Efficacy and safety of guselkumab an antiinterleukin23 monoclonal

Efficacy and safety of guselkumab, an anti-interleukin-23 monoclonal antibody, compared with adalimumab for the continuous treatment of patients with moderate to severe psoriasis: Results from the phase III, double-blinded, placebo- and active comparator–controlled VOYAGE 1 trial Andrew Blauvelt, MD, MBA, Kim A. Papp, MD, Ph. D, Christopher E. M. Griffiths, MD, Bruce Randazzo, MD, Ph. D, Yasmine Wasfi, MD, Ph. D, Yaung-Kaung Shen, Ph. D, Shu Li, Ph. D, Alexa B. Kimball, MPH, MD Journal of the American Academy of Dermatology Volume 76 Issue 3 Pages 405 -417 (March 2017) DOI: 10. 1016/j. jaad. 2016. 11. 041 Copyright © 2016 American Academy of Dermatology, Inc. Terms and Conditions
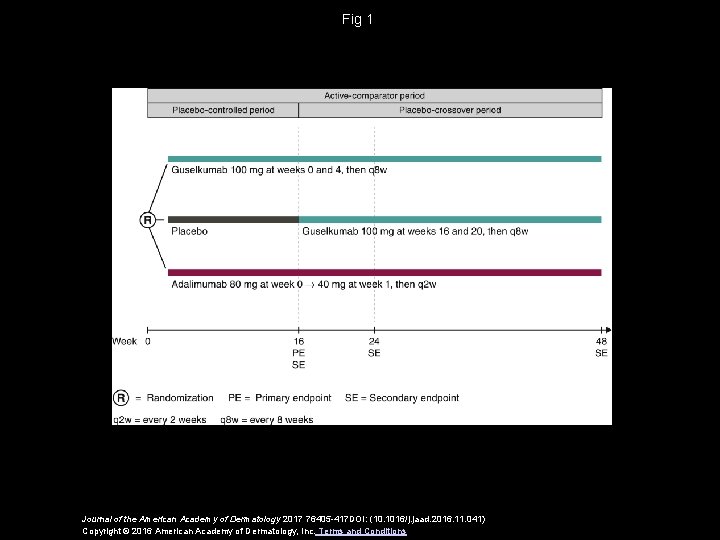
Fig 1 Journal of the American Academy of Dermatology 2017 76405 -417 DOI: (10. 1016/j. jaad. 2016. 11. 041) Copyright © 2016 American Academy of Dermatology, Inc. Terms and Conditions
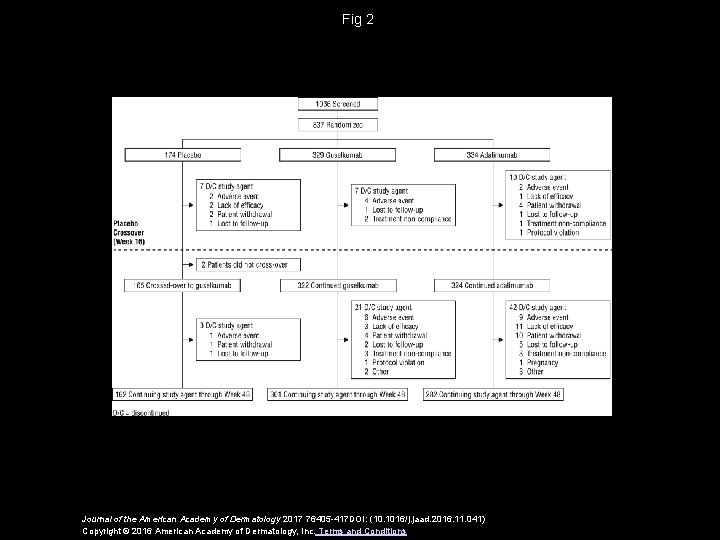
Fig 2 Journal of the American Academy of Dermatology 2017 76405 -417 DOI: (10. 1016/j. jaad. 2016. 11. 041) Copyright © 2016 American Academy of Dermatology, Inc. Terms and Conditions

Fig 3 Journal of the American Academy of Dermatology 2017 76405 -417 DOI: (10. 1016/j. jaad. 2016. 11. 041) Copyright © 2016 American Academy of Dermatology, Inc. Terms and Conditions
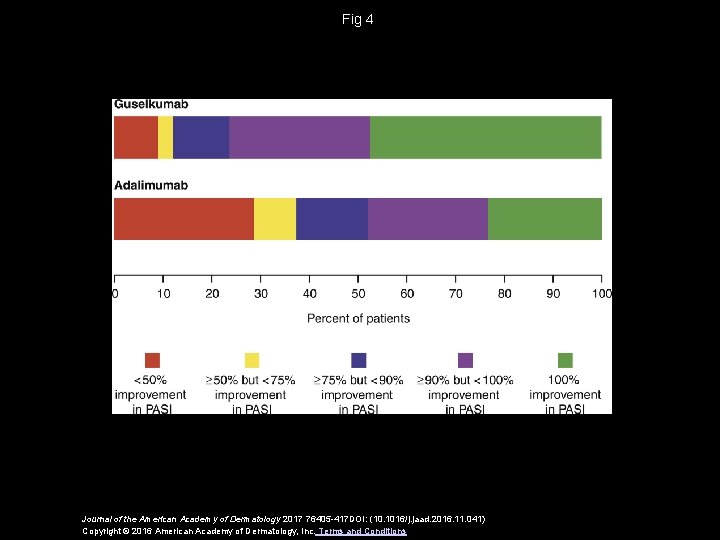
Fig 4 Journal of the American Academy of Dermatology 2017 76405 -417 DOI: (10. 1016/j. jaad. 2016. 11. 041) Copyright © 2016 American Academy of Dermatology, Inc. Terms and Conditions
- Slides: 5