Efficacy and safety of subcutaneous and intravenous rituximab

Efficacy and safety of subcutaneous and intravenous rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone in first-line diffuse large B-cell lymphoma: the randomized Mab. Ease study by Pieternella Lugtenburg, Irit Avivi, Henriette Berenschot, Osman Ilhan, Jean Pierre Marolleau, Arnon Nagler, Antonio Rueda, Monica Tani, Mehmet Turgut, Stuart Osborne, Rodney Smith, and Michael Pfreundschuh haematol Volume 102(11): 1913 -1922 October 27, 2017 © 2017 by Ferrata Storti Foundation
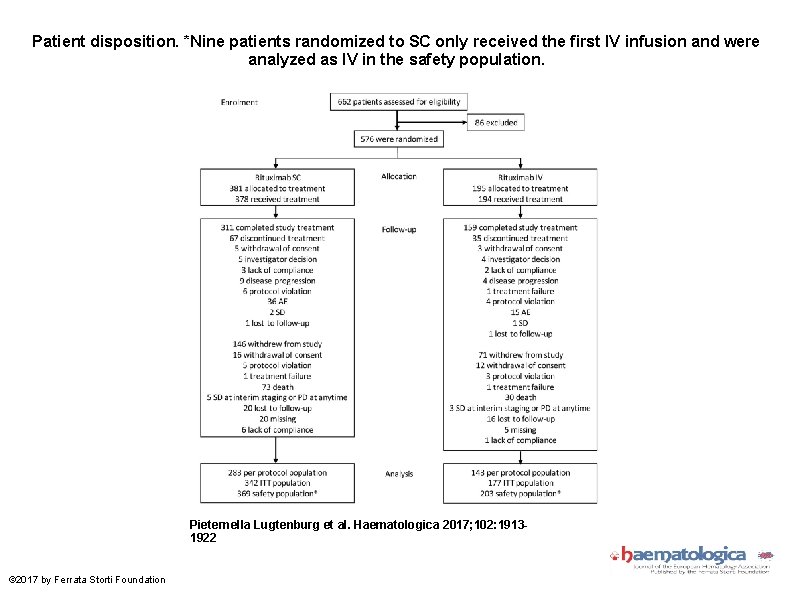
Patient disposition. *Nine patients randomized to SC only received the first IV infusion and were analyzed as IV in the safety population. Pieternella Lugtenburg et al. Haematologica 2017; 102: 19131922 © 2017 by Ferrata Storti Foundation
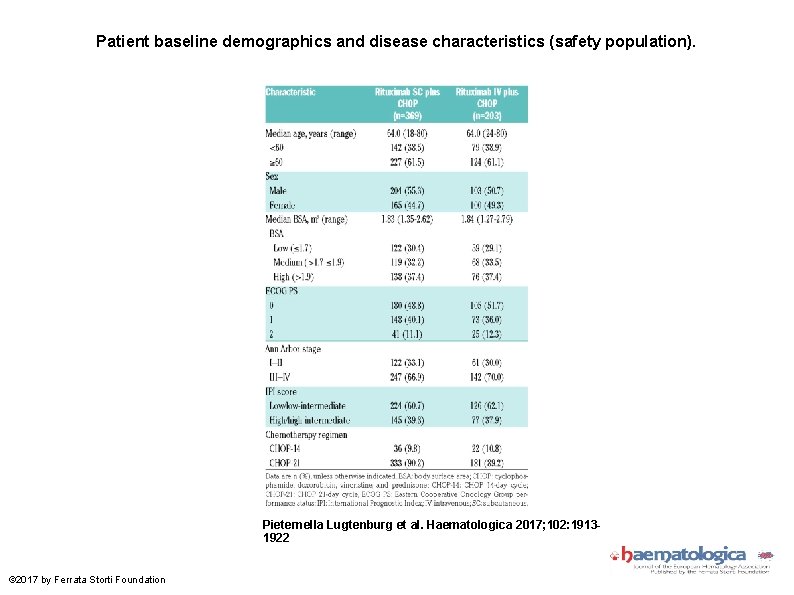
Patient baseline demographics and disease characteristics (safety population). Pieternella Lugtenburg et al. Haematologica 2017; 102: 19131922 © 2017 by Ferrata Storti Foundation
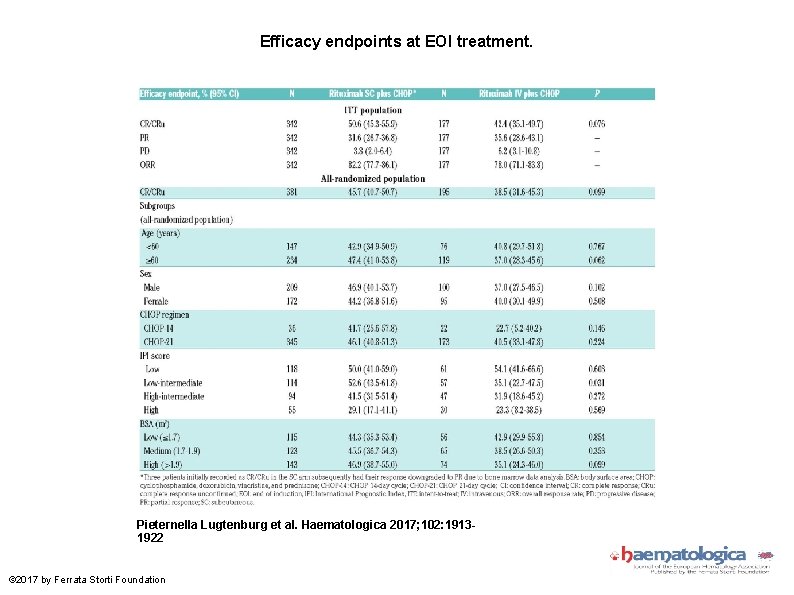
Efficacy endpoints at EOI treatment. Pieternella Lugtenburg et al. Haematologica 2017; 102: 19131922 © 2017 by Ferrata Storti Foundation
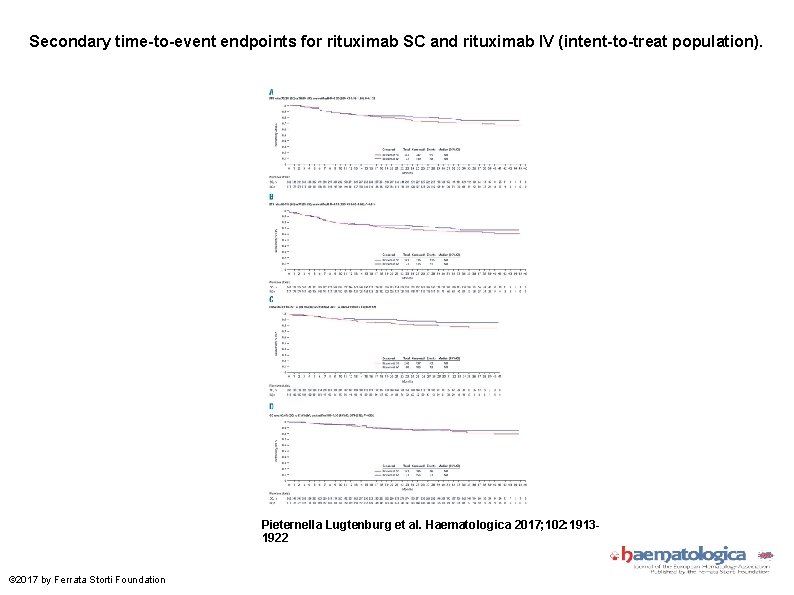
Secondary time-to-event endpoints for rituximab SC and rituximab IV (intent-to-treat population). Pieternella Lugtenburg et al. Haematologica 2017; 102: 19131922 © 2017 by Ferrata Storti Foundation
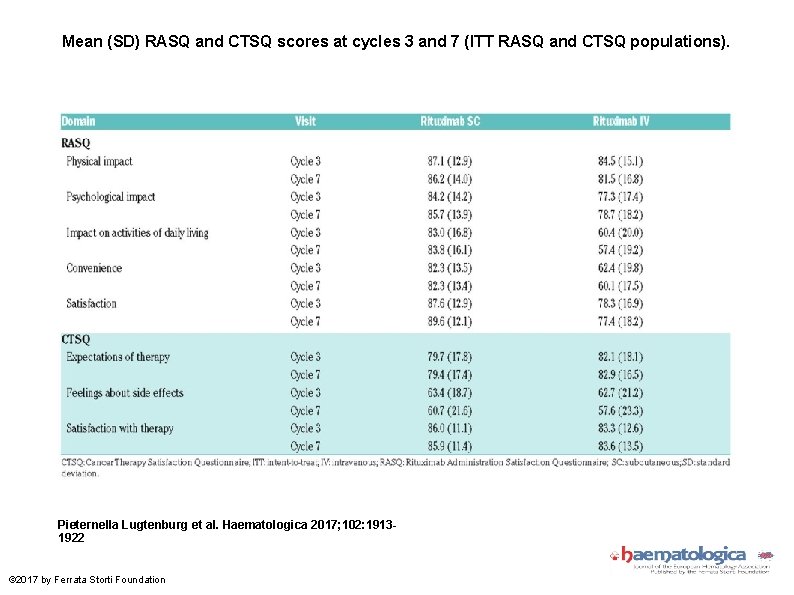
Mean (SD) RASQ and CTSQ scores at cycles 3 and 7 (ITT RASQ and CTSQ populations). Pieternella Lugtenburg et al. Haematologica 2017; 102: 19131922 © 2017 by Ferrata Storti Foundation
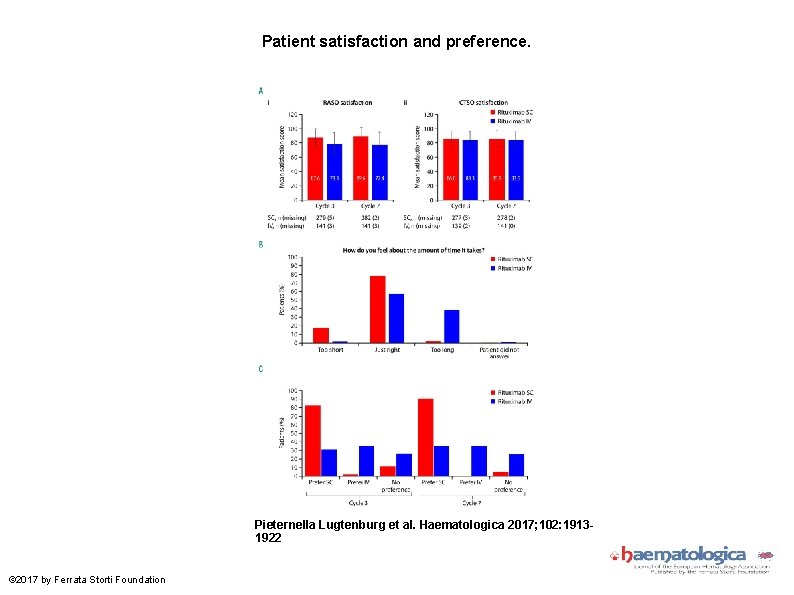
Patient satisfaction and preference. Pieternella Lugtenburg et al. Haematologica 2017; 102: 19131922 © 2017 by Ferrata Storti Foundation
- Slides: 7