Effects of Oxygen on the Activation of Methane







- Slides: 28

Effects of Oxygen on the Activation of Methane over Pt Surfaces and Clusters. Corneliu Buda, 1 Matthew Neurock, 1 Cathy Chin 2 and Enrique Iglesia 2 1 Department of Chemical Engineering, University of Virginia, Charlottesville, VA. 2 Department of Chemical Engineering, University of California, Berkeley, CA. January 14 th 2009

Goals and Methods ØElucidate the mechanisms that control the activation of methane and establish the kinetics over different metal surfaces and clusters. Ø Explore surface morphology and the influence of oxygen coverage over methane first step dissociation. Ø All calculations performed using plane wave density functional theory enclosed by VASP package.

Previous Results

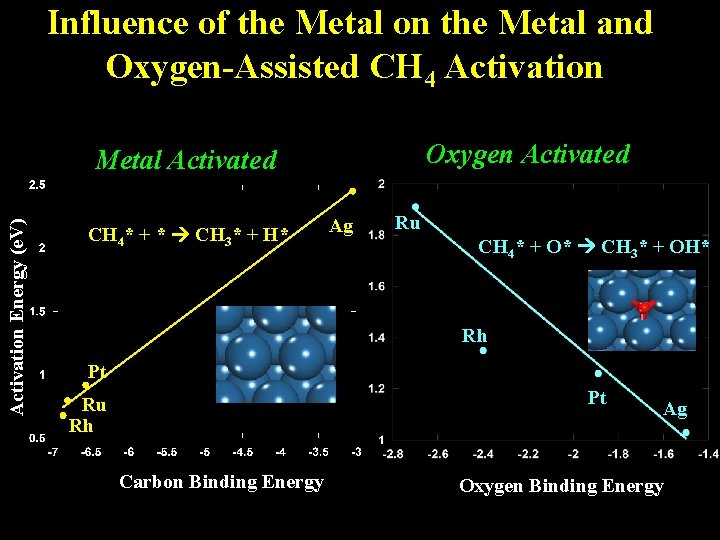
Influence of the Metal on the Metal and Oxygen-Assisted CH 4 Activation Oxygen Activated Activation Energy (e. V) Metal Activated CH 4* + * CH 3* + H* Ag Ru CH 4* + O* CH 3* + OH* Rh Pt Pt Ru Rh Carbon Binding Energy Ag Oxygen Binding Energy

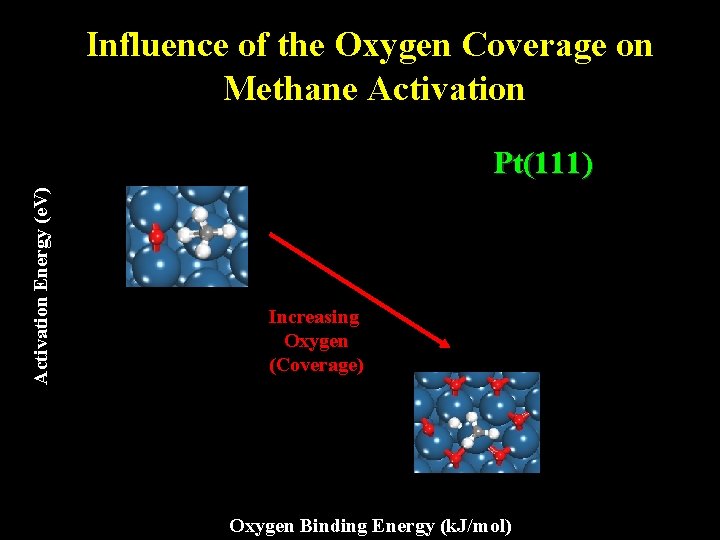
Influence of the Oxygen Coverage on Methane Activation Energy (e. V) Pt(111) Increasing Oxygen (Coverage) Oxygen Binding Energy (k. J/mol)

Barriers for O 2 Dissociation and Recombination Ea (k. J/mol) Pt(111) 2 O* O 2* 2 O* Coverage (ML)

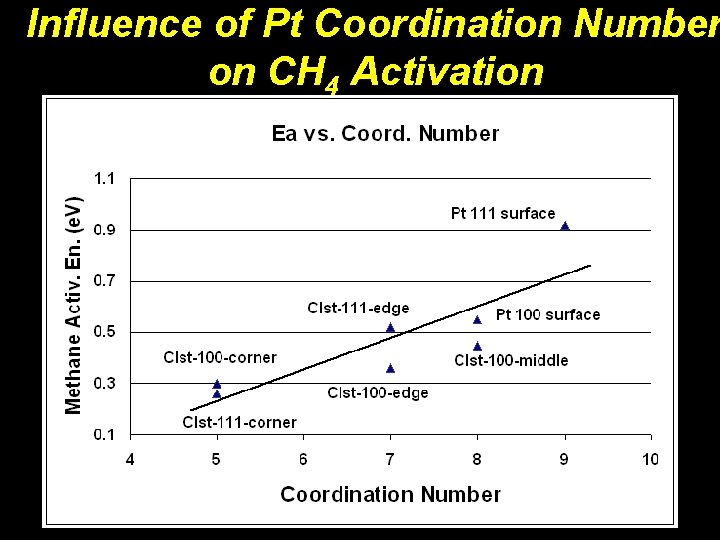
Influence of Pt Coordination Number on CH 4 Activation

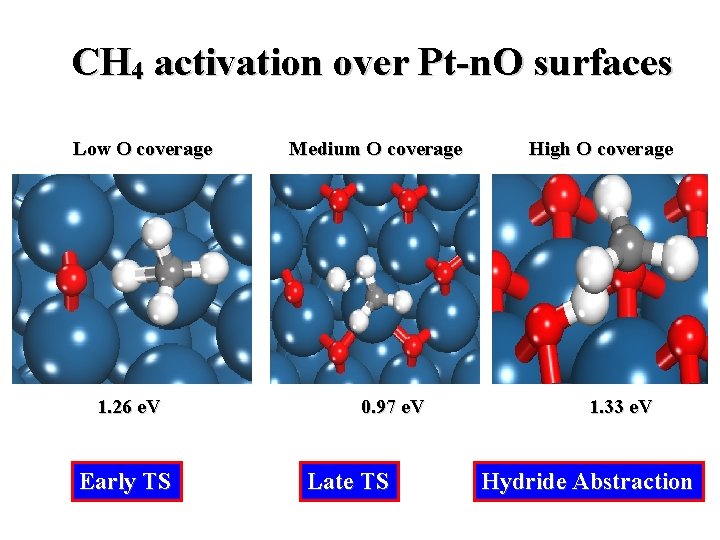
CH 4 activation over Pt-n. O surfaces Low O coverage 1. 26 e. V Early TS Medium O coverage 0. 97 e. V Late TS High O coverage 1. 33 e. V Hydride Abstraction

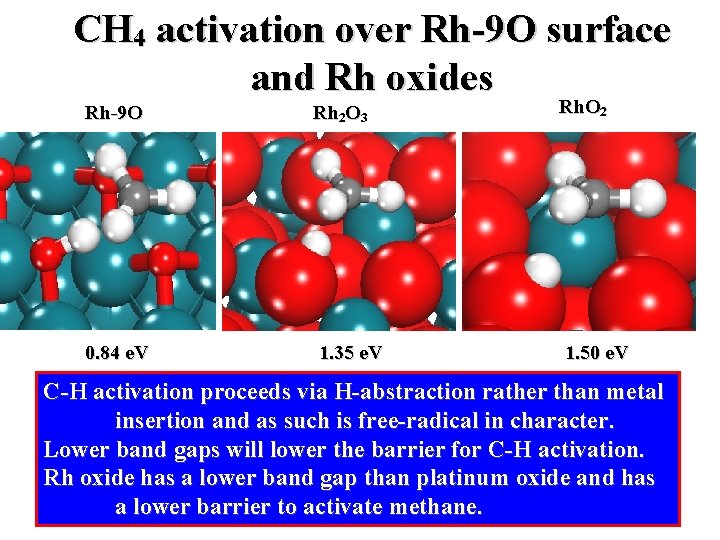
CH 4 activation over Rh-9 O surface and Rh oxides Rh-9 O Rh 2 O 3 Rh. O 2 0. 84 e. V 1. 35 e. V 1. 50 e. V C-H activation proceeds via H-abstraction rather than metal insertion and as such is free-radical in character. Lower band gaps will lower the barrier for C-H activation. Rh oxide has a lower band gap than platinum oxide and has a lower barrier to activate methane.

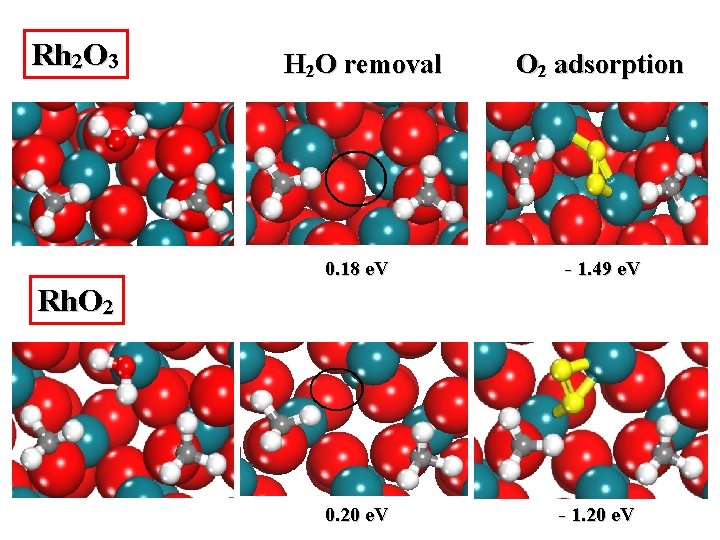
Rh 2 O 3 H 2 O removal O 2 adsorption 0. 18 e. V - 1. 49 e. V 0. 20 e. V - 1. 20 e. V Rh. O 2

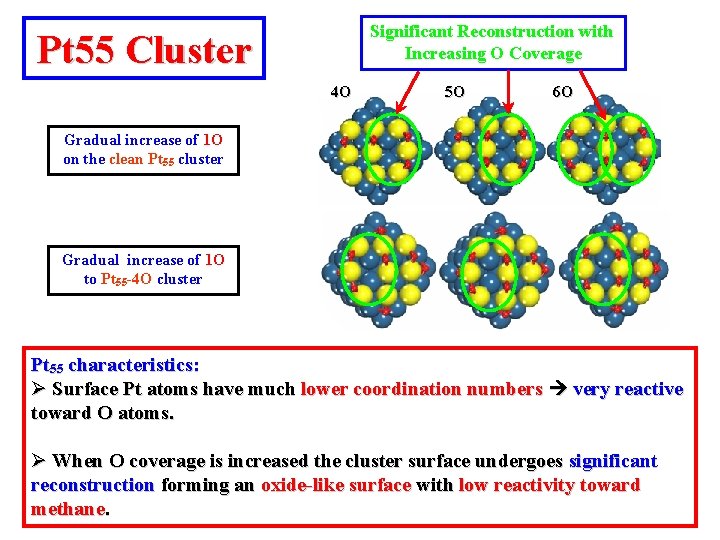
Significant Reconstruction with Increasing O Coverage Pt 55 Cluster 4 O 5 O 6 O Gradual increase of 1 O on the clean Pt 55 cluster Gradual increase of 1 O to Pt 55 -4 O cluster Pt 55 characteristics: Ø Surface Pt atoms have much lower coordination numbers very reactive toward O atoms. Ø When O coverage is increased the cluster surface undergoes significant reconstruction forming an oxide-like surface with low reactivity toward methane.

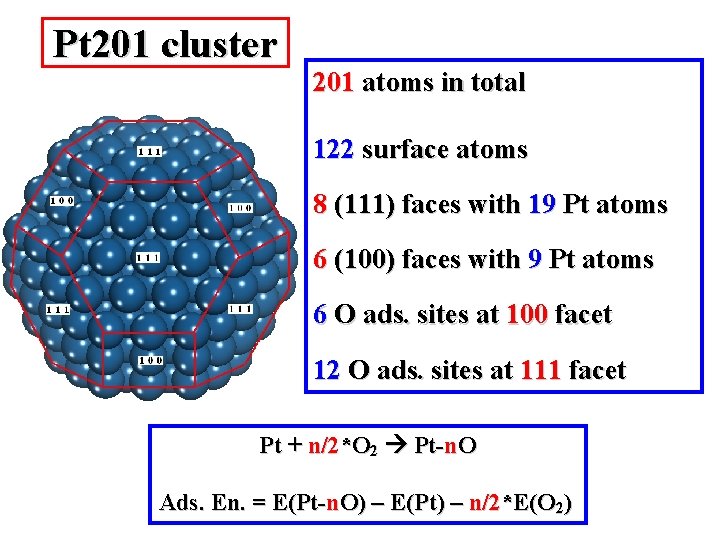
Pt 201 cluster 201 atoms in total 122 surface atoms 8 (111) faces with 19 Pt atoms 6 (100) faces with 9 Pt atoms 6 O ads. sites at 100 facet 12 O ads. sites at 111 facet Pt + n/2*O 2 Pt-n. O Ads. En. = E(Pt-n. O) – E(Pt) – n/2*E(O 2)

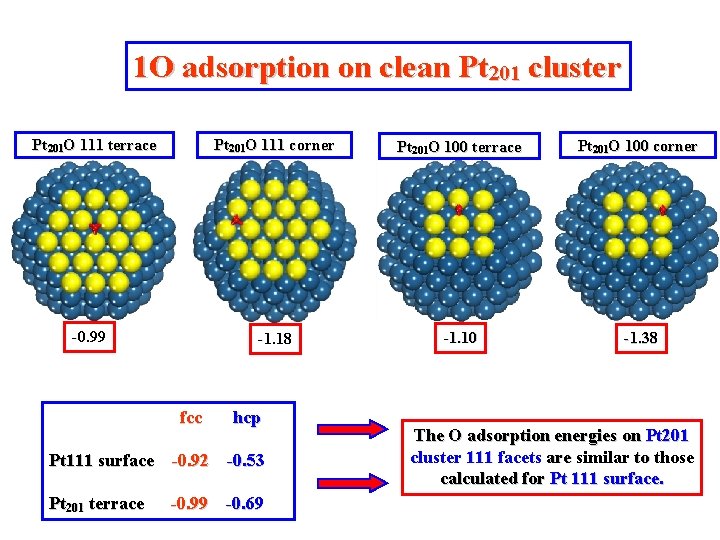
1 O adsorption on clean Pt 201 cluster Pt 201 O 111 terrace -0. 99 fcc Pt 201 O 111 corner Pt 201 O 100 terrace Pt 201 O 100 corner -1. 18 -1. 10 -1. 38 hcp Pt 111 surface -0. 92 -0. 53 Pt 201 terrace -0. 99 -0. 69 The O adsorption energies on Pt 201 cluster 111 facets are similar to those calculated for Pt 111 surface.

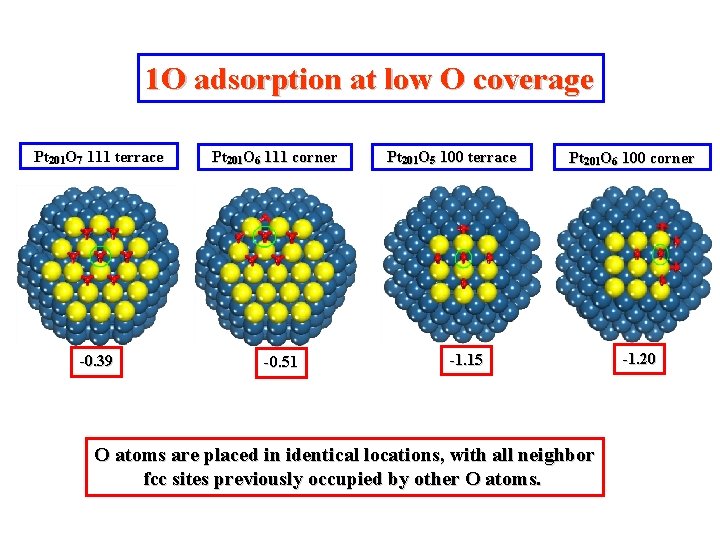
1 O adsorption at low O coverage Pt 201 O 7 111 terrace -0. 39 Pt 201 O 6 111 corner -0. 51 Pt 201 O 5 100 terrace Pt 201 O 6 100 corner -1. 15 O atoms are placed in identical locations, with all neighbor fcc sites previously occupied by other O atoms. -1. 20

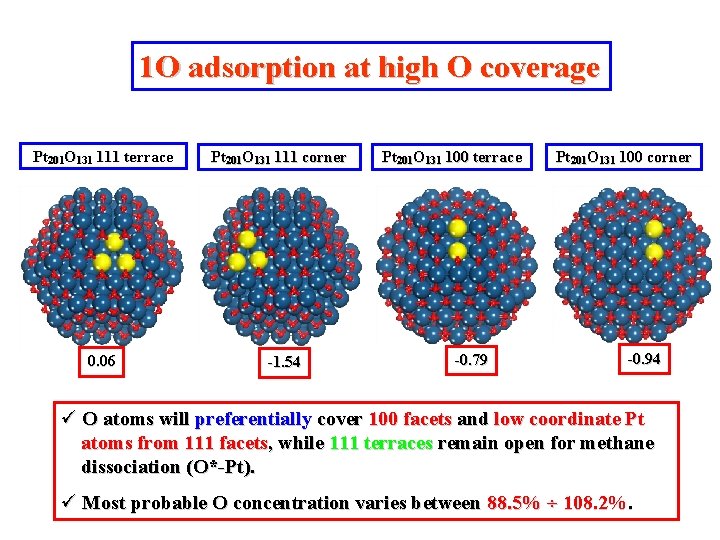
1 O adsorption at high O coverage Pt 201 O 131 111 terrace 0. 06 Pt 201 O 131 111 corner -1. 54 Pt 201 O 131 100 terrace -0. 79 Pt 201 O 131 100 corner -0. 94 ü O atoms will preferentially cover 100 facets and low coordinate Pt atoms from 111 facets, while 111 terraces remain open for methane dissociation (O*-Pt). ü Most probable O concentration varies between 88. 5% ÷ 108. 2%.

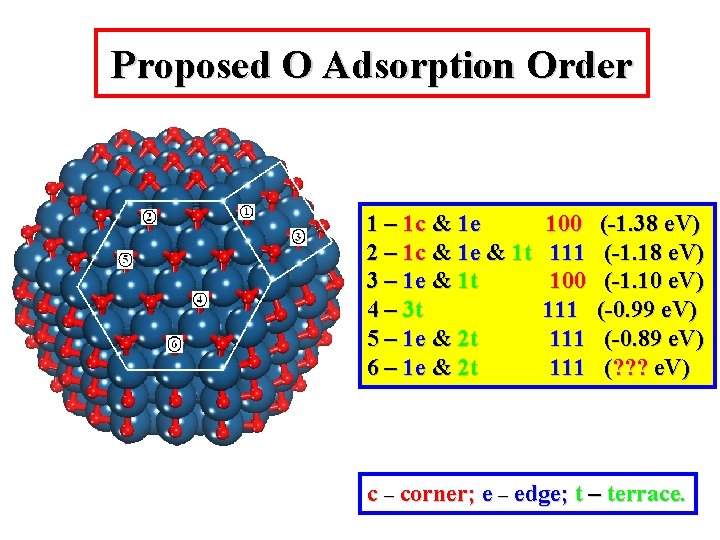
Proposed O Adsorption Order 1 – 1 c & 1 e 2 – 1 c & 1 e & 1 t 3 – 1 e & 1 t 4 – 3 t 5 – 1 e & 2 t 6 – 1 e & 2 t 100 111 111 (-1. 38 e. V) (-1. 10 e. V) (-0. 99 e. V) (-0. 89 e. V) (? ? ? e. V) c – corner; e – edge; t – terrace.

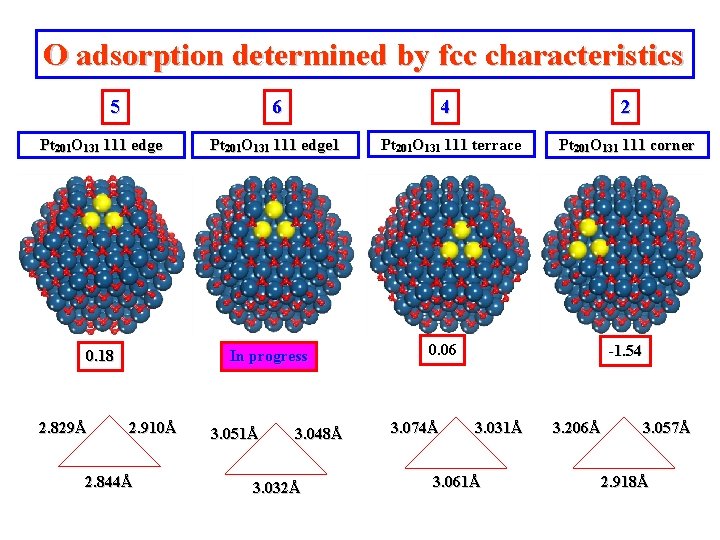
O adsorption determined by fcc characteristics 5 6 Pt 201 O 131 111 edge 0. 18 2. 829Å 4 Pt 201 O 131 111 edge 1 In progress 2. 910Å 2. 844Å 3. 051Å 3. 048Å 3. 032Å 2 Pt 201 O 131 111 terrace Pt 201 O 131 111 corner 0. 06 3. 074Å -1. 54 3. 031Å 3. 061Å 3. 206Å 3. 057Å 2. 918Å

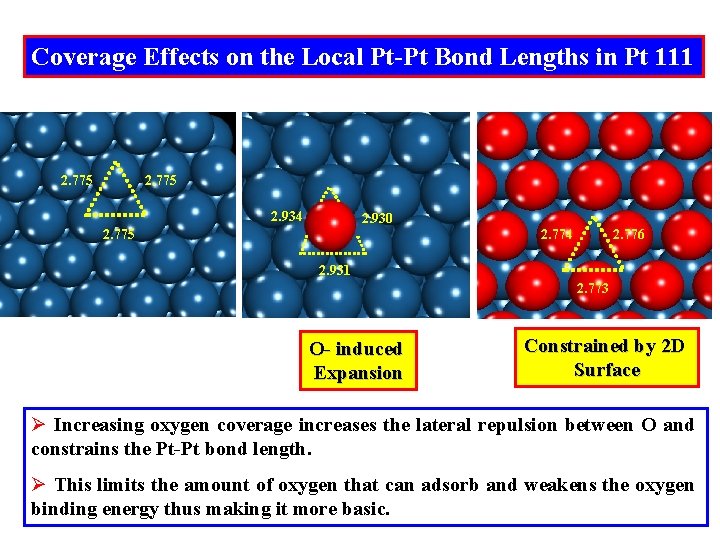
Coverage Effects on the Local Pt-Pt Bond Lengths in Pt 111 2. 775 2. 934 2. 930 2. 775 2. 774 2. 776 2. 931 2. 773 O- induced Expansion Constrained by 2 D Surface Ø Increasing oxygen coverage increases the lateral repulsion between O and constrains the Pt-Pt bond length. Ø This limits the amount of oxygen that can adsorb and weakens the oxygen binding energy thus making it more basic.

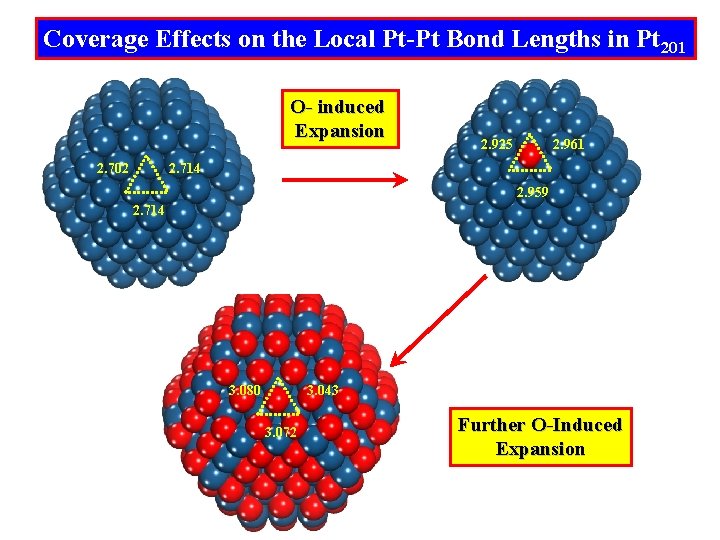
Coverage Effects on the Local Pt-Pt Bond Lengths in Pt 201 O- induced Expansion 2. 702 2. 925 2. 961 2. 714 2. 959 2. 714 3. 080 3. 043 3. 072 Further O-Induced Expansion

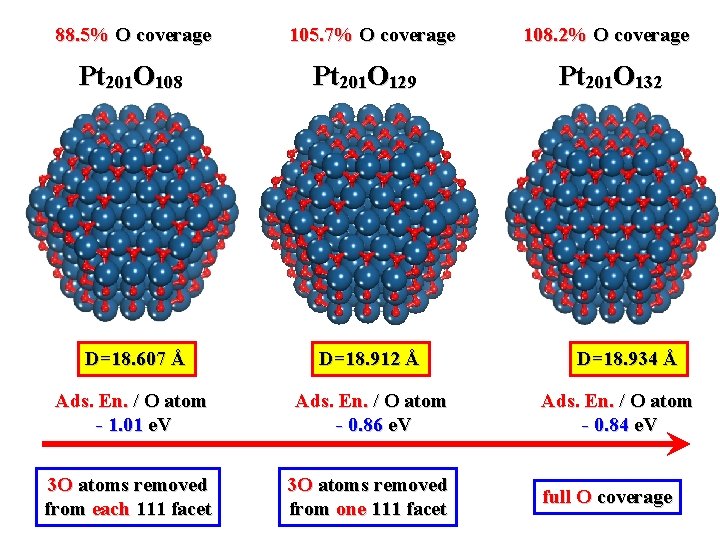
88. 5% O coverage 105. 7% O coverage Pt 201 O 108 Pt 201 O 129 D=18. 607 Å D=18. 912 Å Ads. En. / O atom - 1. 01 e. V Ads. En. / O atom - 0. 86 e. V 3 O atoms removed from each 111 facet 3 O atoms removed from one 111 facet 108. 2% O coverage Pt 201 O 132 D=18. 934 Å Ads. En. / O atom - 0. 84 e. V full O coverage

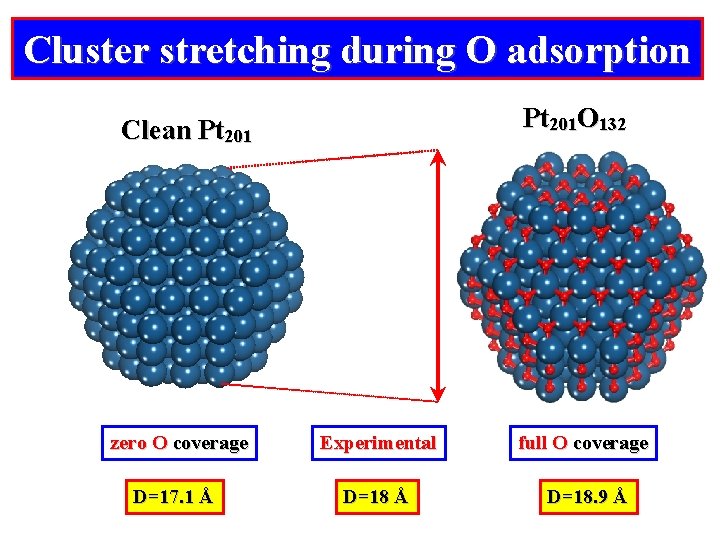
Cluster stretching during O adsorption Pt 201 O 132 Clean Pt 201 zero O coverage Experimental full O coverage D=17. 1 Å D=18. 9 Å

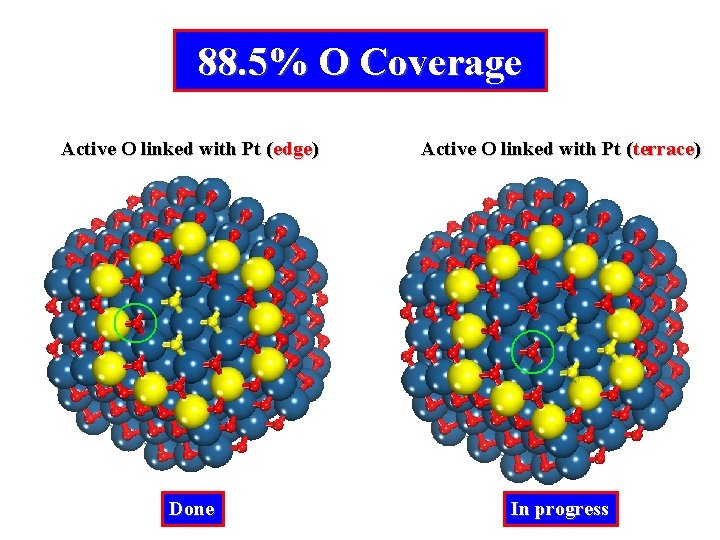
88. 5% O Coverage Active O linked with Pt (edge) Active O linked with Pt (terrace) Done In progress

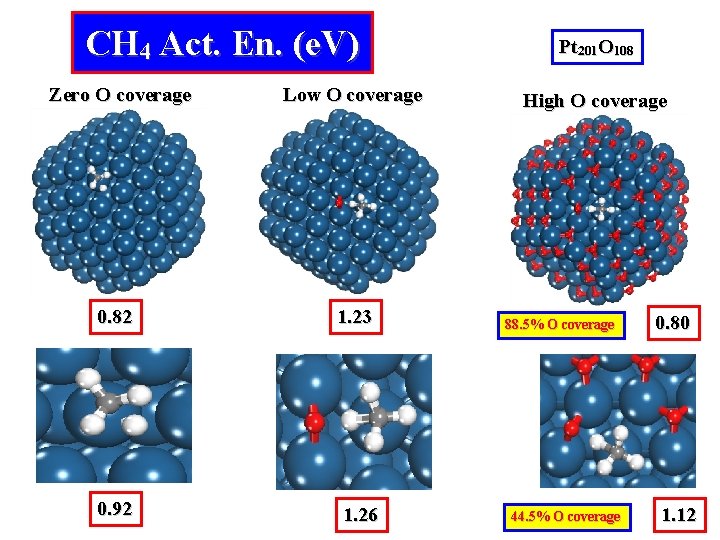
CH 4 Act. En. (e. V) Zero O coverage Low O coverage 0. 82 1. 23 0. 92 1. 26 Pt 201 O 108 High O coverage 88. 5% O coverage 44. 5% O coverage 0. 80 1. 12

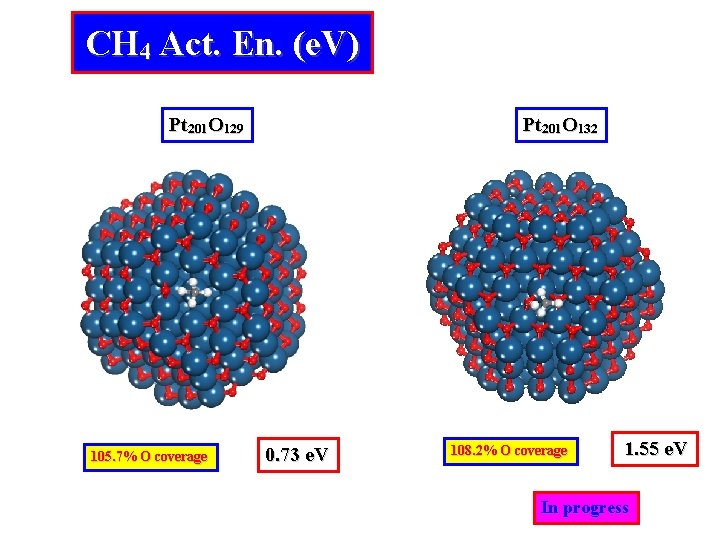
CH 4 Act. En. (e. V) Pt 201 O 129 105. 7% O coverage Pt 201 O 132 0. 73 e. V 108. 2% O coverage 1. 55 e. V In progress

Summary Ø O adsorption energies to the central atoms in the 111 terrace of Pt 201 cluster are quite similar with Pt 111 surface. Ø Pt 201 cluster elongates when O is adsorbed, but the surface maintains its initial configuration, while for smaller cluster as Pt 55 it begin to take on some surface oxide-like characteristics. Ø Methane activation energy has a lower values on the Pt 201 cluster at high O coverage than on clean cluster.

Future Interest

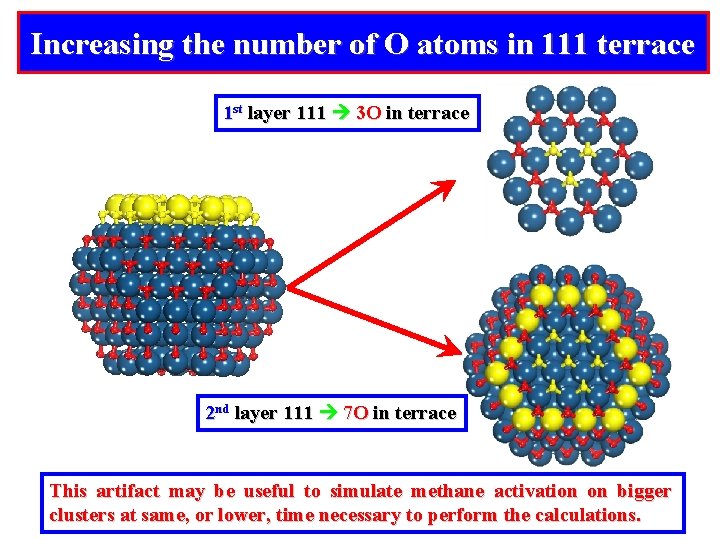
Increasing the number of O atoms in 111 terrace 1 st layer 111 3 O in terrace 2 nd layer 111 7 O in terrace This artifact may be useful to simulate methane activation on bigger clusters at same, or lower, time necessary to perform the calculations.

Acknowledgements BP Methane Conversion Cooperative Program