Efficient Modeling of Excitable Cells Using Hybrid Automata

Efficient Modeling of Excitable Cells Using Hybrid Automata Radu Grosu SUNY at Stony Brook Joint work with Pei Ye, Emilia Entcheva and Scott A. Smolka

Talk Outline 1. Biological Background 2. Motivation 3. Computational Background 4. Hybrid Automata 5. HA Models of Excitable Cells 6. Simulation Results 7. Conclusions & Future Work

Main Goal • Computational Efficiency: – Making large-scale simulation practical • Formal Analysis (in the future): – Reachability – Safety – Liveness
Background • Excitable cells – Neurons – Cardiac myocytes – Skeletal muscle cells • Different concentrations of ions inside and outside of cells form: – Trans-membrane potential – Ion currents cross the cell membrane through channels

Squid Giant Axon (Animation from Marine Bilogical Laboratory, MA) 1. Squid at rest. 2. Mantle opens. Water enters the mantle cavity. 3. A signal from the brain is sent to the stellate ganglion which is connected to nerve cells (axons) distributed through the mantle. 4. Nerve impulses travel the length of these axons. 5. The muscles contract synchronously, rapidly closing the mantle. 6. Water is forced out through the siphon, producing a jet action.
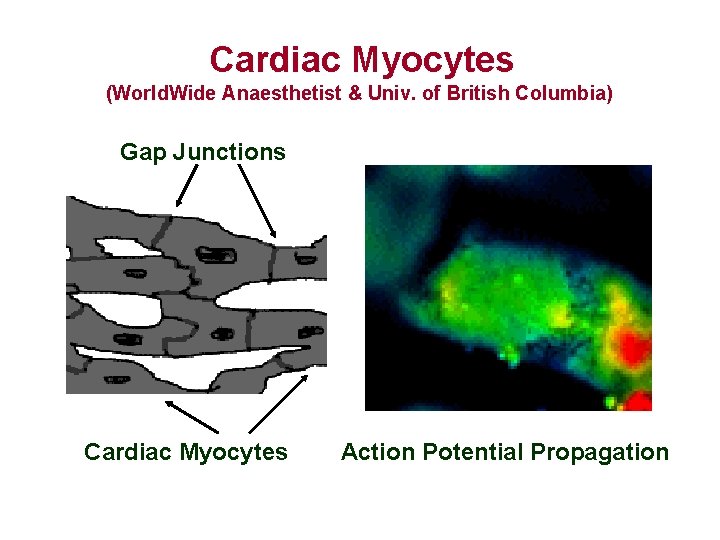
Cardiac Myocytes (World. Wide Anaesthetist & Univ. of British Columbia) Gap Junctions Cardiac Myocytes Action Potential Propagation

2 D Simulations of Atrial Fibrillation (Kneller et al. , Mc. Gill) Single Spiral Wave Fast Spiral Wave Breakup Atrial Fibrillation
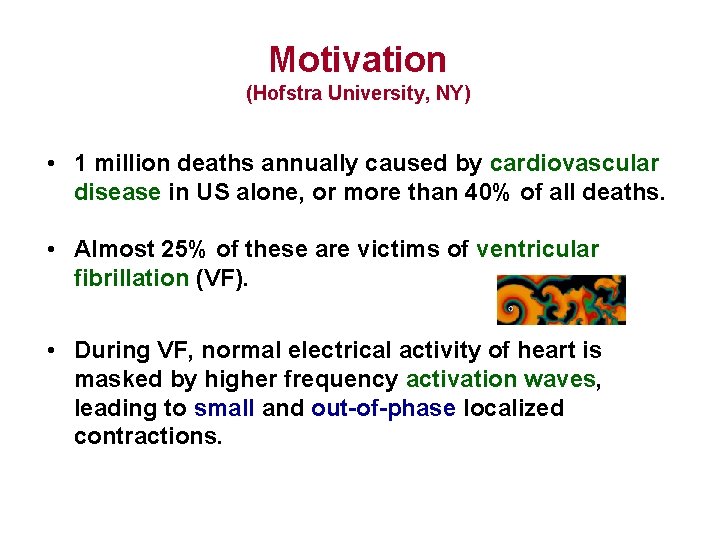
Motivation (Hofstra University, NY) • 1 million deaths annually caused by cardiovascular disease in US alone, or more than 40% of all deaths. • Almost 25% of these are victims of ventricular fibrillation (VF). • During VF, normal electrical activity of heart is masked by higher frequency activation waves, leading to small and out-of-phase localized contractions.

Mathematical Models • Hodgkin-Huxley (HH) model – Membrane potential for squid giant axon – Developed in 1952 – Framework for the following models • Luo-Rudy (LRd) model – Model for cardiac cells of guinea pig – Developed in 1991 • Neo-Natal Rat (NNR) model – Being developed in Stony Brook University by Emilia Entcheva et al.

Who? Alan Lloyd Hodgkin *1914 +1998 Andrew Fielding Huxley *1917 Nobel Preis for Physiology or Medicine in 1963 "for their discoveries concerning the ionic mechanisms involved in excitation and inhibition in the peripheral and central portions of the nerve cell membrane"
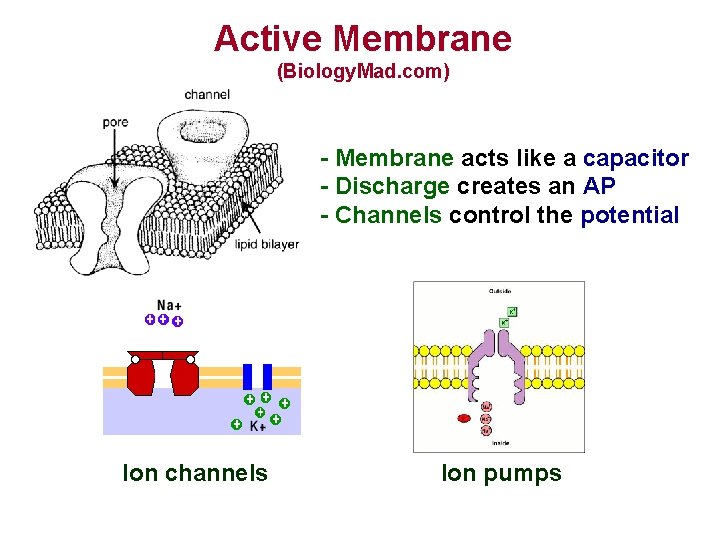
Active Membrane (Biology. Mad. com) - Membrane acts like a capacitor - Discharge creates an AP - Channels control the potential Ion channels Ion pumps
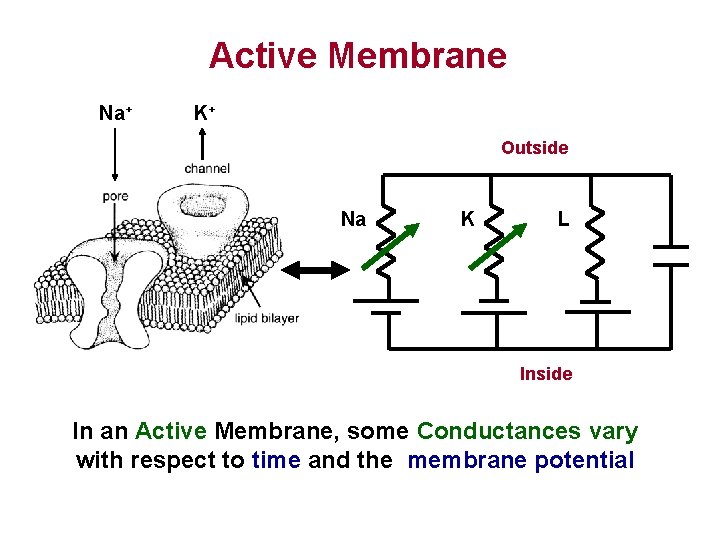
Active Membrane Na+ K+ Outside Na K L Inside In an Active Membrane, some Conductances vary with respect to time and the membrane potential
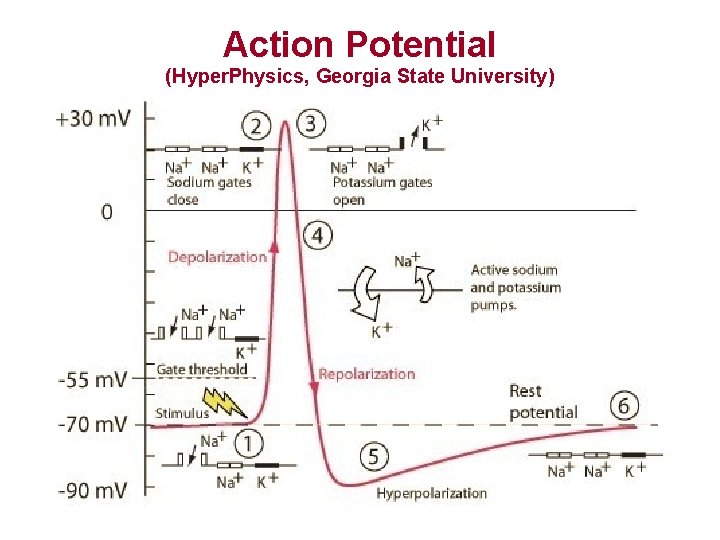
Action Potential (Hyper. Physics, Georgia State University)
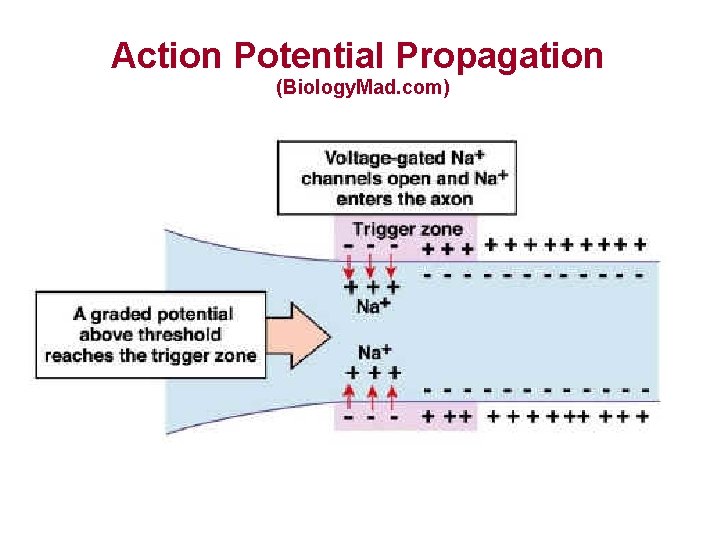
Action Potential Propagation (Biology. Mad. com)
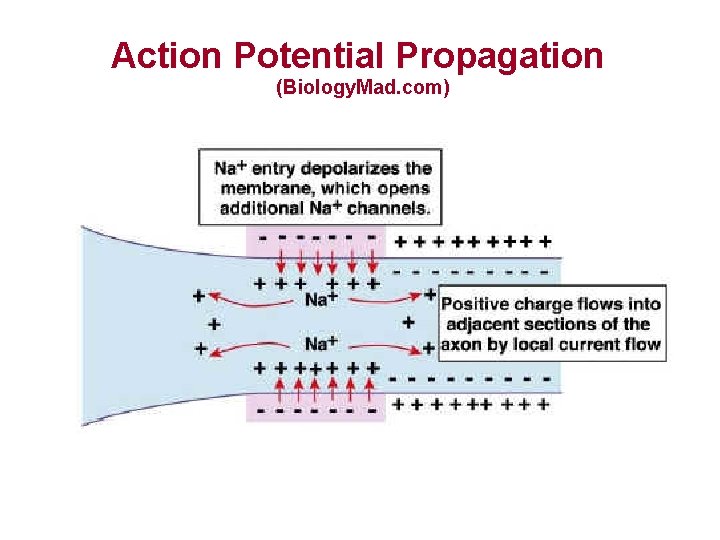
Action Potential Propagation (Biology. Mad. com)
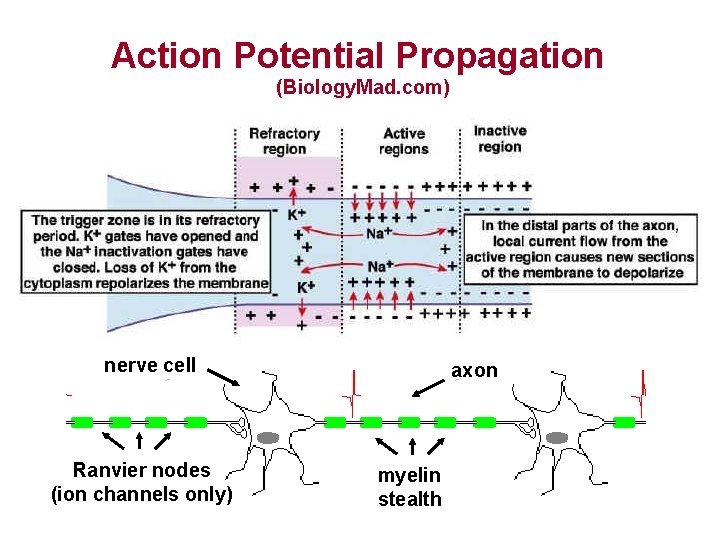
Action Potential Propagation (Biology. Mad. com) nerve cell Ranvier nodes (ion channels only) axon myelin stealth
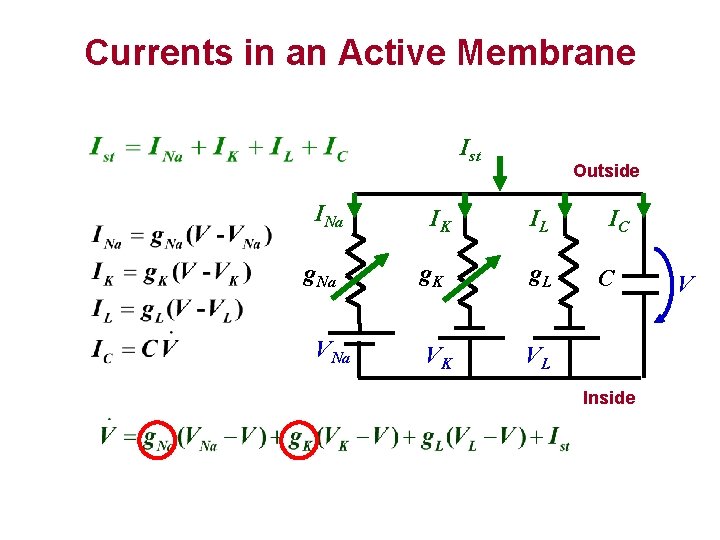
Currents in an Active Membrane Ist INa g. Na VNa IK Outside IL g. K g. L VK VL IC C Inside V

The Potasium Channel (Pictures from B. Babadi, Univ. of Teheran) • Has four similar slow subunits. • A subunit can be either “open” or “closed”. • Channel is open iff all 4 subunits are open.

Kinetics of Potasium Subunits (Pictures from B. Babadi, Univ. of Teheran)

The Sodium Channel (Pictures from B. Babadi, Univ. of Teheran) m m • Has three similar fast subunits and a single slow subunit. h m

The Full Hodgkin-Huxley Model

Hodgkin-Huxley Model in Action (Applet of A. Fodor, Stanford)

Hybrid Automata (HA) (Alur, Henzinger, Sifakis and others) • Combine both – Continuous behavior (Differential Equations) – Discrete transitions • Advantages – Simplicity – Rich descriptive ability
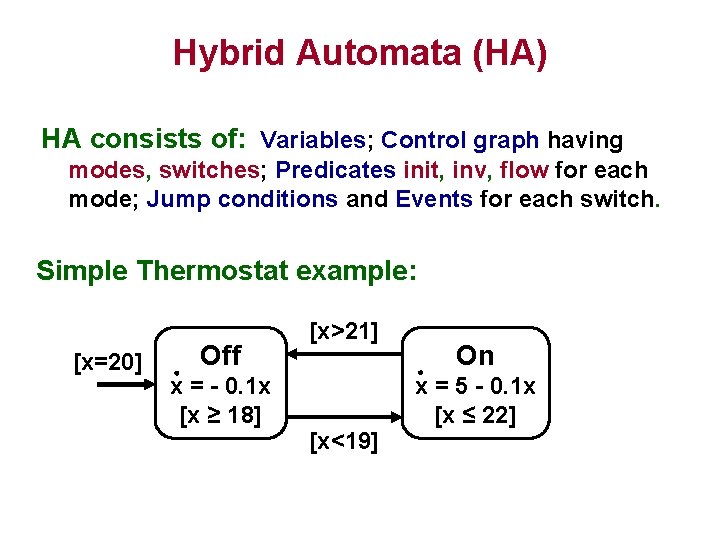
Hybrid Automata (HA) HA consists of: Variables; Control graph having modes, switches; Predicates init, inv, flow for each mode; Jump conditions and Events for each switch. Simple Thermostat example: [x=20] Off x = - 0. 1 x [x ≥ 18] [x>21] [x<19] On x = 5 - 0. 1 x [x ≤ 22]
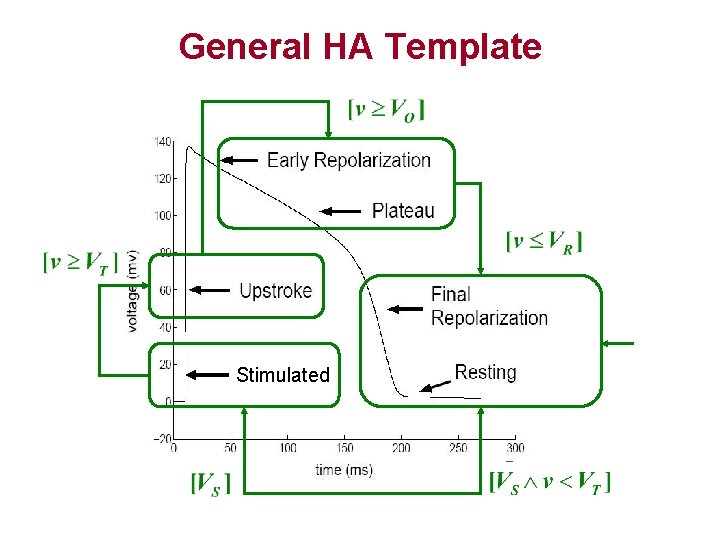
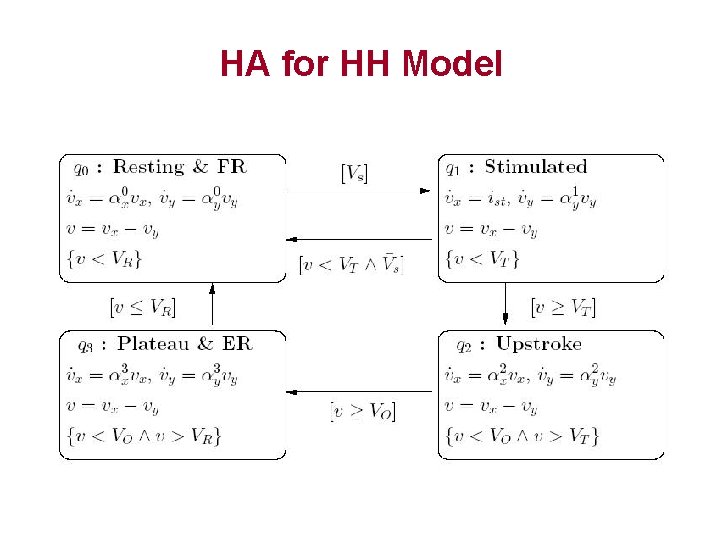
General HA Template Stimulated

Assumptions for the Flows 1. Each mode corresponds to an open/closed configuration of the gates. • Gate dependence on V is factored into the modes. 2. Sodium and potasium gates (conductances) are mutually independent of each other. 3. Gate (conductance) behavior within a mode is given by a linear differential equation • A step function approximation is too crude.

Assumptions for the Flows Problem: Equation (3) is nonlinear. Solution: Assume the inward (INa) and outward (IK+ IL) currents are linear!

Is this justified? (Applet of A. Fodor, Stanford)

Assumptions for the Flows Hence: Now take: Then:
HA for HH Model
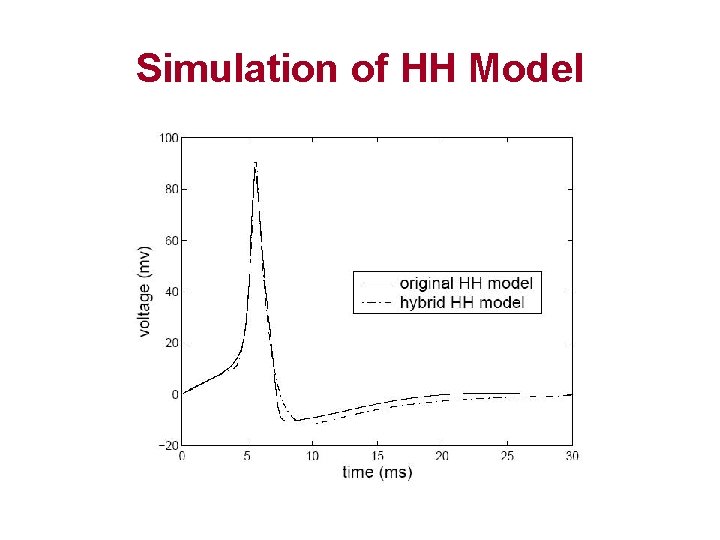
Simulation of HH Model
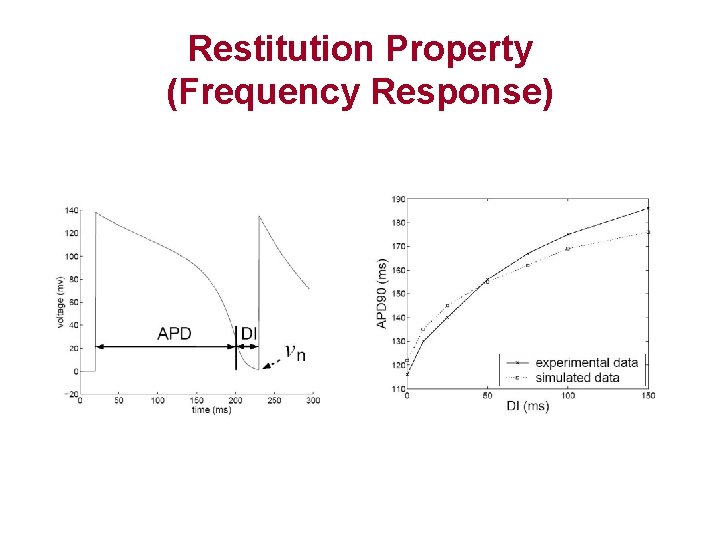
Restitution Property (Frequency Response)

New Features for HA Models 1. Capture dependence on the Ca 2+ ion: – Add new flow variable vz 2. Capture restitution nonlinearity: – Add new state variable vn remembering voltage value when stimulus occurs. – Adjust AP slope with cycle constant f: – Adjust AP height & duration with constants g, h:

HA for NNR Model
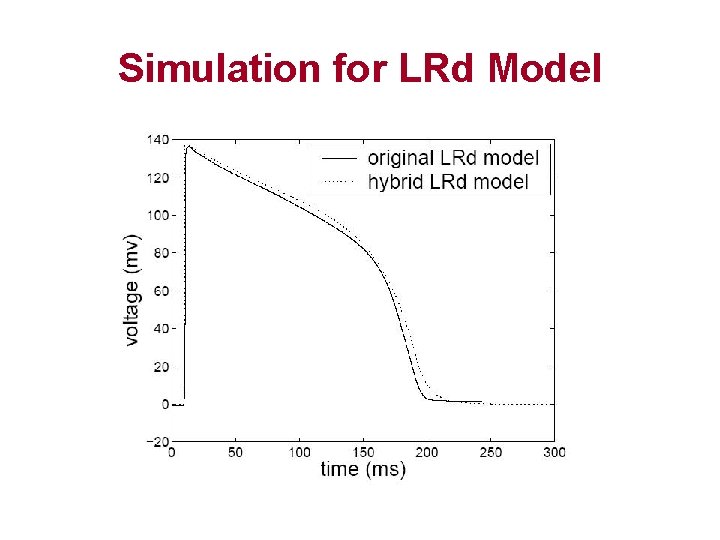
Simulation for LRd Model
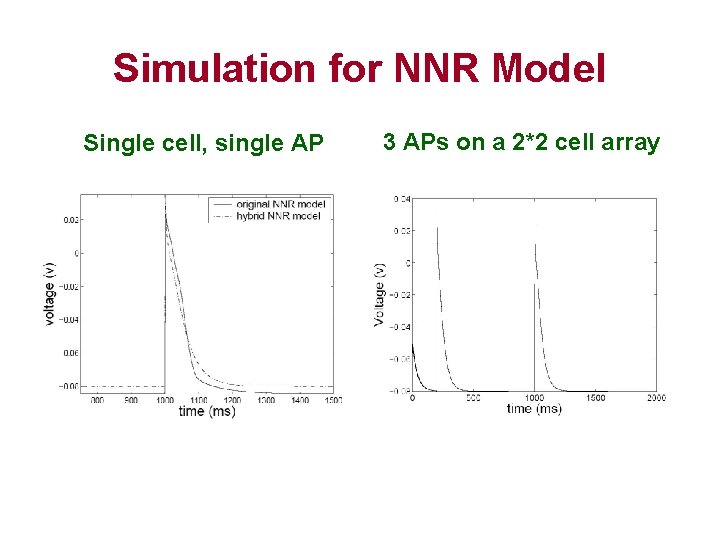
Simulation for NNR Model Single cell, single AP 3 APs on a 2*2 cell array

Large-scale Spatial Simulation for NNR Model Re-entry on a 400*400 cell array
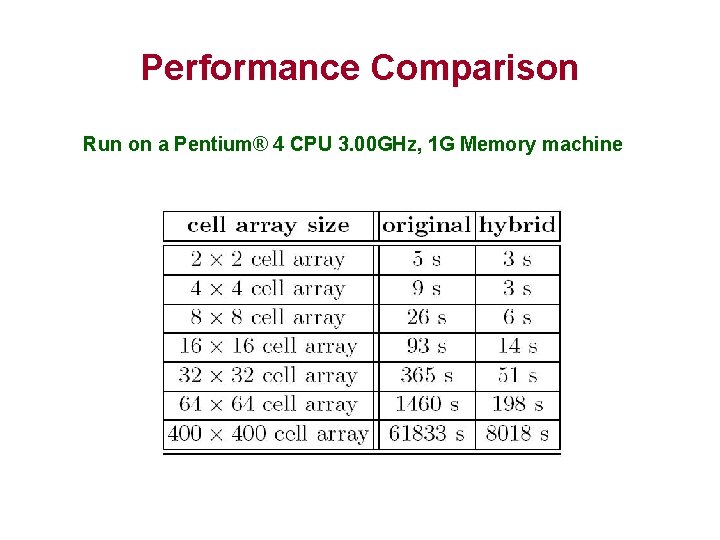
Performance Comparison Run on a Pentium® 4 CPU 3. 00 GHz, 1 G Memory machine

Conclusion • Cell excitation used to be modeled by ODE systems – Hodgkin-Huxley – Luo-Rudy – Neo-Natal Rat • Hybrid automata approach combines – Differential equations – Discrete mode switches • Simulation by using Hybrid automata – Accurate – Efficient – Easily extended to other complex biological systems

Future Work • Use optimization techniques to automatically derive HA model parameters. • Develop simpler spatial model to further improve efficiency (FDM vs. FEM). • Formal analysis: ventricular fibrillation as a reachability property. • Long-term work: improved pacemaker/defibrillator technology, communicate with prosthesis robots.
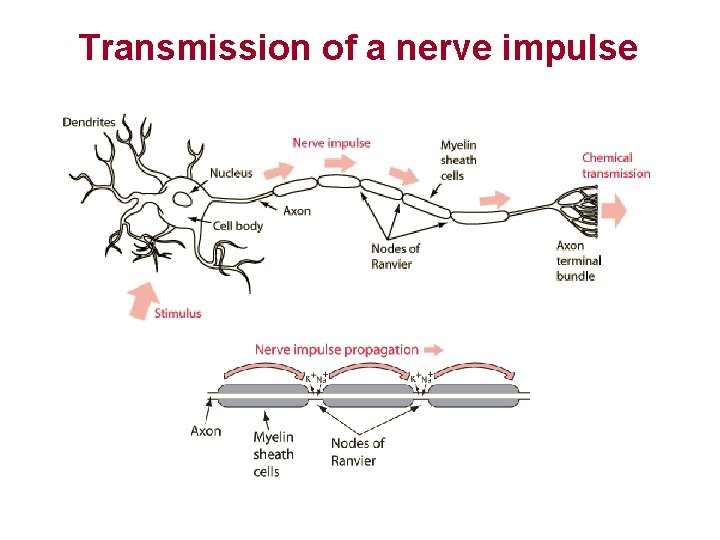
Transmission of a nerve impulse
Ions and Channels of Excitable Cells 2+ Ca Cell K+ K+ Na+ Ca 2+ K+ K+ Na+ K+ Ca 2+ channel K+ Na+ Ca 2+ Na+ Cell K+ K+ Ca 2+ K+ Na+ Ca 2+ Na+

The Giant Axon of Squid
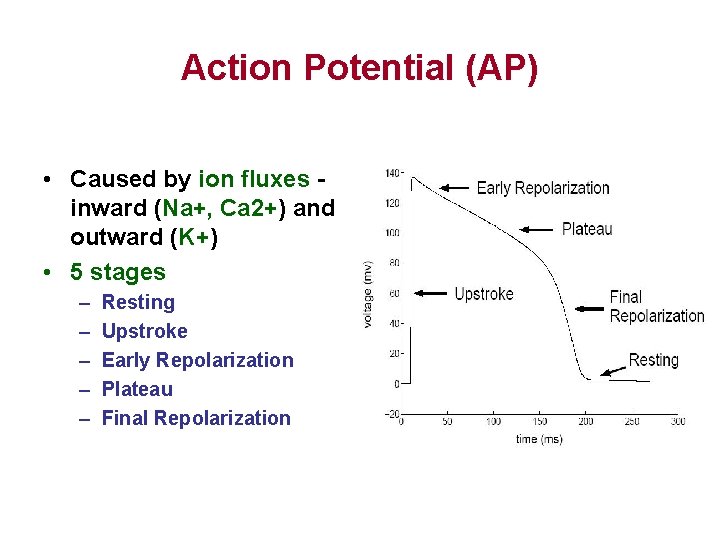
Action Potential (AP) • Caused by ion fluxes inward (Na+, Ca 2+) and outward (K+) • 5 stages – – – Resting Upstroke Early Repolarization Plateau Final Repolarization

Restitution Property • Excitable cells respond differently to stimuli with different frequency. • Each cycle is characterized by: – Action Potential Duration (APD) – Diastolic Interval (DI) • Longer DI, longer APD

Hodgkin-Huxley Model • • • C: Cell capacitance V: Trans-membrane voltage gna, gk, g. L: Maximum channel conductance Ena, Ek, EL: Reversal potential m, n, h: Ion channel gate variables Ist: Stimulation current

Two Ways of Abstraction • Rational method: derive the flow functions from the differential equations in the original model • Empirical method: use curve-fitting techniques to get the flow functions with the form chosen (here we use the form ).

General HA Template • 4 control modes: – – Resting and Final repolarization (FR) Stimulated Upstroke Early repolarization (ER) and Plateau • Threshold voltage monitoring mode switches – Vo, VT and VR • Event VS represents the presence of stimulus

HA for LRd Model

New Features of HA for LRd and NNR Model • Add vz to capture dependence on the Ca 2+ ion • Use vn to remember the current voltage when the next stimulus occurs. – determines the time cell stays in mode ER and plateau – Thus, APD will change with DI • For NNR model, define and thus the threshold voltages are also influenced by DI.
- Slides: 50