Effect of the PCSK 9 Inhibitor Evolocumab on

Effect of the PCSK 9 Inhibitor, Evolocumab, on the Composition of Coronary Atherosclerosis: Insights from the GLAGOV Trial SJ Nicholls, H Kassahun, DM Brennan, K Wolski, J Yang, R Somaratne, SM Wasserman and SE Nissen

Disclosures • Research support: Astra. Zeneca, Amgen, Anthera, Eli Lilly, Esperion, Novartis, Cerenis, The Medicines Company, Resverlogix, Infra. Re. Dx, Roche, Sanofi. Regeneron and Lipo. Science • Consulting and honoraria: Astra. Zeneca, Eli Lilly, Anthera, Omthera, Merck, Takeda, Resverlogix, Sanofi-Regeneron, CSL Behring, Esperion, Boehringer Ingelheim

Background • The PCSK 9 inhibitor, evolocumab, lowers LDL-C, induces plaque regression on IVUS and reduces CV events in statin-treated ASCVD patients • The impact of PCSK 9 inhibition on the composition of coronary atheroma has not been investigated. • Virtual histology is an experimental technique which characterizes plaque composition and may have utility in assessing effects of anti-atherosclerotic therapies.
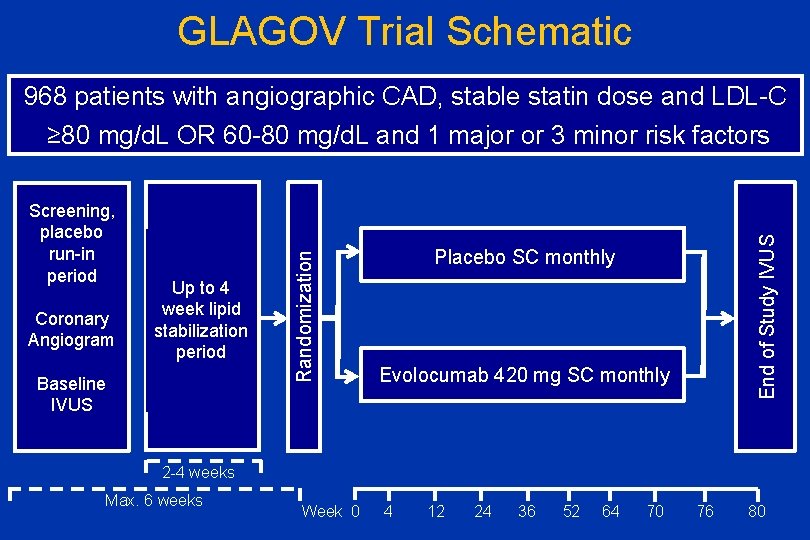
GLAGOV Trial Schematic 968 patients with angiographic CAD, stable statin dose and LDL-C Coronary Angiogram Up to 4 week lipid stabilization period Baseline IVUS End of Study IVUS Screening, placebo run-in period Randomization ≥ 80 mg/d. L OR 60 -80 mg/d. L and 1 major or 3 minor risk factors Placebo SC monthly Evolocumab 420 mg SC monthly 2 -4 weeks Max. 6 weeks Week 0 4 12 24 36 52 64 70 76 80

GLAGOV VH Substudy • Determine if evolocumab produced changes in VH- derived plaque components (dense calcium, fibrous, fibrofatty, necrotic core) compared with placebo in 331 patients with evaluable VH imaging. • The prespecified statistical plan sought to compare changes in volumetric measures, adjusting for baseline values and multiple comparisons. • The primary endpoint was the absolute change in dense calcium volume from baseline to week 78.
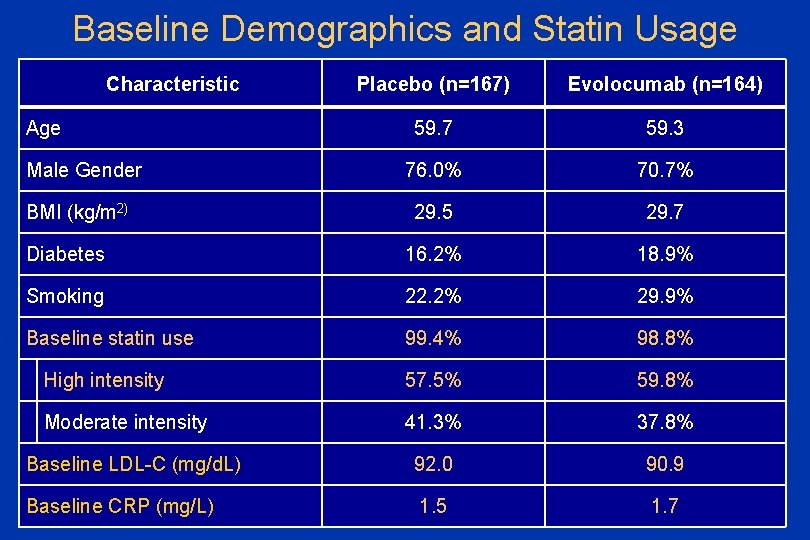
Baseline Demographics and Statin Usage Characteristic Placebo (n=167) Evolocumab (n=164) 59. 7 59. 3 76. 0% 70. 7% 29. 5 29. 7 Diabetes 16. 2% 18. 9% Smoking 22. 2% 29. 9% Baseline statin use 99. 4% 98. 8% High intensity 57. 5% 59. 8% Moderate intensity 41. 3% 37. 8% Baseline LDL-C (mg/d. L) 92. 0 90. 9 Baseline CRP (mg/L) 1. 5 1. 7 Age Male Gender BMI (kg/m 2)
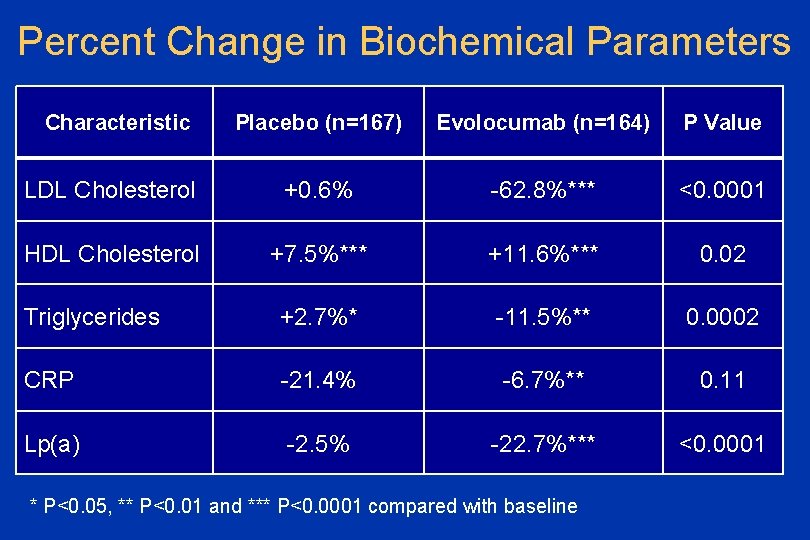
Percent Change in Biochemical Parameters Characteristic Placebo (n=167) Evolocumab (n=164) P Value LDL Cholesterol +0. 6% -62. 8%*** <0. 0001 HDL Cholesterol +7. 5%*** +11. 6%*** 0. 02 Triglycerides +2. 7%* -11. 5%** 0. 0002 CRP -21. 4% -6. 7%** 0. 11 Lp(a) -2. 5% -22. 7%*** <0. 0001 * P<0. 05, ** P<0. 01 and *** P<0. 0001 compared with baseline
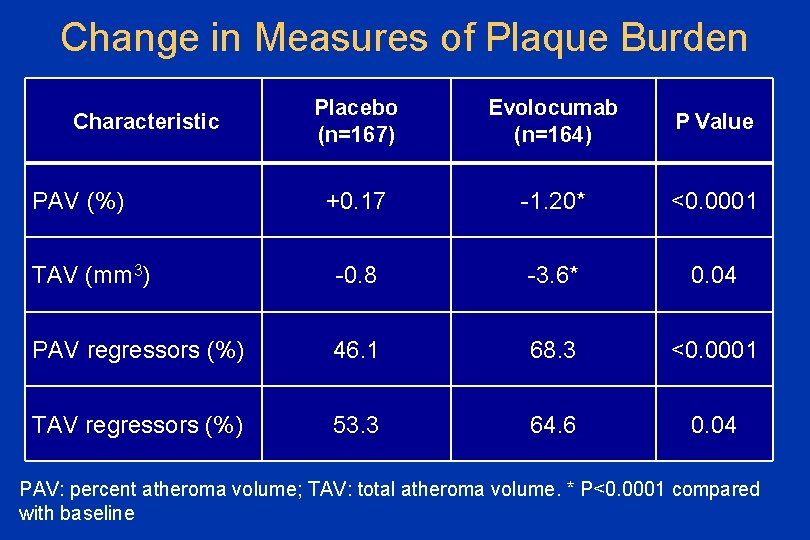
Change in Measures of Plaque Burden Placebo (n=167) Evolocumab (n=164) P Value +0. 17 -1. 20* <0. 0001 TAV (mm 3) -0. 8 -3. 6* 0. 04 PAV regressors (%) 46. 1 68. 3 <0. 0001 TAV regressors (%) 53. 3 64. 6 0. 04 Characteristic PAV (%) PAV: percent atheroma volume; TAV: total atheroma volume. * P<0. 0001 compared with baseline
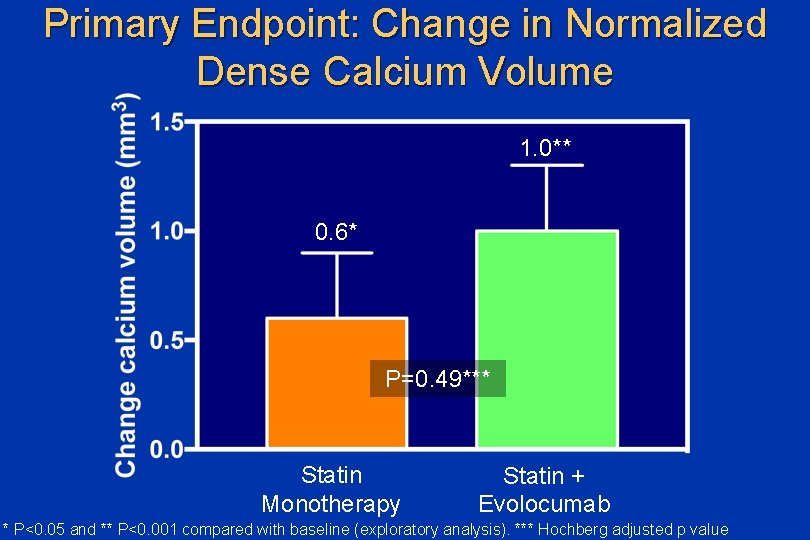
Primary Endpoint: Change in Normalized Dense Calcium Volume 1. 0** 0. 6* P=0. 49*** Statin Monotherapy Statin + Evolocumab * P<0. 05 and ** P<0. 001 compared with baseline (exploratory analysis). *** Hochberg adjusted p value
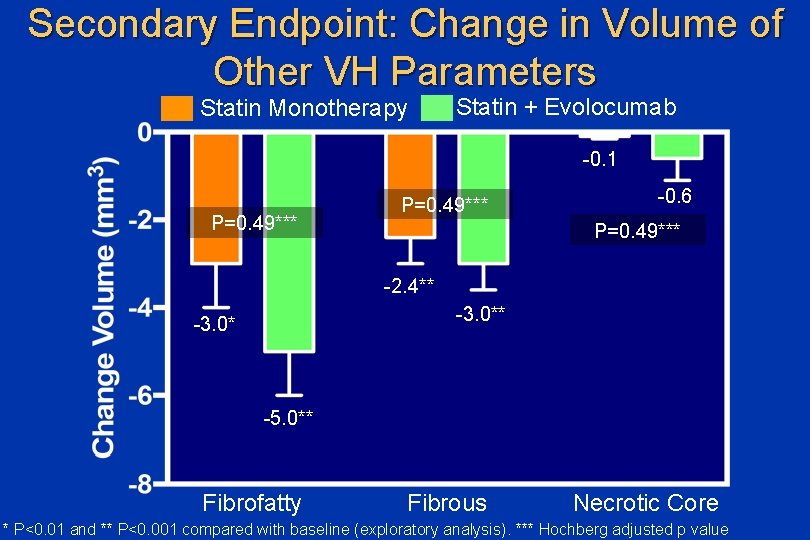
Secondary Endpoint: Change in Volume of Other VH Parameters Statin Monotherapy Statin + Evolocumab -0. 1 P=0. 49*** -0. 6 P=0. 49*** -2. 4** -3. 0* -5. 0** Fibrofatty Fibrous Necrotic Core * P<0. 01 and ** P<0. 001 compared with baseline (exploratory analysis). *** Hochberg adjusted p value
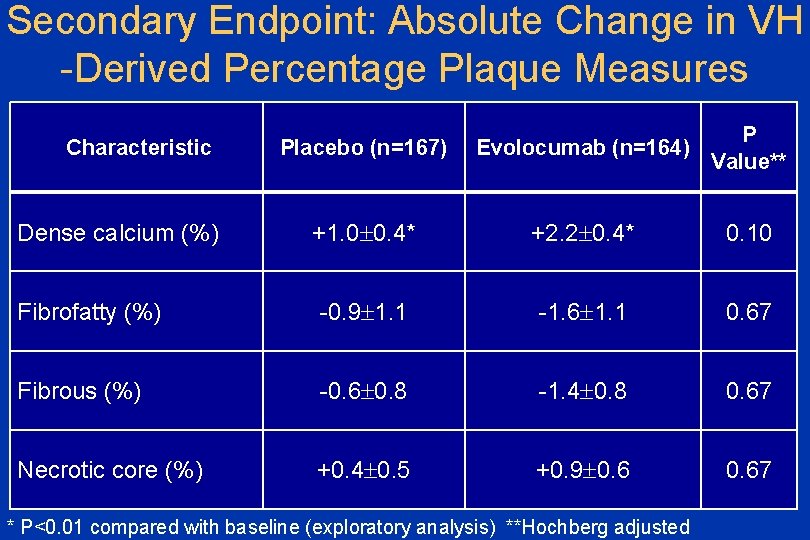
Secondary Endpoint: Absolute Change in VH -Derived Percentage Plaque Measures Placebo (n=167) Evolocumab (n=164) P Value** +1. 0 0. 4* +2. 2 0. 4* 0. 10 Fibrofatty (%) -0. 9 1. 1 -1. 6 1. 1 0. 67 Fibrous (%) -0. 6 0. 8 -1. 4 0. 8 0. 67 Necrotic core (%) +0. 4 0. 5 +0. 9 0. 67 Characteristic Dense calcium (%) * P<0. 01 compared with baseline (exploratory analysis) **Hochberg adjusted
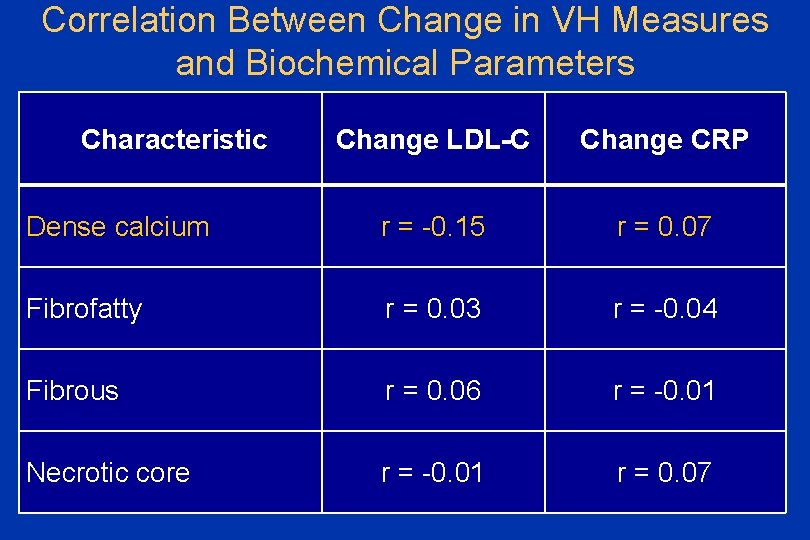
Correlation Between Change in VH Measures and Biochemical Parameters Characteristic Change LDL-C Change CRP Dense calcium r = -0. 15 r = 0. 07 Fibrofatty r = 0. 03 r = -0. 04 Fibrous r = 0. 06 r = -0. 01 Necrotic core r = -0. 01 r = 0. 07
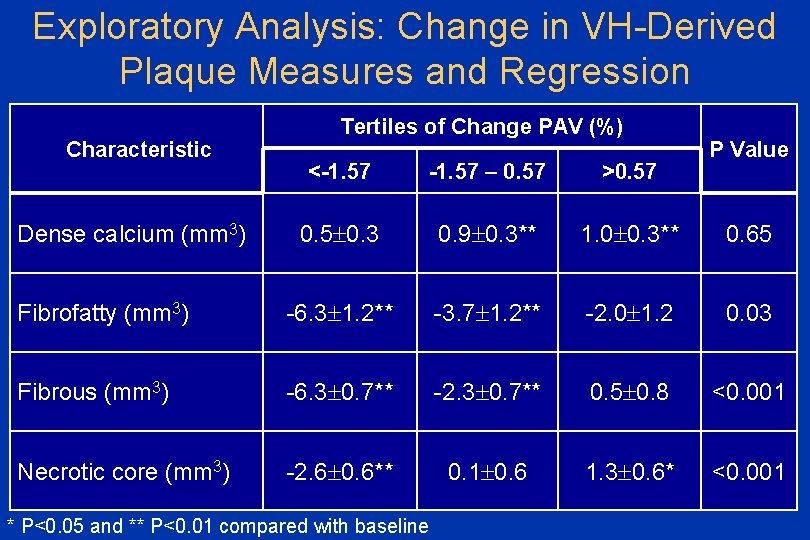
Exploratory Analysis: Change in VH-Derived Plaque Measures and Regression Characteristic Tertiles of Change PAV (%) P Value <-1. 57 – 0. 57 >0. 57 0. 5 0. 3 0. 9 0. 3** 1. 0 0. 3** 0. 65 Fibrofatty (mm 3) -6. 3 1. 2** -3. 7 1. 2** -2. 0 1. 2 0. 03 Fibrous (mm 3) -6. 3 0. 7** -2. 3 0. 7** 0. 5 0. 8 <0. 001 Necrotic core (mm 3) -2. 6 0. 6** 0. 1 0. 6 1. 3 0. 6* <0. 001 Dense calcium (mm 3) * P<0. 05 and ** P<0. 01 compared with baseline
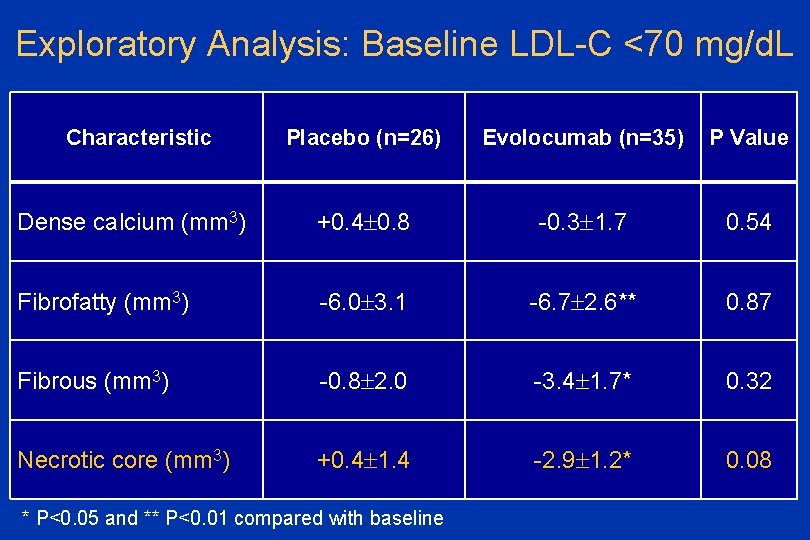
Exploratory Analysis: Baseline LDL-C <70 mg/d. L Characteristic Placebo (n=26) Evolocumab (n=35) P Value Dense calcium (mm 3) +0. 4 0. 8 -0. 3 1. 7 0. 54 Fibrofatty (mm 3) -6. 0 3. 1 -6. 7 2. 6** 0. 87 Fibrous (mm 3) -0. 8 2. 0 -3. 4 1. 7* 0. 32 Necrotic core (mm 3) +0. 4 1. 4 -2. 9 1. 2* 0. 08 * P<0. 05 and ** P<0. 01 compared with baseline

Conclusion • Evolocumab on a background of optimal statin therapy produced robust lowering of LDL cholesterol and plaque regression by conventional IVUS. • However, VH imaging failed to detect any difference between treatment groups for individual plaque components. • These findings further fuel uncertainty regarding the utility of VH imaging in drug development to assess the effect of anti-atherosclerotic therapies.

Final Thoughts • The inverse correlation between changes in LDL cholesterol and plaque calcium supports prior reports in studies of high intensity statins. • While the underlying mechanism remains uncertain, it questions the use of serial calcium scoring to monitor responses to lipid lowering interventions. • VH yields predictable, but not incremental information, in the setting of plaque regression. It will be of interest to see if other modalities can provide better insights.
- Slides: 16