Effects of an AngiotensinConverting Enzyme Inhibitor Ramipril on

Effects of an Angiotensin-Converting Enzyme Inhibitor, Ramipril, on Death from Cardiovascular Causes, Myocardial Infarction, and Stroke in High-Risk Patients The Heart Outcomes Prevention Evaluation (HOPE) Study Investigators N Engl J Med, January 20, 2000

HOPE - Background • ACEIs improve the outcome in patients with LV dysfunction, whether or not they have symptomatic heart failure. • This study assessed the role of an ACEI, ramipril, in patients who were at high risk for cardiovascular events but who did not have LV dysfunction or heart failure. N Engl J Med, January 20, 2000

HOPE - Design • A total of 9, 297 high-risk patients, > 55 years old, who had evidence of vascular disease or diabetes plus one other cardiovascular risk factor and who were not known to have a low EF or heart failure were randomly assigned to receive ramipril (10 mg per day) or matching placebo for a mean of 5 years. • The primary outcome was a composite of MI, stroke or death from cardiovascular causes. Each of these endpoints were also analyzed separately. • Secondary endpoints were death from any cause, the need for revascularization, hospitalization for unstable angina or heart failure, and complications related to diabetes. N Engl J Med, January 20, 2000
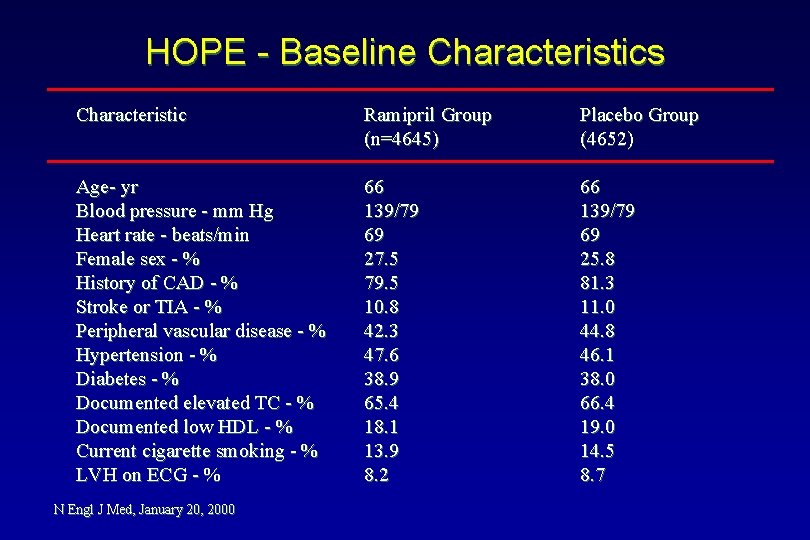
HOPE - Baseline Characteristics Characteristic Ramipril Group (n=4645) Placebo Group (4652) Age- yr Blood pressure - mm Hg Heart rate - beats/min Female sex - % History of CAD - % Stroke or TIA - % Peripheral vascular disease - % Hypertension - % Diabetes - % Documented elevated TC - % Documented low HDL - % Current cigarette smoking - % LVH on ECG - % 66 139/79 69 27. 5 79. 5 10. 8 42. 3 47. 6 38. 9 65. 4 18. 1 13. 9 8. 2 66 139/79 69 25. 8 81. 3 11. 0 44. 8 46. 1 38. 0 66. 4 19. 0 14. 5 8. 7 N Engl J Med, January 20, 2000
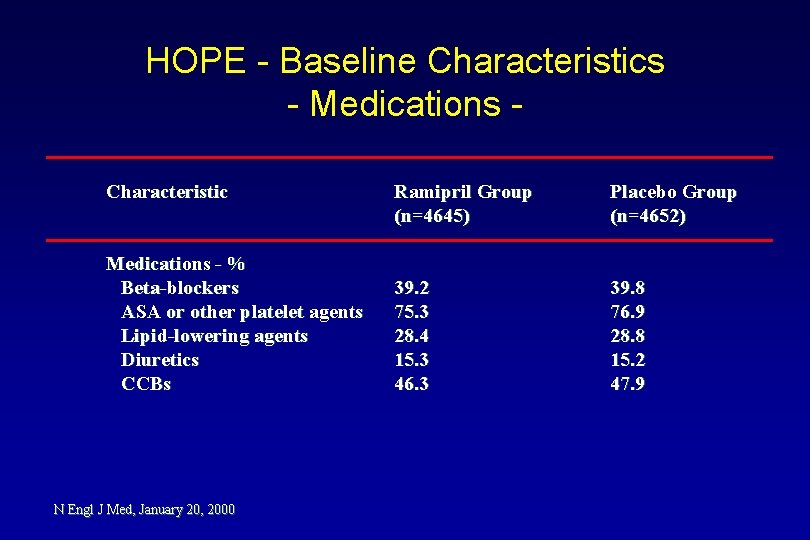
HOPE - Baseline Characteristics - Medications Characteristic Medications - % Beta-blockers ASA or other platelet agents Lipid-lowering agents Diuretics CCBs N Engl J Med, January 20, 2000 Ramipril Group (n=4645) Placebo Group (n=4652) 39. 2 75. 3 28. 4 15. 3 46. 3 39. 8 76. 9 28. 8 15. 2 47. 9

HOPE - Compliance • More patients in the ramipril group versus the placebo group stopped treatment because of cough (7. 3% versus 1. 8%) or hypotension or dizziness (1. 9% versus 1. 5%) • By contrast, more patients in the placebo group than in the ramipril group stopped treatment because of uncontrolled hypertension (3. 9% versus 2. 3%) or because of a clinical event - a primary or secondary outcome (9. 0% versus 6. 7%) • 5. 3% of patients in the ramipril group and 7. 2% of patients in the placebo group received open label ACEI for heart failure N Engl J Med, January 20, 2000
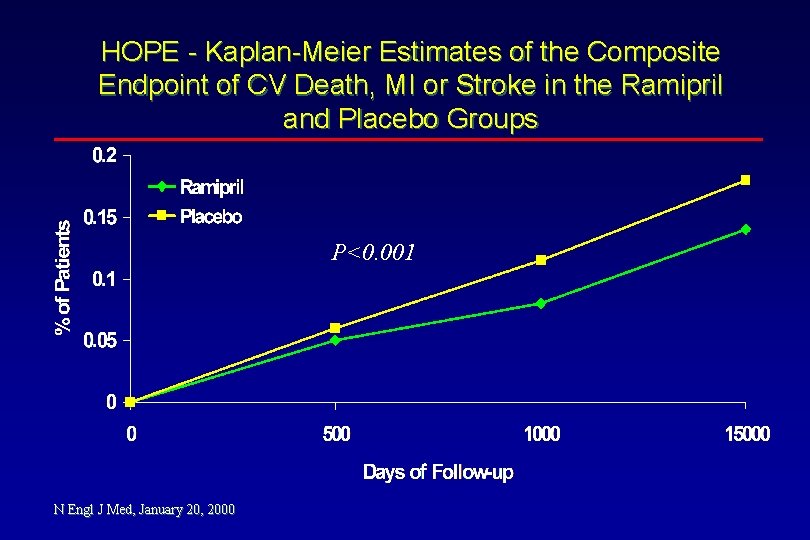
HOPE - Kaplan-Meier Estimates of the Composite Endpoint of CV Death, MI or Stroke in the Ramipril and Placebo Groups P<0. 001 N Engl J Med, January 20, 2000
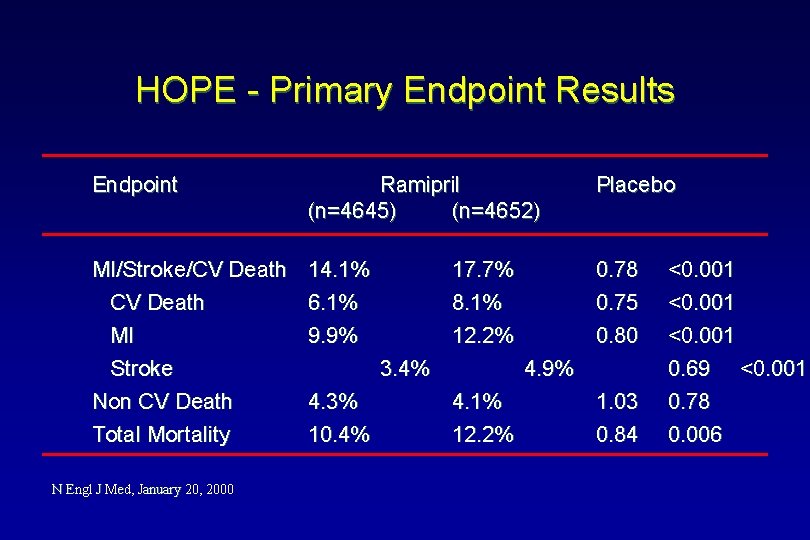
HOPE - Primary Endpoint Results Endpoint Ramipril (n=4645) (n=4652) Placebo MI/Stroke/CV Death MI Stroke Non CV Death Total Mortality 14. 1% 6. 1% 9. 9% 0. 78 0. 75 0. 80 N Engl J Med, January 20, 2000 17. 7% 8. 1% 12. 2% 3. 4% 4. 3% 10. 4% 4. 9% 4. 1% 12. 2% 1. 03 0. 84 <0. 001 0. 69 <0. 001 0. 78 0. 006
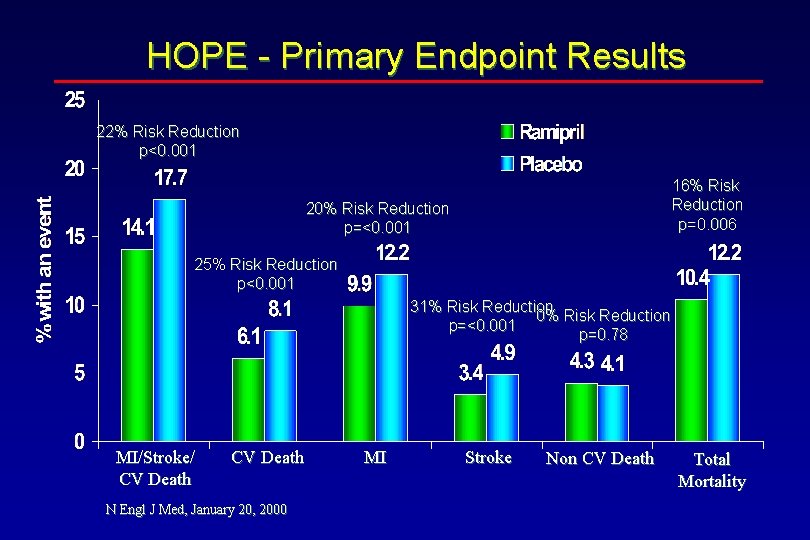
HOPE - Primary Endpoint Results 22% Risk Reduction p<0. 001 16% Risk Reduction p=0. 006 20% Risk Reduction p=<0. 001 25% Risk Reduction p<0. 001 31% Risk Reduction 0% Risk Reduction p=<0. 001 p=0. 78 MI/Stroke/ CV Death N Engl J Med, January 20, 2000 MI Stroke Non CV Death Total Mortality

HOPE - Secondary and Other Endpoint Results Endpoint value Ramipril (n=4645) Placebo RR P (n=46532) Secondary Outcomes - % Revascularization 16. 0 18. 6 <0. 001 Hospitalization for UA 12. 2 12. 4 0. 98 Complications/DM 6. 2 7. 4 <0. 03 Hospitalization for HF 3. 3 3. 8 0. 87 Other Outcomes - % Heart failure 9. 2 11. 7 0. 77 Cardiac arrest 0. 8 1. 2 0. 63 Worsening angina 23. 8 26. 2 0. 003 UA = unstable angina; DM = diabetes mellitus; HF = heart failure New diagnosis of DM 3. 7 5. 3 0. 68 N Engl J Med, January 20, 2000 Unstable angina with ECG changes 3. 9 4. 0 0. 84 0. 68 0. 84 0. 19 <0. 001 0. 03 0. 89 0. 002 0. 96
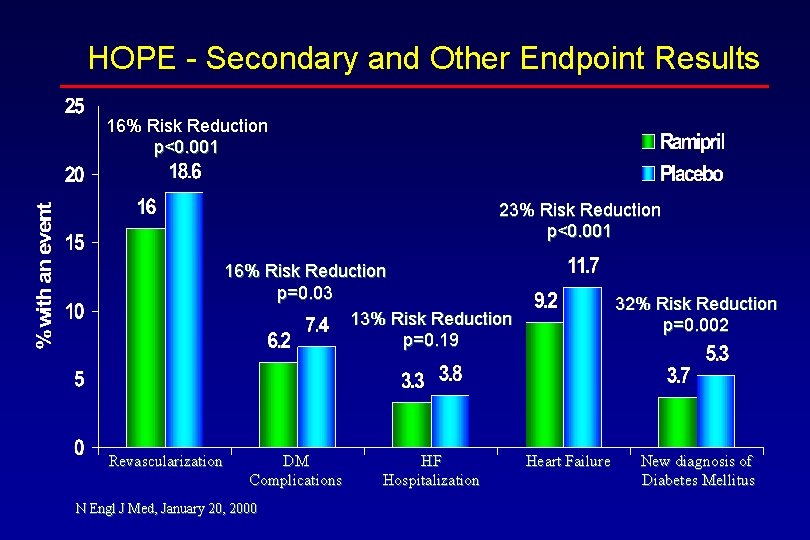
HOPE - Secondary and Other Endpoint Results 16% Risk Reduction p<0. 001 23% Risk Reduction p<0. 001 16% Risk Reduction p=0. 03 13% Risk Reduction p=0. 19 Revascularization DM Complications N Engl J Med, January 20, 2000 HF Hospitalization 32% Risk Reduction p=0. 002 Heart Failure New diagnosis of Diabetes Mellitus
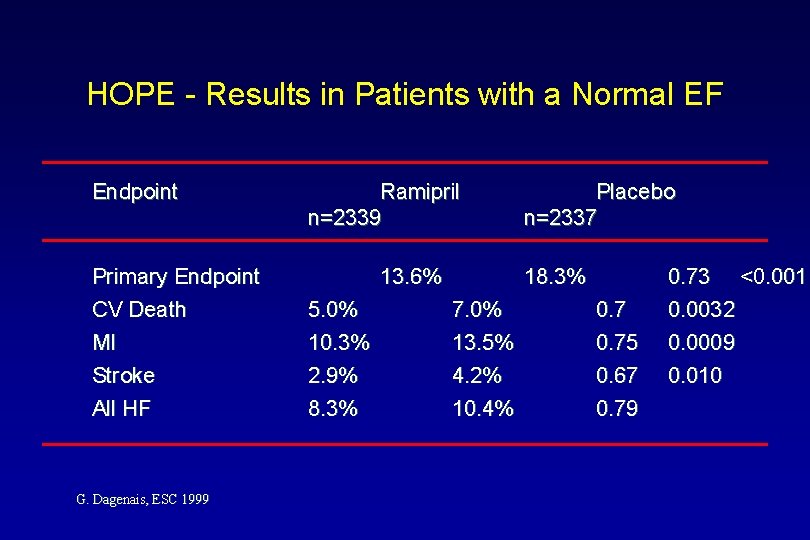
HOPE - Results in Patients with a Normal EF Endpoint Primary Endpoint CV Death MI Stroke All HF G. Dagenais, ESC 1999 Ramipril n=2339 13. 6% 5. 0% 10. 3% 2. 9% 8. 3% Placebo n=2337 18. 3% 7. 0% 13. 5% 4. 2% 10. 4% 0. 75 0. 67 0. 79 0. 73 <0. 001 0. 0032 0. 0009 0. 010
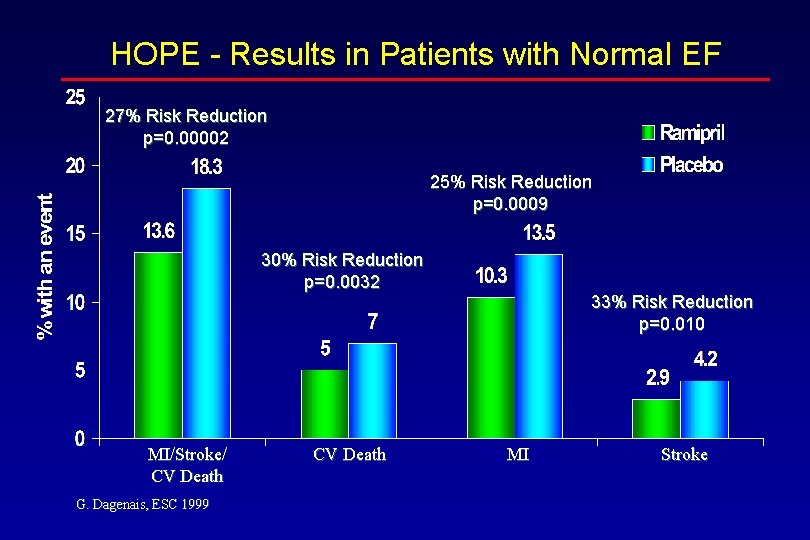
HOPE - Results in Patients with Normal EF 27% Risk Reduction p=0. 00002 25% Risk Reduction p=0. 0009 30% Risk Reduction p=0. 0032 MI/Stroke/ CV Death G. Dagenais, ESC 1999 CV Death 33% Risk Reduction p=0. 010 MI Stroke
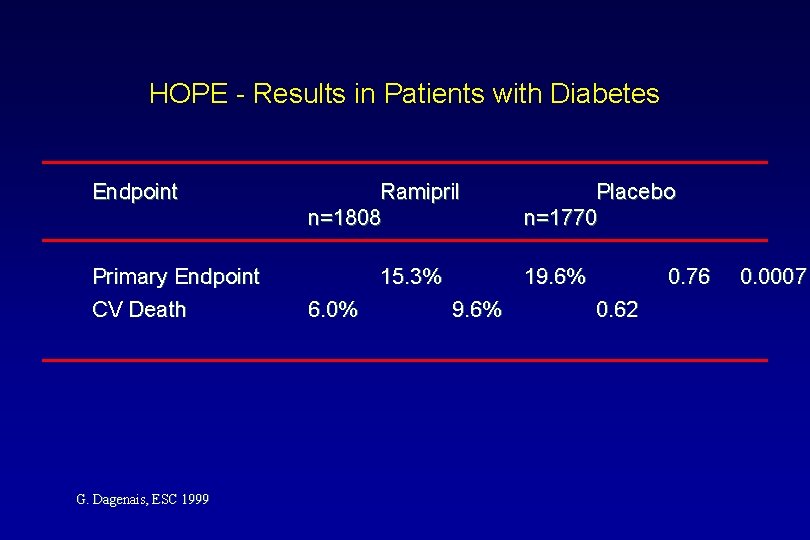
HOPE - Results in Patients with Diabetes Endpoint Primary Endpoint CV Death G. Dagenais, ESC 1999 Ramipril n=1808 15. 3% 6. 0% Placebo n=1770 19. 6% 0. 76 0. 62 0. 0007
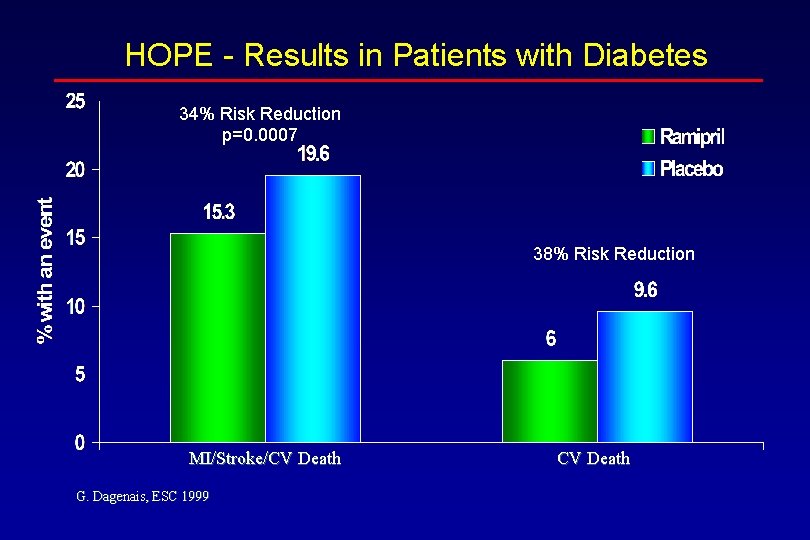
HOPE - Results in Patients with Diabetes 34% Risk Reduction p=0. 0007 38% Risk Reduction MI/Stroke/CV Death G. Dagenais, ESC 1999 CV Death
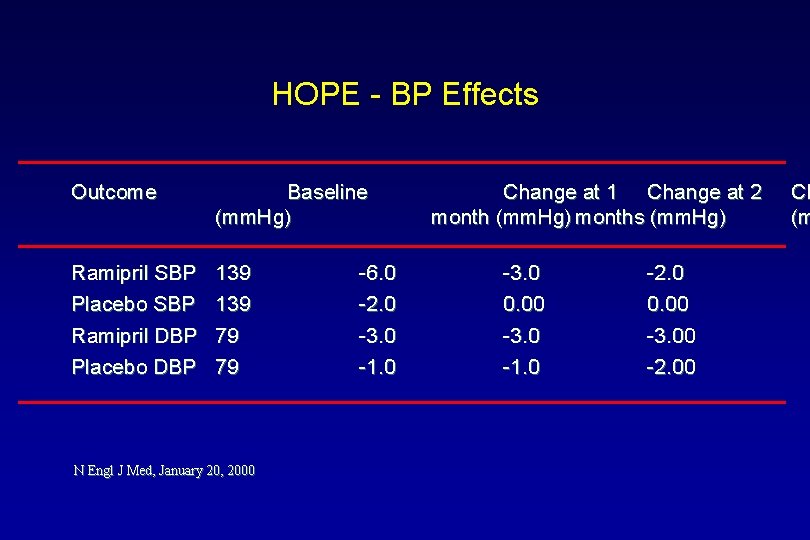
HOPE - BP Effects Outcome Baseline (mm. Hg) Ramipril SBP Placebo SBP Ramipril DBP Placebo DBP 139 79 79 N Engl J Med, January 20, 2000 -6. 0 -2. 0 -3. 0 -1. 0 Change at 1 Change at 2 month (mm. Hg) months (mm. Hg) -3. 0 0. 00 -3. 0 -1. 0 -2. 0 0. 00 -3. 00 -2. 00 Ch (m
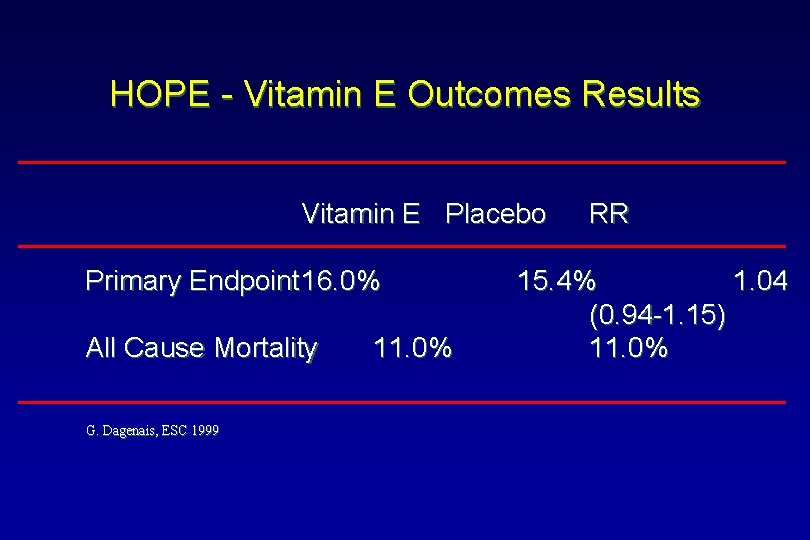
HOPE - Vitamin E Outcomes Results Vitamin E Placebo Primary Endpoint 16. 0% All Cause Mortality G. Dagenais, ESC 1999 11. 0% RR 15. 4% 1. 04 (0. 94 -1. 15) 11. 0%
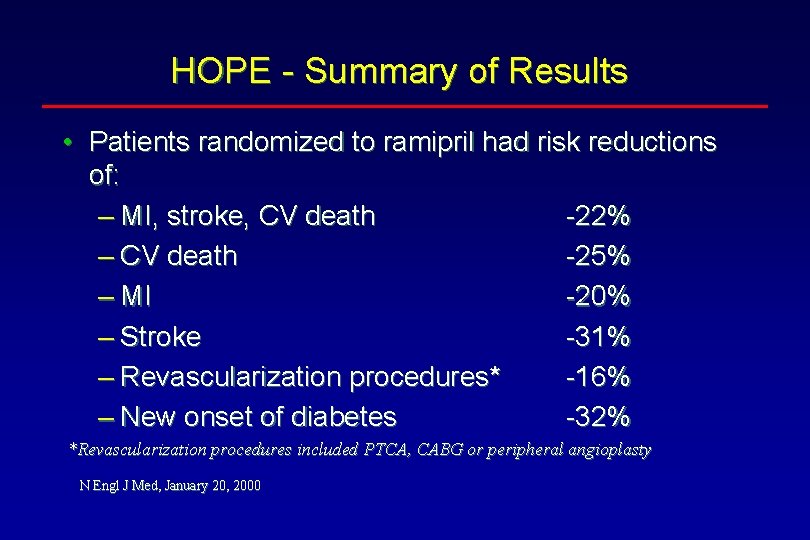
HOPE - Summary of Results • Patients randomized to ramipril had risk reductions of: – MI, stroke, CV death -22% – CV death -25% – MI -20% – Stroke -31% – Revascularization procedures* -16% – New onset of diabetes -32% *Revascularization procedures included PTCA, CABG or peripheral angioplasty N Engl J Med, January 20, 2000

HOPE - Summary of Results (continued) • The beneficial effect of treatment with ramipril on the composite outcome was consistently observed among the following predefined subgroups: – patients with and without diabetes – men and women – those with and without evidence of cardiovascular disease – those < 65 years of age and those > 65 year of age – those with and without hypertension at baseline* – those with and without microalbuminuria *A reduction of 2 mm Hg in diastolic blood pressure (as seen in this trial) might at best account for about 40% of the reduction in the rate of stroke and about 25% of the reduction of MI. N Engl J Med, January 20, 2000

HOPE - Summary of Results (continued) There was a clear benefit of ramipril among patients: with and without evidence of coronary artery disease at baseline with and without a history of myocardial infarction with a documented EF > 40% (27% risk reduction, p<0. 001) Benefits were also observed whether or not patients were also taking (at randomization): ASA or other antiplatelet agents beta-blockers lipid-lowering agents antihypertensive agents N Engl J Med, January 20, 2000

HOPE - Conclusions • “Ramipril significantly reduces the rates of death, myocardial infarction, and stroke in a broad range of high-risk patients who are not known to have a low ejection fraction or heart failure” N Engl J Med, January 20, 2000
- Slides: 21