Efficacy and Safety of CarfilzomibPomalidomide Dexamethasone KPd in

Efficacy and Safety of Carfilzomib-Pomalidomide. Dexamethasone (KPd) in Relapsed and/or Refractory Multiple Myeloma: Pooled Analysis of Two Multicenter Single Arm Studies Andrzej Jakubowiak, 1 Jagoda Jasielec, 1 Cara Rosenbaum, 2 Robert Z. Orlowski, 3 Adam Cohen, 4 William Bensinger, 5 Karim Iskander, 6 Khalid Mezzi, 6 Tim Welliver, 6 Akeem Yusuf, 6 Brian Durie 7 1 University of Chicago Medicine, Chicago, IL, USA; 2 Weill Cornell Medical College, New York, NY, USA; 3 The University of Texas MD Anderson Cancer Center, Houston, TX, USA; Cancer Center, University of Pennsylvania, Philadelphia, PA, USA; 5 Swedish Medical Center, Seattle, WA, USA; 6 Amgen Inc. , Thousand Oaks, CA, USA; 7 Cedars. Sinai Medical Center, Samuel Oschin Comprehensive Cancer Institute, Los Angeles, CA, USA 4 Abramson

Disclosure Statement: Andrzej Jakubowiak, MD, Ph. D Consultant: ABBVIE, AMGEN, BMS, GSK, Janssen, Karyopharm, Skyline. Dx, Sanofi, Takeda Speaker’s Bureau: None Grant/Research Support: ABBVIE, AMGEN, BMS, GSK, Janssen, Karyopharm, Sanofi, Takeda Stock Shareholder: None Honoraria: ABBVIE, AMGEN, BMS, GSK, Janssen, Karyopharm, Skyline. Dx, Sanofi, Takeda Employment: None Other: None Discussion of off-label drug use: carfilzomib, elotuzumab, daratumumab, selinexor, ixazomib This study was supported by Amgen Inc. Medical writing support was provided by Jacqueline Sayyah and Shawn Lee, of Amgen Inc. The authors would like to thank Viktor Chirikov and Hrishi Kale from Pharmerit, inc. , and Tara Kervin from SCI, inc. for data analyses

Background • Most multiple myeloma patients receive lenalidomide-based frontline treatment and many progress while on lenalidomide-based therapy • Pomalidomide-based regimens may be an option for multiple myeloma (MM) patients who have been exposed or are refractory to lenalidomide • Carfilzomib-pomalidomide-dexamethasone (KPd) has been shown to be an efficacious and well-tolerated regimen in relapsed and/or refractory MM (RRMM) in two phase 1/2 studies: – NCT 01665794 (MMRC study [n=66])1: A multi-center phase 1/2 MMRC study of RRMM patients with median prior 2 lines of therapy. 91% refractory to lenalidomide, 85% prior IMi. D and PI exposure. ORR was 77% and m. PFS was 16. 8 months – NCT 01464034 (AMy. C study [n=117])2: A multi-center phase 1/2 AMy. C study of RRMM patients with median 5 prior lines of therapy. 100% refractory to lenalidomide, 97% prior IMi. D and PI exposure. ORR was 50% and m. PFS was 9. 7 months • A pooled analysis of individual patient data from these two multicenter trials would provide a more robust assessment of the efficacy and safety of KPd in RRMM, including in several relevant patient subgroups 1 Jakubowiak A, et al. EHA meeting, 2017. 2 Shah J, et al. Blood. 2015; 12: 2284 -2290 MMRC, Mulitple Myeloma Research Consortium; AMy. C , Academic Myeloma Consortium; ORR, overall response rate; m. PFS, median progression free survival

KPd Pooled Analysis Study Design • Patient-level data from two multicenter phase 1/2 studies of KPd were combined (n=183)1, 2 Treatment Schedule Carfilzomib: 20/27 mg/m 2 IV Days 1, 2, 8, 9, 15, 16 Pomalidomide 4 mg Days 1 -21 Dexamethasone 40 mg (cycle 1 -4); 20 mg (cycle >4) Days 1, 8, 15, +/-22 28 -day cycles until PD or unacceptable toxicity 1 Jakubowiak A, et al. EHA meeting, 2017. 2 Shah J, et al. Blood. 2015; 12: 2284 -2290. Primary end point for pooled analysis: • ORR Secondary end points for pooled analysis included: • ≥VGPR rate • PFS • OS • Safety

Study Eligibility • NCT 016657941 and NCT 014640342 included in this analysis were single-arm trials of KPd with dose escalation and dose expansion phases • Patients receiving carfilzomib at 20/27 mg/m 2 as initial planned treatment are included in this analysis 3 Key inclusion criteria 1 Jakubowiak A, et al. EHA meeting, 2017. 2 Shah J, et al. Blood. 2015; 12: 2284 -2290. § § § § RRMM with prior lenalidomide treatment Lenalidomide-refractory disease Measurable disease per IMWG ECOG performance status of 0 to 2 Absolute neutrophil count ≥ 1. 0 x 109/L Hemoglobin ≥ 8. 0 g/d. L Platelet ≥ 75 x 109/L (1); Platelet ≥ 50 x 109/L (2) Cr. Cl ≥ 30 m. L/min (1) ; Cr. Cl ≥ 50 m. L/min(2) 3 Subjects who received at least one dose of study drug were included in the pooled analysis.
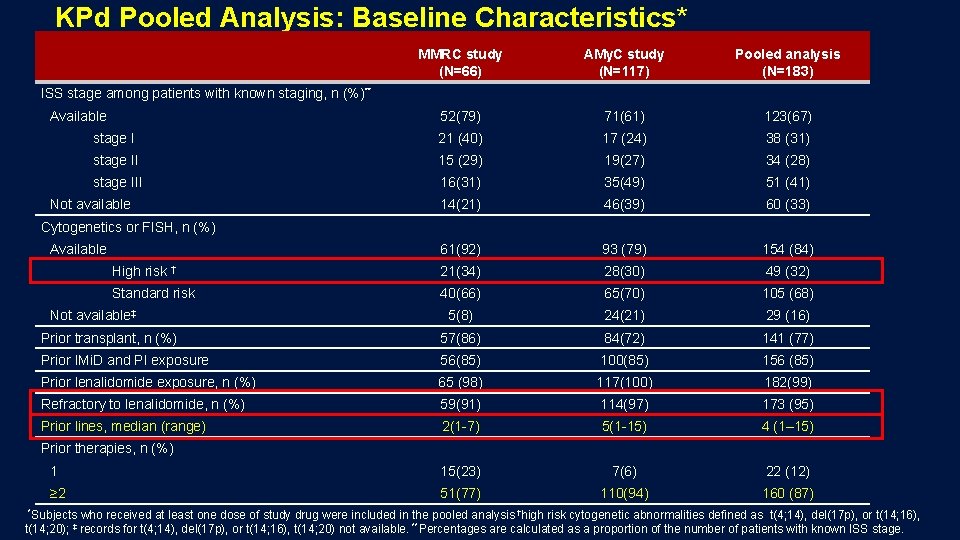
KPd Pooled Analysis: Baseline Characteristics* MMRC study (N=66) AMy. C study (N=117) Pooled analysis (N=183) 52(79) 71(61) 123(67) stage I 21 (40) 17 (24) 38 (31) stage II 15 (29) 19(27) 34 (28) stage III 16(31) 35(49) 51 (41) 14(21) 46(39) 60 (33) 61(92) 93 (79) 154 (84) High risk † 21(34) 28(30) 49 (32) Standard risk 40(66) 65(70) 105 (68) 5(8) 24(21) 29 (16) Prior transplant, n (%) 57(86) 84(72) 141 (77) Prior IMi. D and PI exposure 56(85) 100(85) 156 (85) Prior lenalidomide exposure, n (%) 65 (98) 117(100) 182(99) Refractory to lenalidomide, n (%) 59(91) 114(97) 173 (95) Prior lines, median (range) 2(1 -7) 5(1 -15) 4 (1– 15) 1 15(23) 7(6) 22 (12) ≥ 2 51(77) 110(94) 160 (87) ISS stage among patients with known staging, n (%)** Available Not available Cytogenetics or FISH, n (%) Available Not available‡ Prior therapies, n (%) *Subjects who received at least one dose of study drug were included in the pooled analysis †high risk cytogenetic abnormalities defined as t(4; 14), del(17 p), or t(14; 16), t(14; 20); ‡ records for t(4; 14), del(17 p), or t(14; 16), t(14; 20) not available. **Percentages are calculated as a proportion of the number of patients with known ISS stage.
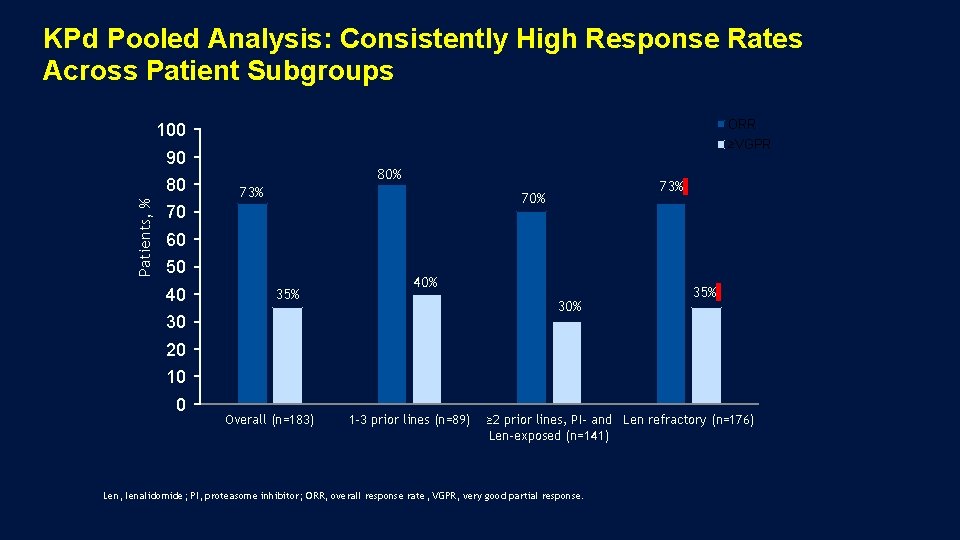
KPd Pooled Analysis: Consistently High Response Rates Across Patient Subgroups ORR 100 ≥VGPR 90 Patients, % 80 80% 73% 70 60 50 40 35% 40% 30 35% 20 10 0 Overall (n=183) 1 -3 prior lines (n=89) ≥ 2 prior lines, PI- and Len refractory (n=176) Len-exposed (n=141) Len, lenalidomide; PI, proteasome inhibitor; ORR, overall response rate, VGPR, very good partial response.
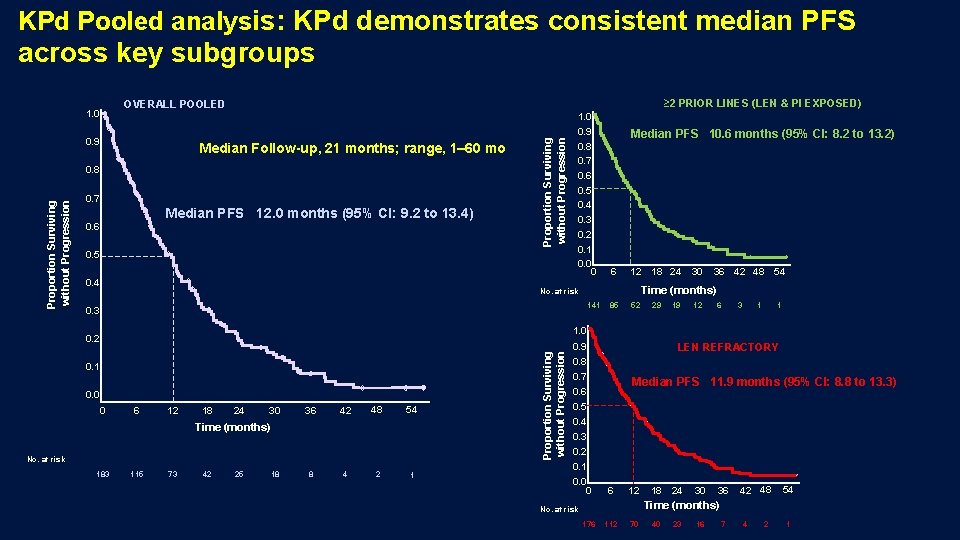
KPd Pooled analysis: KPd demonstrates consistent median PFS across key subgroups ≥ 2 PRIOR LINES (LEN & PI EXPOSED) 0. 9 Median Follow-up, 21 months; range, 1– 60 mo Proportion Surviving without Progression 0. 8 0. 7 Median PFS 12. 0 months (95% CI: 9. 2 to 13. 4) 0. 6 Proportion Surviving without Progression OVERALL POOLED 1. 0 0. 5 0. 4 1. 0 0. 9 0. 8 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 0. 1 0. 0 0 Median PFS 10. 6 months (95% CI: 8. 2 to 13. 2) 6 12 18 24 85 52 30 36 42 48 54 Time (months) No. at risk 141 0. 3 29 19 12 6 3 1 1 1. 0 0. 1 0. 0 0 6 12 18 24 30 36 42 48 54 Time (months) No. at risk 183 115 73 42 25 18 8 4 2 1 Proportion Surviving without Progression 0. 2 0. 9 LEN REFRACTORY 0. 8 0. 7 Median PFS 11. 9 months (95% CI: 8. 8 to 13. 3) 0. 6 0. 5 0. 4 0. 3 0. 2 0. 1 0. 0 0 6 12 18 24 30 36 42 48 54 Time (months) No. at risk 176 112 70 40 23 16 7 4 2 1

KPd Pooled analysis: KPd demonstrates consistent median OS across key subgroups ≥ 2 PRIOR LINES (LEN & PI EXPOSED) 1. 0 OVERALL POOLED 1. 0 0. 9 Median OS 29. 6 months (95% CI: 26. 3 to 34. 0) Proportion Surviving 0. 8 0. 7 Proportion Surviving 0. 9 Median OS 28. 6 months (95% CI: 23. 6 to 33. 2) 0. 8 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 0. 1 0. 0 0. 6 0 6 12 24 30 36 42 48 54 14 7 4 Time (months) No. at risk 0. 5 18 141 128 104 82 69 43 28 0. 4 1. 0 0. 2 0. 1 0. 0 0 6 12 18 24 30 36 42 48 54 Time (months) Proportion Surviving 0. 3 0. 8 0. 6 0. 5 0. 4 0. 3 0. 2 0. 0 167 140 114 96 61 Median OS 29. 6 months (95% CI: 25. 9 to 34. 0) 0. 7 0. 1 No. at risk 183 LEN REFRACTORY 0. 9 41 22 10 0 6 12 18 161 135 110 6 No. at risk 176 24 30 36 42 48 54 22 10 6 Time (months) 93 59 39

KPd pooled analysis : Adverse Events Summary Category TEAS, n (%) Any grade AE Grade 3 -4 AE Leading to treatment discontinuation Grade 5 AEs* Hematologic grade 3 -4 TEAEs in >10% of patients, n (%) Anemia Thrombocytopenia Neutropenia Non-hematologic grade 3 -4 TEAEs in ≥ 5% of patients , n (%) Fatigue Pneumonia Hypercalcemia Dyspnea Grade 3 -4 TEAEs of clinical interest, n (%) Hypertension Congestive cardiac failure Peripheral neuropathy Renal failure *Grade 5 AEs: General (4); Vascular (1); Infections (2); Neoplasms (1). (n=183) 183 (100) 136 (74) 18 (28) 8 (4) 31 (17) 20 (11) 50 (27) 11 (6) 28 (15) 19 (10) 10 (5) 8 (4) 4 (2) 1 (1) 0 (0)
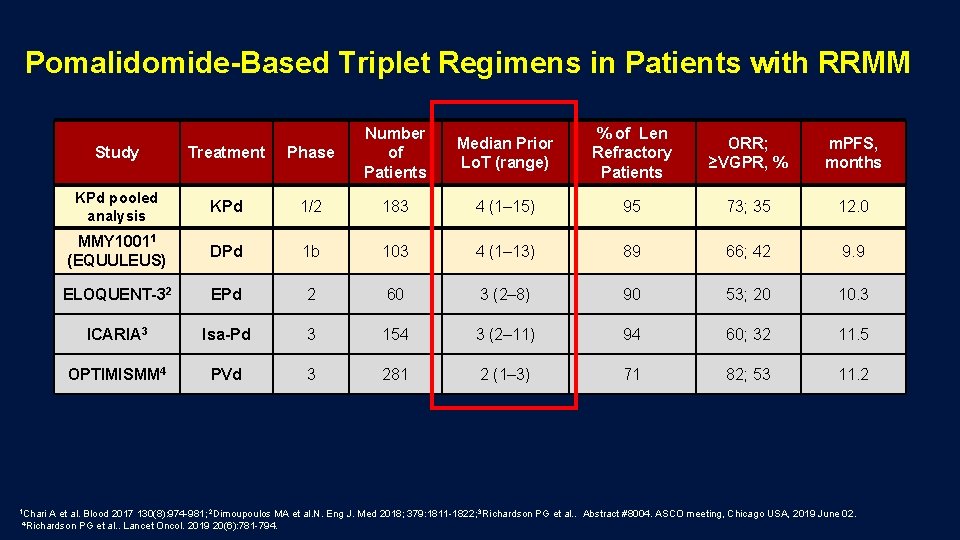
Pomalidomide-Based Triplet Regimens in Patients with RRMM 1 Chari Study Treatment Phase Number of Patients Median Prior Lo. T (range) % of Len Refractory Patients ORR; ≥VGPR, % m. PFS, months KPd pooled analysis KPd 1/2 183 4 (1– 15) 95 73; 35 12. 0 MMY 10011 (EQUULEUS) DPd 1 b 103 4 (1– 13) 89 66; 42 9. 9 ELOQUENT-32 EPd 2 60 3 (2– 8) 90 53; 20 10. 3 ICARIA 3 Isa-Pd 3 154 3 (2– 11) 94 60; 32 11. 5 OPTIMISMM 4 PVd 3 281 2 (1– 3) 71 82; 53 11. 2 A et al. Blood 2017 130(8): 974 -981; 2 Dimoupoulos MA et al. N. Eng J. Med 2018; 379: 1811 -1822; 3 Richardson PG et al. . Abstract #8004. ASCO meeting, Chicago USA, 2019 June 02. PG et al. . Lancet Oncol. 2019 20(6): 781 -794. 4 Richardson

Conclusions • Based on this analysis of pooled data from 2 multi-center trials, KPd regimen shows – ORR 73%, VGPR 35% – PFS 12. 0 mo, OS, 29. 6 mo in patient population 95% refractory to lenalidomide and median of 4 lines of therapy • Toxicities are manageable and consistent with toxicities in other carfilzomib and pomalidomide studies • With all reservations to pooled analysis and cross-study comparisons, KPd appears as one of the most effective treatment options for lenalidomide refractory patients
- Slides: 12