EFFECT OF CHANGING CONCENTRATION ON RATE OF REACTION








- Slides: 12

EFFECT OF CHANGING CONCENTRATION ON RATE OF REACTION Using the Sunflower application

AIM OF THE TASK The aim of the task is for you to find out why, in terms of particles, changing concentration affects the rate of a chemical reaction.

SUCCESS CRITERIA • Explain what happens to the number of reactant particles by changing their concentration. • Use the terms collision and successful collision confidently. • Link the number of reactant particles with the number of collisions between reactants, and therefore the number of successful collisions between the reactant particles. • Explain how and why the rate of a chemical reaction changes as the number of successful collisions between the reactant particles change.

APPARATUS • Stop watch • Computer • ‘Sunflower Task – Changing Concentration’ worksheet

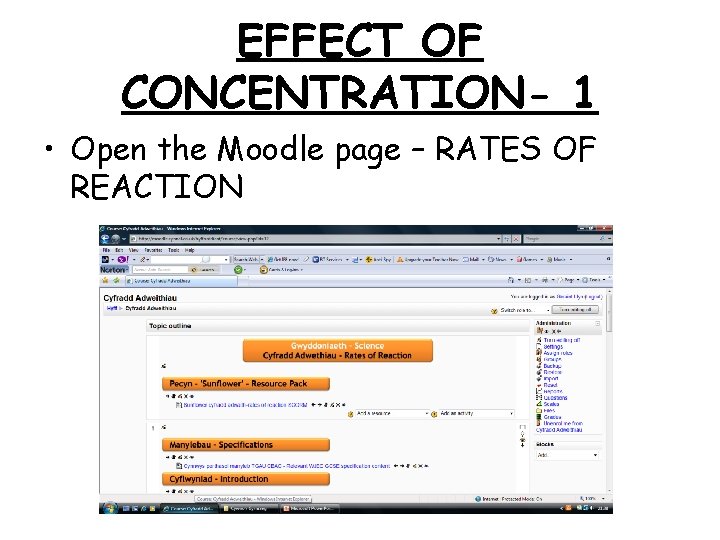
EFFECT OF CONCENTRATION- 1 • Open the Moodle page – RATES OF REACTION

EFFECT OF CONCENTRATION - 2 • Click on the following link: Task 1 – Effect of Changing Concentration

EFFECT OF CONCENTRATION - 3 • This page should appear:

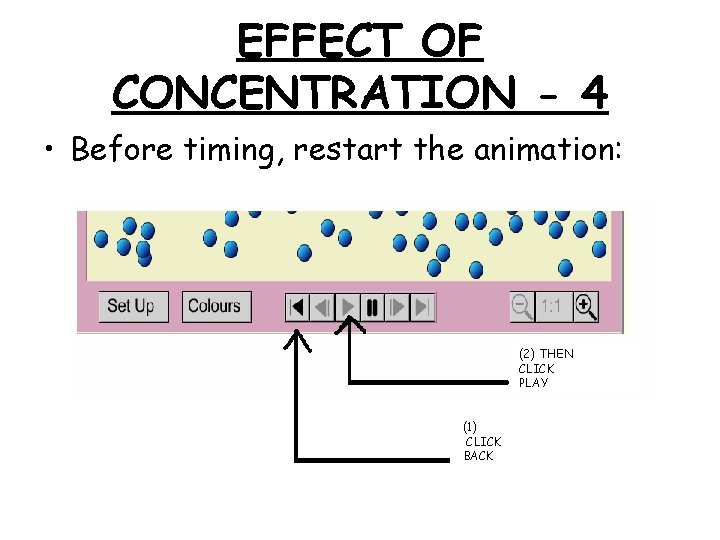
EFFECT OF CONCENTRATION - 4 • Before timing, restart the animation: (2) THEN CLICK PLAY (1) CLICK BACK

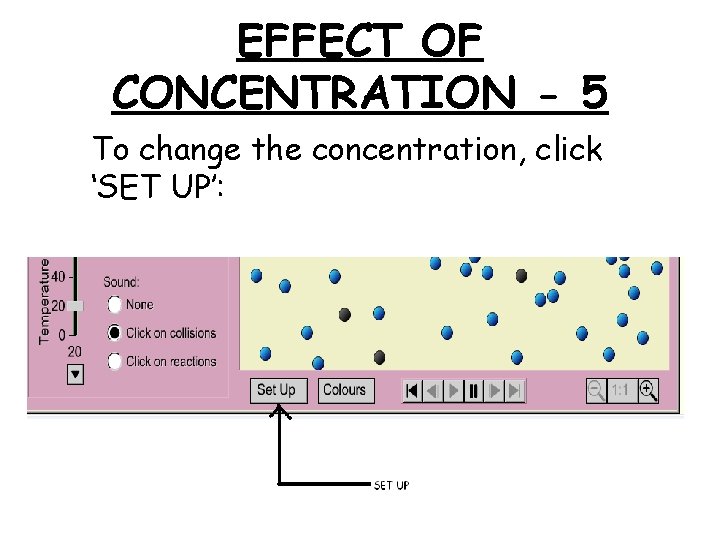
EFFECT OF CONCENTRATION - 5 To change the concentration, click ‘SET UP’:

EFFECT OF CONCENTRATION - 6 • This page should appear:

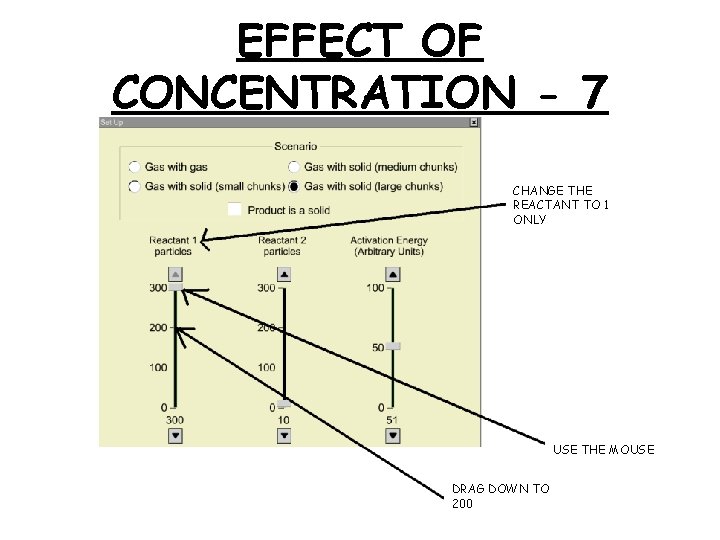
EFFECT OF CONCENTRATION - 7 CHANGE THE REACTANT TO 1 ONLY USE THE MOUSE DRAG DOWN TO 200

SUCCESS CRITERIA • Explain what happens to the number of reactant particles by changing their concentration. • Use the terms collision and successful collision confidently. • Link the number of reactant particles with the number of collisions between reactants, and therefore the number of successful collisions between the reactant particles. • Explain how and why the rate of a chemical reaction changes as the number of successful collisions between the reactant particles change.