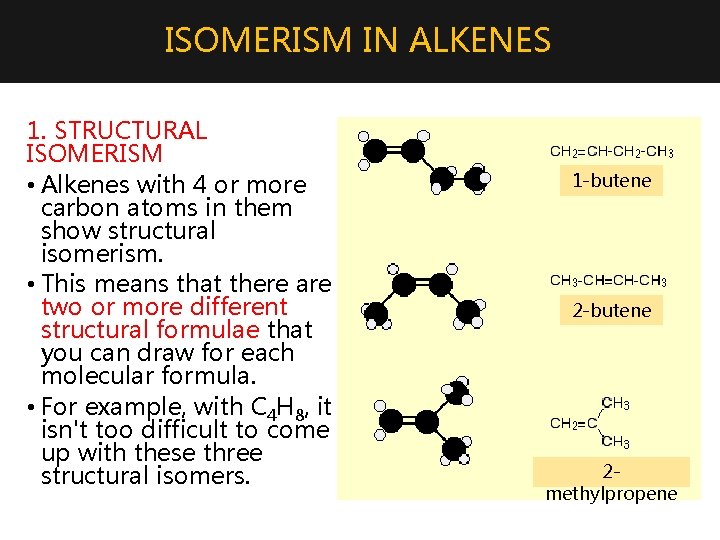
ISOMERISM IN ALKENES 1. STRUCTURAL ISOMERISM • Alkenes with 4 or more carbon atoms in them show structural isomerism. • This means that there are two or more different structural formulae that you can draw for each molecular formula. • For example, with C 4 H 8, it isn't too difficult to come up with these three structural isomers. 1 -butene 2 methylpropene
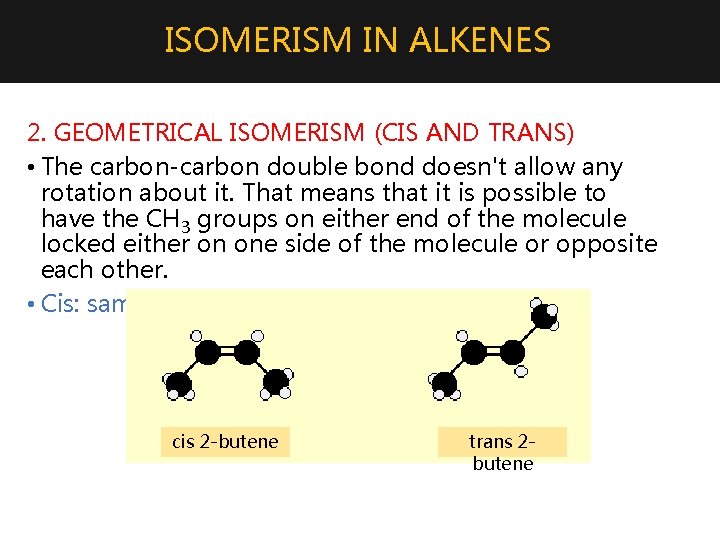
ISOMERISM IN ALKENES 2. GEOMETRICAL ISOMERISM (CIS AND TRANS) • The carbon-carbon double bond doesn't allow any rotation about it. That means that it is possible to have the CH 3 groups on either end of the molecule locked either on one side of the molecule or opposite each other. • Cis: same side, trans: opposite side cis 2 -butene trans 2 butene

MORE NOTES ON ISOMERISM Refer Slide IS 02 c Complete Exercise Sheet ES 07, ES 08, ES 09, ES 10

PHYSICAL PROPERTIES OF ALKENES • Mostly similar to alkanes • Ethene, propene and the various butenes are gases at room temperature • Alkenes are virtually insoluble in water, but dissolve in organic solvents • Density: 0. 6 to 0. 7 g/cm 3 • Melting point: • Increases as the molecule mass increases • Boiling point: • Increases as the no. of carbon increases • Branched alkenes have lower boiling points compared to straight alkenes • For example, isobutene have lower b. p. than straight butene
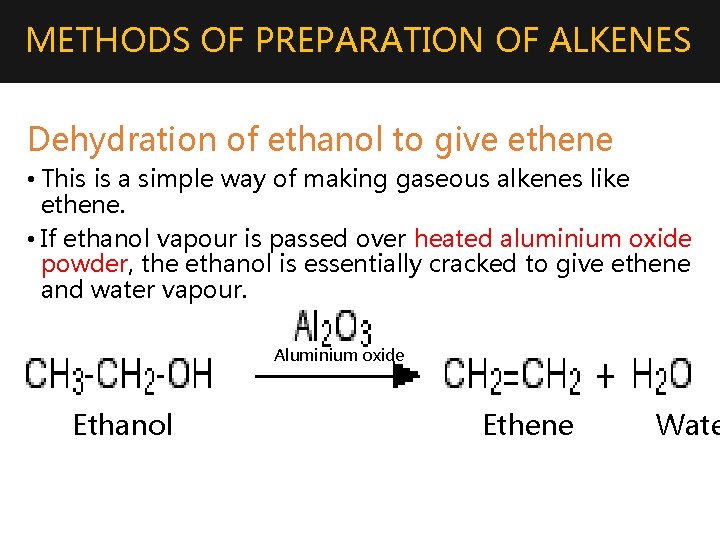
METHODS OF PREPARATION OF ALKENES Dehydration of ethanol to give ethene • This is a simple way of making gaseous alkenes like ethene. • If ethanol vapour is passed over heated aluminium oxide powder, the ethanol is essentially cracked to give ethene and water vapour. Aluminium oxide Ethanol Ethene Wate

IMPORTANT REACTIONS OF ALKENES Some important CHEMICAL REACTIONS of alkenes: 1. Hydrogenation of alkene 2. Halogenation of alkene 3. Hydrohalogenation of alkene

IMPORTANT REACTIONS OF ALKENES 1. Hydrogenation of alkene • Addition of hydrogen • Ethene reacts with hydrogen in the presence of a finely divided nickel catalyst at a temperature of about 150°C. Ethane is produced. • This is a fairly pointless reaction because ethene is a far more useful compound than ethane!

IMPORTANT REACTIONS OF ALKENES 2. Halogenation of alkene • Addition of halogen to alkene, occurs at room temperature • For example, bromine adds to give 1, 2 -dibromoethane. What type of reaction is this? • Reaction with bromine happens at room temperature. • If a gaseous alkene like ethene, bubble it through either pure liquid bromine or a solution of bromine in an organic solvent like tetrachloromethane. The reddish-brown bromine is decolourised as it reacts with the alkene. • A liquid alkene (like cyclohexene) can be shaken with liquid bromine or its solution in tetrachloromethane. • Chlorine reacts faster than bromine. Iodine reacts much, much more slowly. (Reactivity: Cl > Br > I)

ADDITION TO UNSYMMETRICAL ALKENES 3. Hydrohalogenation • The addition of hydrogen halides (HCl, HBr and HI) to alkenes will give haloalkenes (alkyl halides) • Carries out either with the pure reagents or in the presence of a polar solvent such as acetic acid • For example, the addition of HCl to propene gives two products: 2 -chloropropane (major products) and 1 chloropropane (minor products), following Markovnikov’s Markonikov’s Rule In the addition of HX to alkene, hydrogen adds to the doubly bonded carbon that has the greatest number

MANUFACTURING ETHANOL 4. Hydration of alkenes • Addition of water to alkenes will produce alcohol. • For example, ethanol is manufactured by reacting ethene with steam. The reaction is reversible • Only 5% of the ethene is converted into ethanol at each pass through the reactor. By removing the ethanol from the equilibrium mixture and recycling the ethene, it is possible to achieve an overall 95% conversion.
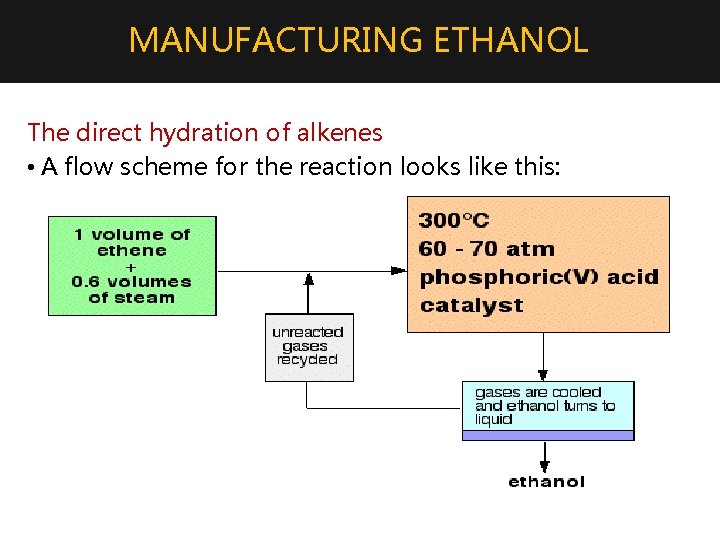
MANUFACTURING ETHANOL The direct hydration of alkenes • A flow scheme for the reaction looks like this:

POLYMERIZATION OF ALKENES

POLYMERIZATION Alkenes can undergo addition reactions with themselves to form a long chain polymer molecule. The reaction is known as addition polymerization where no other byproduct is formed. Intro video - What are polymers? https: //www. youtube. com/watch? v=b. Ji 8 x 7 b. KHq. Q
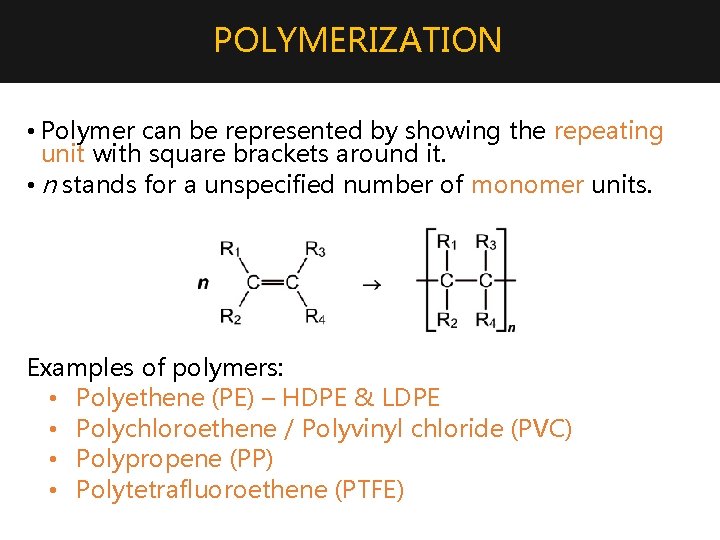
POLYMERIZATION • Polymer can be represented by showing the repeating unit with square brackets around it. • n stands for a unspecified number of monomer units. Examples of polymers: • Polyethene (PE) – HDPE & LDPE • Polychloroethene / Polyvinyl chloride (PVC) • Polypropene (PP) • Polytetrafluoroethene (PTFE)
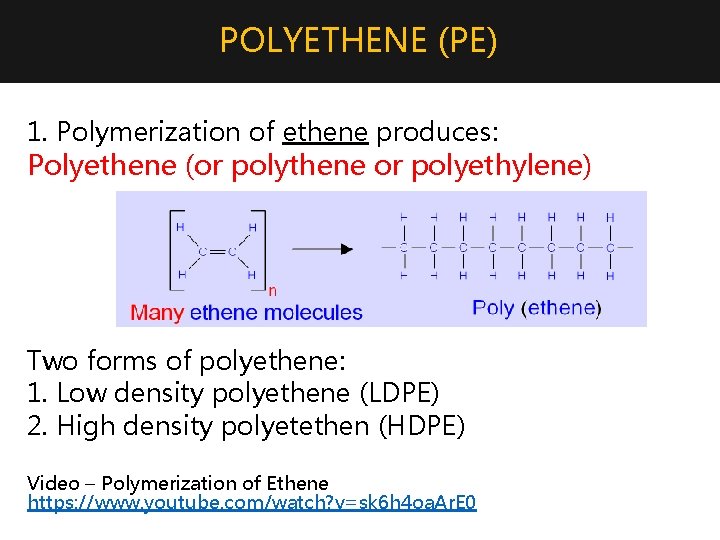
POLYETHENE (PE) 1. Polymerization of ethene produces: Polyethene (or polythene or polyethylene) Two forms of polyethene: 1. Low density polyethene (LDPE) 2. High density polyetethen (HDPE) Video – Polymerization of Ethene https: //www. youtube. com/watch? v=sk 6 h 4 oa. Ar. E 0

POLYETHENE (LDPE) Low density polyethene (LDPE) Manufacturing conditions: Temperature : about 200°C Pressure : about 2000 atmospheres Initiator : a small amount of oxygen as an impurity • The number of molecules joining up is very variable, but is in the region of 2000 to 20000. Uses: • Plastic bags and other similar low strength and flexible sheet materials.

POLYETHENE (HDPE) High density polyethene (HDPE) Manufacturing conditions: Temperature : Pressure : Catalyst : compounds about 60°C low - a few atmospheres Ziegler-Natta catalysts or other metal Uses: • Plastic milk bottles and similar containers, washing up bowls, plastic pipes and so on. Look for the letters HDPE near the recycling symbol.

POLYPROPENE (PP) 2. Polymerization of propene (or polypropylene) produces: Polypropene (or polypropylene) (PP) Manufacture: It is obtained by polymerising propylene in the presence of Ziegler-Natta catalyst. Uses: • Packaging and labelling, textiles, stationery, plastic parts and reusable containers, laboratory equipment

POLYPROPENE (PP)

POLYCHLOROETHENE / POLYVINYL CHLORIDE 3. Polymerization of chloroethene (or vinyl chloride) produces: Polychloroethene (or polyvinyl chloride) (PVC) Manufacturing conditions: Temperature : about 325 -350 K Pressure : about 13 atmospheres Initiator : organic peroxides • Plasticizers are added to the poly (chloroethene) to reduce the effectiveness of these attractions and make the plastic more flexible. The more plasticizer you add, the more flexible it becomes. Uses: • Guttering, plastic windows, electrical cable insulation, sheet materials for flooring and other uses, footwear, clothing, and so on
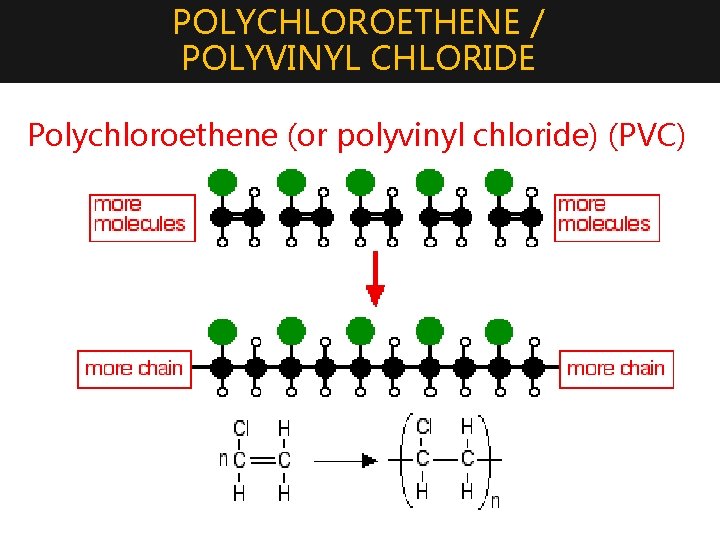
POLYCHLOROETHENE / POLYVINYL CHLORIDE Polychloroethene (or polyvinyl chloride) (PVC)

POLYTETRAFLUOROETHENE (PTFE) 4. Polymerization of tetrafluoroethene produces: Polytetrafluoroethene (PTFE) (Teflon) Manufacturing: The monomer is transformed into the polymer, PTFE, by radical polymerization. The reaction is carried out by passing TFE into water containing a radical initiator, e. g. ammonium persulfate, (NH 4)2 S 2 O 8, at 310 -350 K and a pressure of 10 -20 atm. Uses: • It is chemically inert and resistant to attack by corrosive reagent. Has non-stick properties - which are the basis for its most familiar uses in non-stick kitchen and garden utensils. For the same reason, it can also be used in
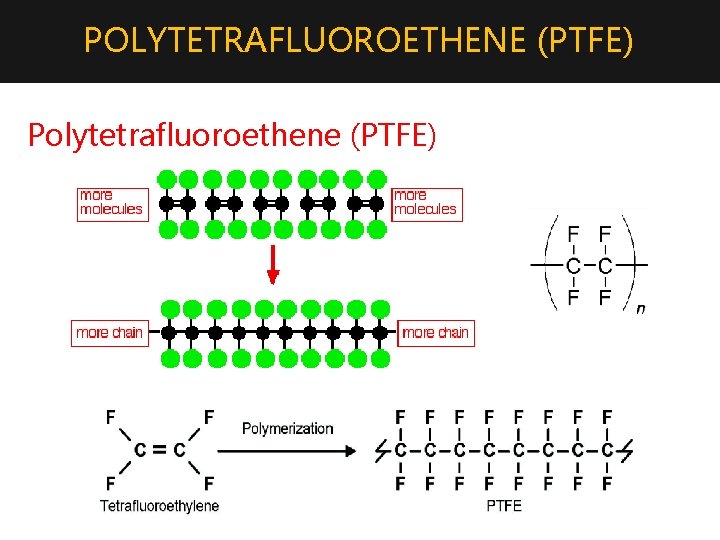
POLYTETRAFLUOROETHENE (PTFE) Polytetrafluoroethene (PTFE)
Complete ES 17

THE CHEMISTRY OF RUBBER

NATURAL RUBBER • Natural rubber is a hydrocarbon polymer built up from the monomer isoprene. Raw rubber obtained from milky sap (latex) of the rubber tree does not possess the characteristics of the rubber with which are familiar. In order to give it strength and elasticity it is vulcanised. In the vulcanisation process, raw rubber is mixed with small amount of sulphur and heated. The sulphur reacts with the polymer molecules forming a cross-linked network.

NATURAL RUBBER • The crosslinking gives mechanical strength to the rubber. In addition, fillers such as carbon black and zinc oxide are usually added to the crude rubber before vulcanisation in order to improve its wearing characteristics.

SYNTHETIC RUBBER Synthetic rubber a. Neoprene rubber. It was the first synthetic rubber manufactured on large scale. The monomer, chloroprene, is prepared from acetylene. Neoprene is particularly resistant to chemical action and is therefore used in making hoses for petrol and oil, and containers for corrosive chemicals. b. Styrene-Butadiene Rubber (SBR). It is a copolymer made from styrene and 1, 3 -butadiene.
- Slides: 87