Effect Measure Modification Ritz EPI 200 B Winter

Effect Measure Modification Ritz EPI 200 B Winter 2010

Effect Measure Modification Consider the combined effects of two exposures on disease occurrence: n Is the effect of one risk factor modified by another risk factor. Definition: Let C be a covariate unaffected by a study exposure E. Effect-measure modification is non-uniformity or heterogeneity of a measure of the E effect on the outcome D across levels of C. That is, according to the effect measure being used, the true (net causal) effect varies over levels of a covariate exposure. Thus, lack of effect measure modification refers to the uniformity or homogeneity of the effect measure. A parallel concept is statistical interaction, which refers to heterogeneity of a measure of the E-D association across levels of another covariate C in the source population (although the term is often used synonymous with effect measure modification)

Effect Measure Modification Effect measure modification is often a very important concept for several reasons: n It is often a key concern when analyzing data and when interpreting statistical results – e. g. , to determine whether we should be estimating a common effect measure or how to select predictors for a statistical model. n It is sometimes used to operationalize specific hypotheses regarding the effects of two exposures – e. g. , we might hypothesize that the effect of one exposure should vary in a certain way over categories of another exposure. n It is relevant for generalizing results across studies and populations.

Effect Measure Modification cont. The assessment of effect modification (and statistical interaction) depends on the specific measure used to quantify the effect; That is, the assessment of effect modification is measure dependent or model dependent (thus the term “effect-measure modification” which is often simply termed “effect modification”). For example, if the risk ratio for the E effect is non-null and homogeneous (uniform) across categories (strata) of C, the risk difference for the E effect will be heterogeneous across C; and if the risk difference is non-null and uniform, the risk ratio will be heterogeneous. Furthermore the risk difference and the risk ratio could exhibit opposite (or the same) direction of change across C.
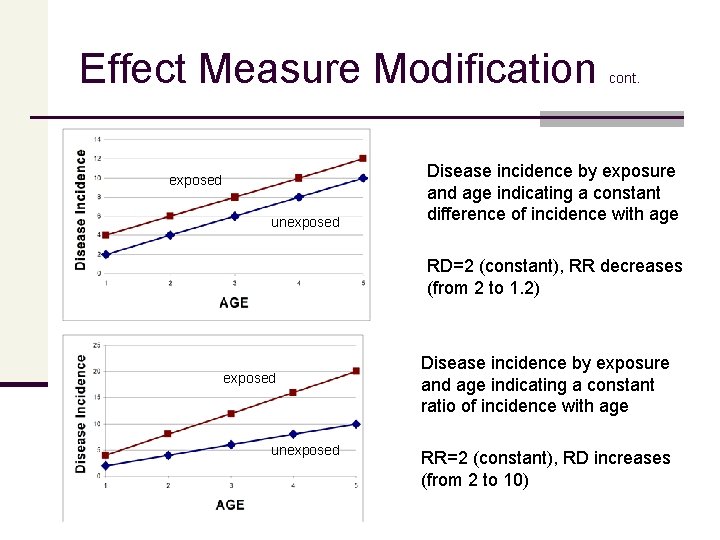
Effect Measure Modification exposed unexposed cont. Disease incidence by exposure and age indicating a constant difference of incidence with age RD=2 (constant), RR decreases (from 2 to 1. 2) exposed unexposed Disease incidence by exposure and age indicating a constant ratio of incidence with age RR=2 (constant), RD increases (from 2 to 10)

Examples: Effect Measure Modification n The following table gives the counterfactual risks of a disease in 4 source populations, by gender, if everyone in that population were exposed and if everyone were unexposed n Thus, gender is being assessed as a potential modifier of the exposure effect; I. e. we want to know whether the exposure effect differs for men and women. n Also shown in the table are the causal risk ratio (RR) and the causal risk difference (RD) for men and women in each population.
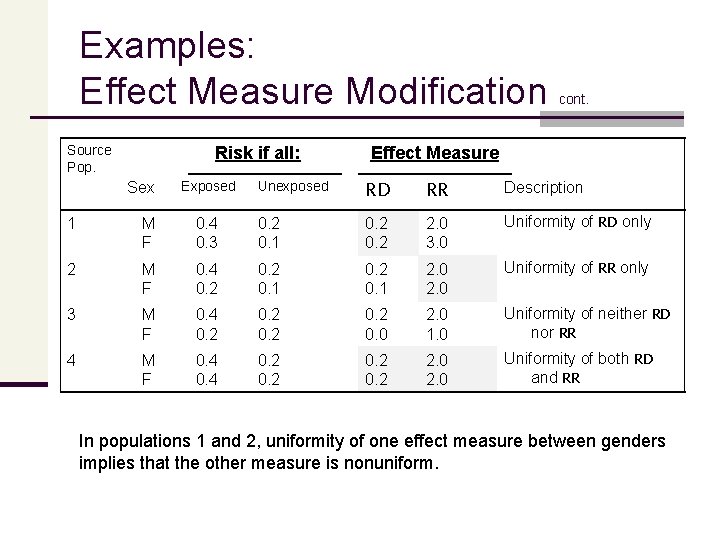
Examples: Effect Measure Modification Source Pop. Risk if all: Sex Exposed cont. Effect Measure Unexposed RD RR Description 1 M F 0. 4 0. 3 0. 2 0. 1 0. 2 2. 0 3. 0 Uniformity of RD only 2 M F 0. 4 0. 2 0. 1 2. 0 Uniformity of RR only 3 M F 0. 4 0. 2 0. 0 2. 0 1. 0 Uniformity of neither RD nor RR 4 M F 0. 4 0. 2 2. 0 Uniformity of both RD and RR In populations 1 and 2, uniformity of one effect measure between genders implies that the other measure is nonuniform.

Examples: Effect Measure Modification cont. n Gender n modifies the RR (but not the RD) in population 1; n modifies the RD (but not the RR) in population 2; n modifies both effect measures in population 3. n is not a risk factor in population 4 (under either exposure condition), thus, it does not modify RR or RD in that population. Thus, we see that the assessment of effect measure modification is measure or model dependent; and that for a covariate to be a modifier, it must have an effect in at least one exposure level. Note: We cannot represent effect measure modification in a DAG since these are scale free, while effect measure modification is scale dependent

Effect Measure Modification Comment: n Uniformity of the causal risk difference indicates that the effects of the two factors are additive (on the risk scale). n Uniformity of the causal risk ratio indicates that the effects are multiplicative (on the risk scale). n Just as we cannot directly observe the effect (counterfactual contrast) of a single exposure in a population, we also cannot directly observe effect modification. n To assess effect modification, therefore, we can compare measures of association for one exposure over levels of another exposure.

Statistical Interaction - definition Statistical interaction (association measure modification) refers to nonuniformity/heterogeneity of a (true) measure of association between disease and one exposure over levels of another exposure in the source population. Thus, assuming no confounding at each level of the potential modifier, we can use our assessment of statistical interaction to make inferences about effect modification. For example, we might compare the risk ratio (RR) or the risk difference (RD) for the exposure-disease association across different categories of the potential modifier. Note: The terms effect measure modification, statistical interaction, and interaction effect are often used synonymously; e. g. , Rothman & Greenland (pp. 329 -32, ME 2)

Statistical Interaction Using data from a specific study, we estimate the amount of statistical interaction between two exposures in the study group by comparing estimated measures of association (e. g. , RR, RD) for one exposure across different levels of the other exposure. These estimates, however, would almost never perfectly represent the true effect parameters in the source population: Estimates of statistical interaction may reflect random and nonrandom error (i. e. , chance and bias) as well as true effect modification. Thus, the same accuracy problems that threaten the estimation of exposure effect also threaten the estimation of effect measure modification. Furthermore, in the assessment of effect measure modification, the amount of error in estimating the exposure effect might vary across levels of the covariate (e. g. different levels of confounding by gender). Since effect measure modification and statistical interaction are measure dependent, it is important to specify the measure being used for their assessment.
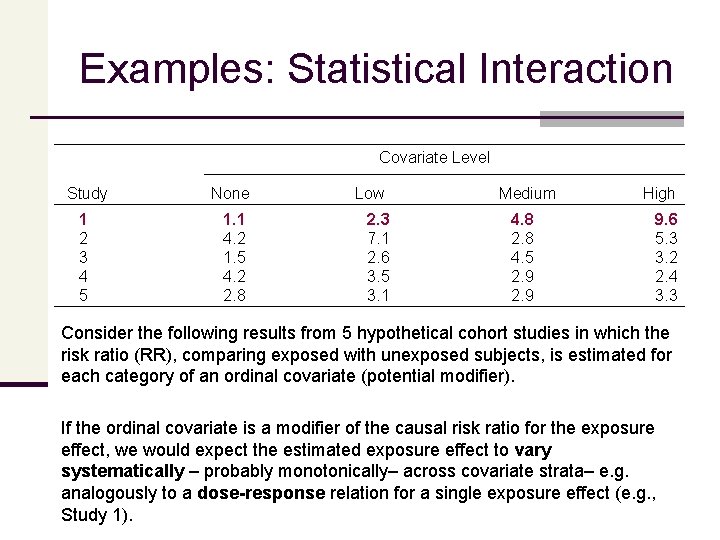
Examples: Statistical Interaction Covariate Level Study None 1 2 3 4 5 1. 1 4. 2 1. 5 4. 2 2. 8 Low 2. 3 7. 1 2. 6 3. 5 3. 1 Medium 4. 8 2. 8 4. 5 2. 9 High 9. 6 5. 3 3. 2 2. 4 3. 3 Consider the following results from 5 hypothetical cohort studies in which the risk ratio (RR), comparing exposed with unexposed subjects, is estimated for each category of an ordinal covariate (potential modifier). If the ordinal covariate is a modifier of the causal risk ratio for the exposure effect, we would expect the estimated exposure effect to vary systematically – probably monotonically– across covariate strata– e. g. analogously to a dose-response relation for a single exposure effect (e. g. , Study 1).

Examples: Statistical Interaction Study None 2 3 4 4. 2 1. 5 4. 2 Covariate Level Low Medium 7. 1 2. 6 3. 5 2. 8 4. 5 2. 9 High 5. 3 3. 2 2. 4 Strong statistical interaction that does not fit this monotonic pattern (e. g. , Study 2 or 3) may reflect • a true non-monotone association or • various sources of bias • e. g. , nondifferential exposure misclassification, where the amount of misclassification is associated with the covariate level, or • covariate misclassification. That is, such misclassification might result in apparent heterogeneity of effect when none exists, or it might distort the true pattern of effect heterogeneity. Note: even a consistent monotonic pattern such as in Study 4 may be interpreted in this manner

Examples: Statistical Interaction Covariate Level Study None 5 2. 8 Low 3. 1 Medium 2. 9 High 3. 3 Apparent heterogeneity of effect, especially minor fluctuations in estimated effect that are not monotonic (e. g. , Study 5), might also be due to random (sampling) error. To deal with random error, we often test for homogeneity of effect, using stratified analysis or model fitting. The basic idea of the stratified test is to construct a summary across strata assuming the null of homogeneity (a ‘common’ effect estimate) and then create a (Chi-squared) summary of the distance from the stratum specific to that common effect estimate. Modeling methods instead introduce product terms to represent heterogeneity, and estimate those, which can be more powerful and (given current software) much easier.

Woolf's Test For Heterogeneity Of A Ratio Measure where g = number of strata; and Under the null hypothesis of homogeneity (no effect measure modification), has a chi square distribution with (g− 1) degrees of freedom (df). The alternative hypothesis is any difference among strata (not necessarily a monotonic trend in the ratio). Comments: Woolf’s test of homogeneity is a type of Wald test, and it can also be applied to test for heterogeneity of risk or rate ratios and risk or rate differences (ME 2: 275 -77). Although strictly speaking it incorrect to do so, Woolf’s test can be applied using the logarithm of the Mantel-Haenszel estimates of the common rate or odds ratios in place of Woolf's estimator of the common effect (a θ). The statistic becomes inaccurate, however, when using MH risk ratios or MH risk and rate difference measures.

Woolf's Test For Heterogeneity A problem with all tests of homogeneity is that there is rarely any reason to think that homogeneity is present; in other words, we should expect the null to be false, so why bother to test it? Failure to reject only indicates we have insufficient precision to distinguish the actual heterogeneity from the null (which is usually the case); I. e. a large p-value tells us that we do not have enough information to distinguish heterogeneity from random noise, I. e. it doesn’t say that homogeneity is present.
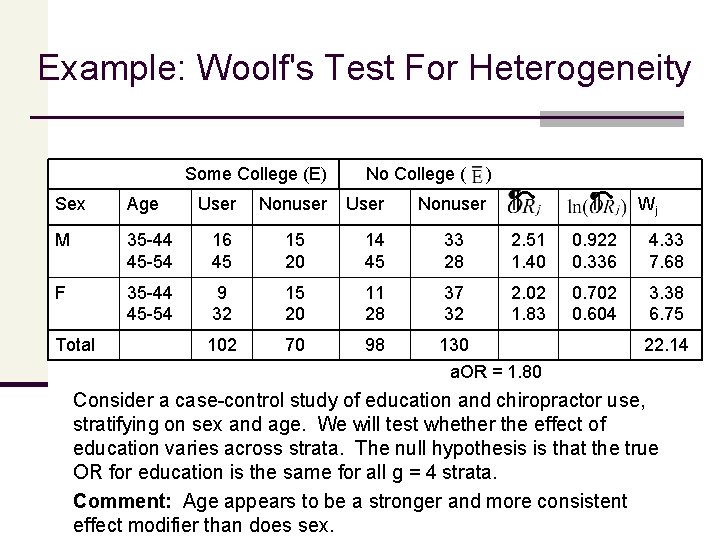
Example: Woolf's Test For Heterogeneity Some College (E) Sex Age M F Total No College ( User ) User Nonuser Wj 35 -44 45 -54 16 45 15 20 14 45 33 28 2. 51 1. 40 0. 922 0. 336 4. 33 7. 68 35 -44 45 -54 9 32 15 20 11 28 37 32 2. 02 1. 83 0. 702 0. 604 3. 38 6. 75 102 70 98 130 22. 14 a. OR = 1. 80 Consider a case-control study of education and chiropractor use, stratifying on sex and age. We will test whether the effect of education varies across strata. The null hypothesis is that the true OR for education is the same for all g = 4 strata. Comment: Age appears to be a stronger and more consistent effect modifier than does sex.

Example: Woolf's Test For Heterogeneity cont. n With g− 1 = 3 df, the 2 -sided P value is 0. 80. Thus, despite the considerable heterogeneity in the observed stratum-specific odds ratios, the P-value derived from Woolf's test is large. n This result does not mean the true odds ratios are homogeneous; it simply means the data are inadequate to discriminate between the observed degree of heterogeneity (1. 4 to 2. 5) and the null (homogeneity).

Component and Joint Effects see ME 2 Chapter 18, pp 329 -342 n Thus far, we have examined interaction effects by estimating the effect of one exposure (the study factor) over categories of another exposure (the modifier). n An alternative way to view the same phenomena separates the combined effects of two exposures into three parts: the two component effects of each exposure– i. e. , its effect in the absence of (in the reference category of) the other exposure; n the joint effect of the two exposures n

Component and Joint Effects Suppose we have two dichotomous exposures, E and F, and suppose we know what the average risk (Rij) in the target population would be under all four combinations of these two exposures–i. e. , the i-th category of E (i = 0, 1) and the j-th category of F (j = 0, 1). F (j=1) (j=0) E (i=1) R 11 R 10 (i=0) R 01 R 00

Component and Joint Effects definitions The component effect of E is the comparison of R 10 with R 00–i. e. , RR 10 = R 10/R 00 or RD 10 = R 10 − R 00. The component effect of F is the comparison of R 01 with R 00–i. e. , RR 01= R 01/R 00 or RD 01 = R 01 − R 00. The joint effect of E and F is the comparison of R 11 with R 00–i. e. , RR 11= R 11/R 00 or RD 11 = R 11 − R 00. F (j=1) (j=0) E (i=1) R 11 R 10 (i=0) R 01 R 00

Component and Joint Effects All three effects involve the same population, with persons unexposed to both E and F ( ) as the joint reference group. To describe the amount of interaction, it is common to compare the joint effect with our expectation of the joint effect under two null conditions: additivity of effects and multiplicativity of effects The expected value of the joint effect under each null condition is derived from the component effects. That is, we express additivity and multiplicativity in terms of component and joint effects. n Comment: It is common to see analyses that examine only multiplicativity. Nonetheless, additivity turns out have a closer connection to basic causal models of interaction (see ME 2, Ch. 18).

Additive Effects F (j=1) (j=0) E (i=1) R 11 R 10 (i=0) R 01 R 00 Consider a target population in which we know what the (possibly counterfactual) average risk (Rij) would be under the i-th category of exposure E and the j-th category of exposure F. Suppose that the causal risk difference (RD) for each exposure is homogeneous over levels of the other exposure– i. e. , R 11−R 01 = R 10−R 00 and R 11−R 10 = R 01−R 00. Then R 11 = R 10 + R 01 − R 00.

Additive Effects F (j=1) (j=0) E (i=1) R 11 R 10 (i=0) R 01 R 00 Subtracting R 00 from each side of Equation (1), we have (R 11−R 00) =(R 10−R 00) + (R 01−R 00), or RD 11 = RD 10 + RD 01. Thus, the joint effect (RD 11) is equal to the sum of the two component effects. (Note: Uniformity of the risk difference means that the two effects are additive on the risk scale; nonuniformity of the risk difference means that these effects are not additive on the risk scale).

Additive Effects F (j=1) cont. (j=0) E (i=1) R 11 R 10 (i=0) R 01 R 00 If RD 11 > RD 10 + RD 01, we say the effects are superadditive or transadditive on the risk scale or there is positive interaction under an additive risk model–i. e. , effect measure modification of the risk difference. If RD 11 < RD 10 + RD 01, we say the effects are subadditive on the risk scale or there is negative interaction under an additive risk model. We can also express additivity of effects in terms of risk ratios (RRij). Dividing both sides of Equation (1) by R 00, we have

Multiplicative Effects F (j=1) (j=0) E (i=1) R 11 R 10 (i=0) R 01 R 00 Suppose now that the causal risk ratio (RR) for each exposure is homogeneous over levels of the other exposure– i. e. , R 11/R 01 = R 10/R 00, or R 11/R 10 = R 01/R 00. Then Dividing both sides of this equation by R 00, we have That is, the joint effect (RR 11) is equal to the product of the two component effects. Thus, uniformity of the risk ratio means that the two effects are multiplicative on the risk scale; nonuniformity of the risk ratio means that these effects are not multiplicative on the risk scale

Multiplicative Effects cont. If RR 11 > RR 10 x RR 01, we say the effects are supermultiplicative on the risk scale or there is positive interaction under a multiplicative risk model–i. e. , effect measure modification of the risk ratio. If RR 11 < RR 10 x RR 01, we say the effects are submultiplicative on the risk scale or there is negative interaction in a multiplicative risk model. Comments: We can also express additivity and multiplicativity of effects in terms of odds or rates instead of risks. This will cause no problem if the disease is rare in all categories. In case-control studies, we use the odds ratio to estimate risk ratios in order to assess interaction effects.
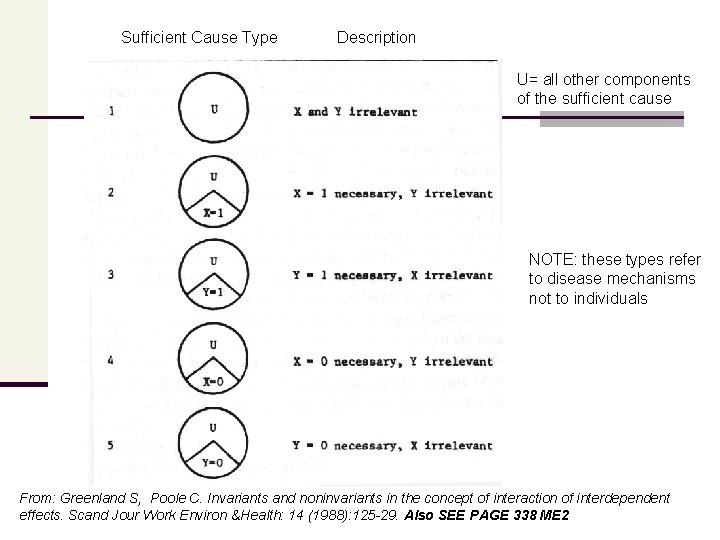
Sufficient Cause Type Description U= all other components of the sufficient cause NOTE: these types refer to disease mechanisms not to individuals From: Greenland S, Poole C. Invariants and noninvariants in the concept of interaction of interdependent effects. Scand Jour Work Environ &Health: 14 (1988): 125 -29. Also SEE PAGE 338 ME 2
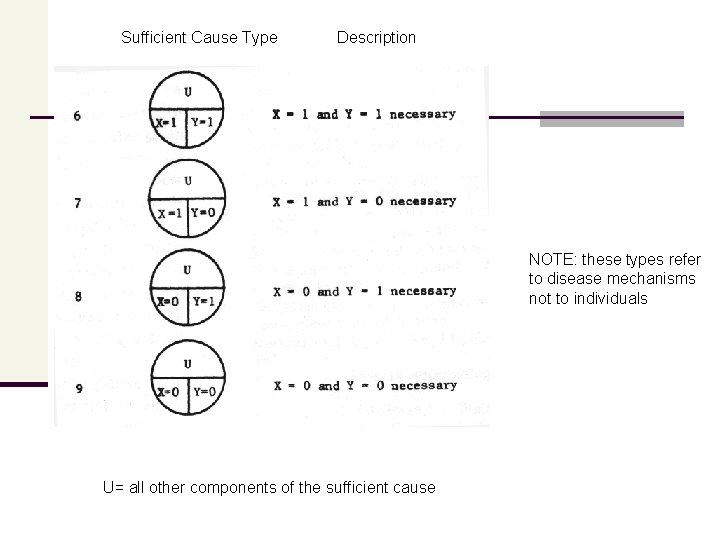
Sufficient Cause Type Description NOTE: these types refer to disease mechanisms not to individuals U= all other components of the sufficient cause
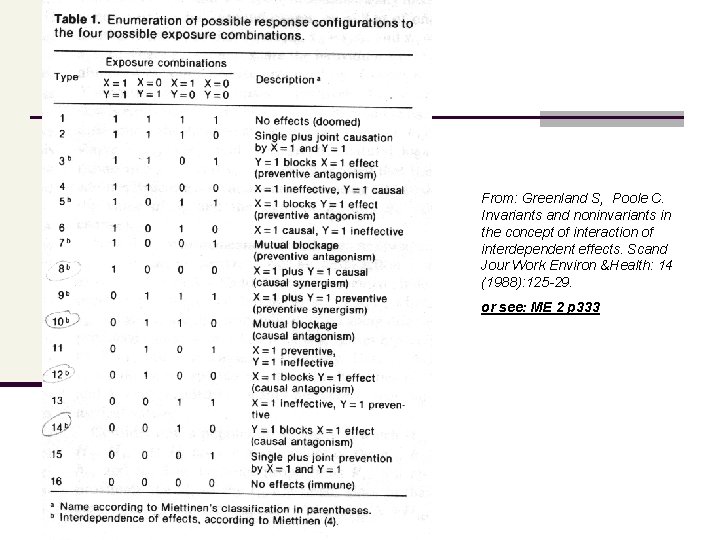
From: Greenland S, Poole C. Invariants and noninvariants in the concept of interaction of interdependent effects. Scand Jour Work Environ &Health: 14 (1988): 125 -29. or see: ME 2 p 333
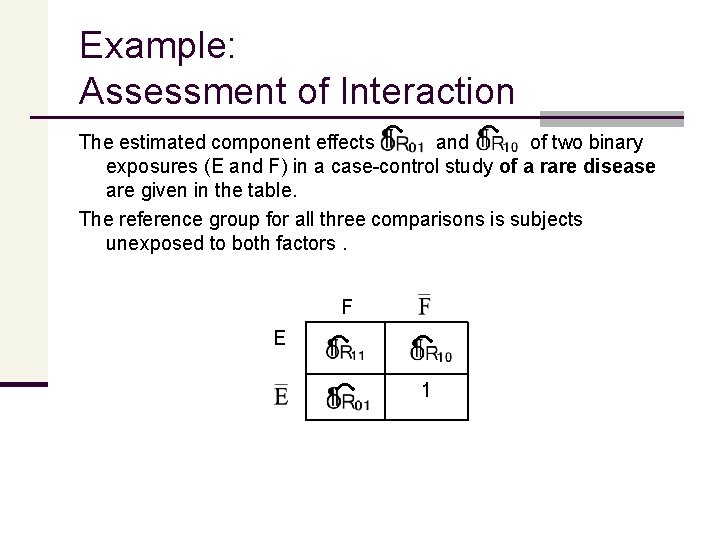
Example: Assessment of Interaction The estimated component effects and of two binary exposures (E and F) in a case-control study of a rare disease are given in the table. The reference group for all three comparisons is subjects unexposed to both factors. F E 1

Example: Assessment of Interaction If the effects of E and F are additive on the risk scale (i. e. , uniformity of the risk difference) we would expect the additive interaction contrast to be zero. (1) If the effects of E and F are multiplicative on the risk scale (i. e. , uniformity of risk ratio), we would expect the multiplicative interaction contrast to be 1. OR 11 OR 10 x OR 01 (2) The estimated joint effect ( ) therefore, can be compared with two null conditions of "no interaction, " corresponding to additivity (risk difference homogeneity) and multiplicativity (risk ratio homogeneity) on the risk scale, respectively.

Example: Assessment of Interaction The Woolf method given earlier is a simple way of testing these conditions. However they are even easier to test by testing n n the fit of an additive risk-regression model (for 1) or a multiplicative risk-regression model (for 2). For a rare outcome, the logistic model (which is a multiplicative odds model) approximates the multiplicative risk model. A more informative analysis step places confidence limits on (1) and (2), to see what sort of interactions are reasonably compatible with the data (assuming only random error is present); This step corresponds to putting confidence limits on the EF product-term coefficient in the regression model (additive for 1, multiplicative for 2).

Effect Measure Modification And Confounding Very often effect measure modification (or interaction) is confused with confounding, perhaps because we can attempt to identify both phenomena with the same general analytic methods–i. e. , stratification or model fitting. Nevertheless, the two concepts are very different: n Effect measure modification refers to heterogeneity of effect, n Confounding refers to bias in the estimation of effect due to lack of comparability between exposure groups. n Effect measure modification is a property of the effect under study, which is often of interest to the investigator; n Confounding is an estimation problem that the investigator tries to prevent or eliminate

Effect Measure Modification And Confounding Consequently, assessments of effect measure modification and confounding are different and may be performed separately for each covariate in a given study. When examining a specific exposure-disease relation, a given risk factor for the disease may be: 1) a modifier and a confounder of the exposure effect; or 2) a modifier and not a confounder; or 3) a confounder but not a modifier; or 4) neither a modifier nor a confounder. To assess effect measure modification n we observe how the estimated effect measure varies across levels of the covariate, or n we compare the estimated joint and component effects of the exposure and covariate. Note again that this assessment is measure (model) dependent.

Effect Measure Modification And Confounding cont. To assess the apparent confounding by a covariate n we “average” the estimated exposure effects across covariate levels (e. g. , using standardization to adjust the covariate distribution to the target population distribution) and n compare this adjusted effect estimate with the estimated effect ignoring the covariate. This is called the change-in-estimate criterion. It is strictly valid only when using risk ratios or risk differences (or mean differences for continuous variables). But for rare diseases (meaning: rare in every category considered) it can also be used with rates or odds. Use of this criterion of course assumes that the covariate is unaffected by exposure and disease.

Example: Effect Modifier Or Confounder Or Both? Consider the effect of smoking on cervical cancer, by race (a risk factor for the disease). The table below shows the number (N, x 1000) of women at risk and the average risk (cases per 100, 000 ascertained over a one year period) of cervical cancer, by smoking status (the exposure) and race (the covariate), in 4 different source cohorts. Each cohort is followed for one year and contains 1 million people, 30% of whom are smokers and 20% of whom are black. Assume that the R values in this table are true risks (not estimates), there are no other confounders in each source population, and that the smokers form the target.
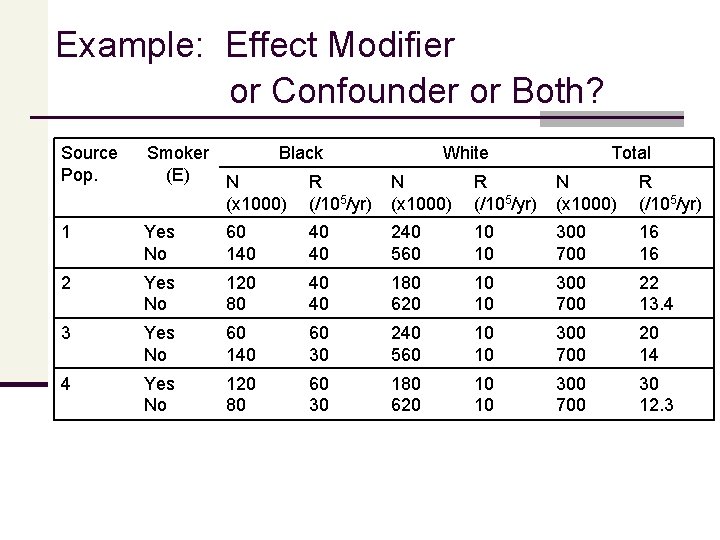
Example: Effect Modifier or Confounder or Both? Source Pop. Smoker Black (E) N R (x 1000) (/105/yr) White Total N (x 1000) R (/105/yr) 1 Yes No 60 140 40 40 240 560 10 10 300 700 16 16 2 Yes No 120 80 40 40 180 620 10 10 300 700 22 13. 4 3 Yes No 60 140 60 30 240 560 10 10 300 700 20 14 4 Yes No 120 80 60 30 180 620 10 10 300 700 30 12. 3

Example: Effect Modifier or Confounder or Both? Source Pop. 1 2 3 4 RR (Smoking) Black White 1. 00 2. 00 1. 00 c. RR 1. 00 1. 64 1. 43 2. 44 s. RR 1. 00 1. 43 1. 67 Race is a Confounder? No Yes Modifier? No No Yes The table on summarizes the race-specific and “overall” effects of smoking in each population and indicates whether race is a confounder or modifier of the smoking effect in each cohort. Note that the overall or summary effects are expressed in two ways: n as crude risk ratios (c. RR), ignoring race; and n as risk ratios internally standardized for race (s. RR). Internal standardization creates a group of nonsmokers with the same race distribution in smokers–i. e. , the unexposed group is “made comparable” to the exposed with respect to race (the potential confounder).

Example: Effect Modifier or Confounder or Both? To determine whether race is a confounder of the smoking effect in each population, n we compare the crude and standardized risk ratios (assuming no other sources of estimation error) n if these two risk ratios are equal, there is no confounding by race in that population; n if these two risk ratios are different, there is confounding by race. Note that confounding by race requires an association between race and smoking in the total source population (as in 2 and 4 but not in 1 or 3).

Example: Effect Modifier or Confounder or Both? To determine whether race is a modifier of the risk ratio for the smoking effect in each population n we compare the risk ratio for blacks with the risk ratio for whites. n n If these two risk ratios are equal, race does not modify the risk ratio in that population; if these two risk ratios are different, race does modify the risk ratio. Unlike confounding, effect measure modification by race does not require an association between race and smoking in the source population. Conclusion: Depending on the pattern of effects and associations in the source population, a cause of disease may be either a confounder for the exposure effect, a modifier of the exposure effect, or neither, or both.

Example: Effect Modifier or Confounder or Both? Some researchers would maintain that if there appears to be appreciable effect measure modification between two exposures then any confounding of either effect by the other exposure is irrelevant. For example, if race appears to modify the exposure effect, we should report race-specific, not summary (adjusted), estimates of effect. The primary reason for this view is that any summary estimate of effect is misleading when there is substantial heterogeneity. Here are three counterarguments to the position that the assessment of effect measure modification always supersedes the control of confounding:

1. Counterargument: Effect measure modification does not supersede control of confounding: Precision and power for studying interaction effects is often poor– much lower than the precision and power for single (main) effects of one of the exposures considered alone (in the same study). Even with no bias, observed interactions may poorly reflect the true degree of effect measure modification in the source population. Confidence intervals for the degree of modification are valuable for making the imprecision clear. These are most easily computed from confidence intervals for product terms (like C x E, E x F, etc. , often called “interaction terms”) in regression models. They often reveal that little can be said or done about heterogeneity, even when much can be said and done about confounding.

2. Counterargument: Effect measure modification does not supersede control of confounding: Even when a covariate is a strong modifier, we still can estimate the impact of the exposure on total disease occurrence (e. g. , attributable or preventable fraction) by standardizing for the covariate as a confounder–i. e. , the presence of effect measure modification does not change the interpretation of the attributable/preventable fraction. In fact, we are always averaging effects for unknown or unmeasured effect measure modifiers. Nevertheless, ignoring interactions is particularly inappropriate in certain situations, such as when the exposure has a positive effect in one group and an inverse effect in another group, i. e. , when there is a crossover effect.

3. Counterargument: Effect measure modification does not supersede control of confounding: If a covariate is a continuous or ordinal variable, we might want to treat it both as a confounder and as a modifier, simultaneously. That is, first we adjust for the covariate as a confounder within larger strata of the same covariate; Then we compare the adjusted estimates among the larger strata to determine whether the covariate is a modifier.

Example: Smoking and risk of cervical cancer in pre- and postmenopausal women When examining the possible effect of smoking on the risk of cervical cancer, we might hypothesize that the smoking effect differs for premenopausal and post-menopausal women, since uterine carcinogenesis is hormonally related. To test this hypothesis, we could treat age both as a potential confounder and as a potential modifier. Thus, first we estimate the smoking effect separately for pre- and postmenopausal women (e. g. , age < 50 vs. age ≥ 50), controlling for age within each group (as a confounder). Such age adjustment might be done by further stratifying the data into 5 -year age categories both for women under 50 and for women 50 years of age and older. Then, we compare the two age-adjusted estimates to assess effect measure modification by age as a proxy for menopausal status. A more sophisticated analysis would model the effect measure as a continuous function of age (e. g. , always increasing or always decreasing with age).

Example: Effect Modifier or Confounder or Both? For purposes of causal inference, we usually want to consider both confounding and effect measure modification in the analysis of observational data. Although the control of confounding is usually more important in a given study than is assessment of effect measure modification, the same covariates are frequently considered as both potential confounders and potential modifiers of the exposure effect. Nevertheless, even though confounding and effect measure modification are assessed statistically with some of the same techniques (e. g. , using stratification), these two concepts are very different and, therefore, have different implications to causal inference. The distinction between confounding and effect measure modification is particularly important to keep in mind when using models: n confounding by a variable may be well controlled by just a few model terms for that factor alone, n whereas modification of the exposure coefficient by the variable requires product terms entered into the model.

Effect Measure Modification: Examples EPI 200 B Winter 2008
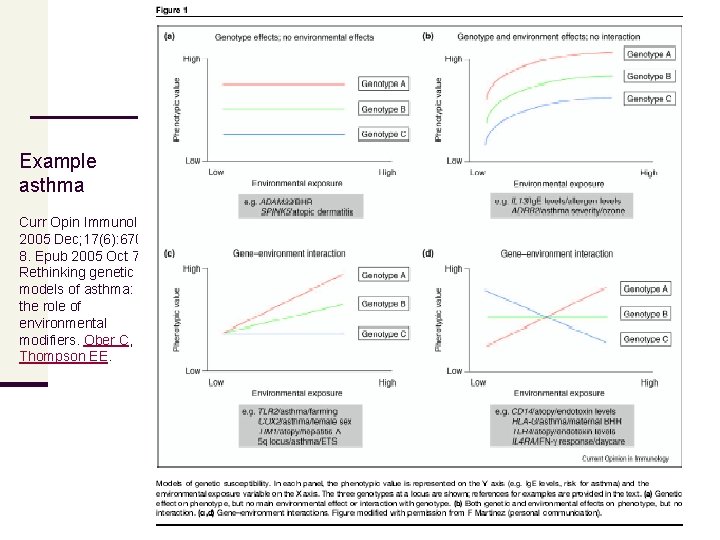
Example asthma Curr Opin Immunol. 2005 Dec; 17(6): 6708. Epub 2005 Oct 7. Rethinking genetic models of asthma: the role of environmental modifiers. Ober C, Thompson EE.
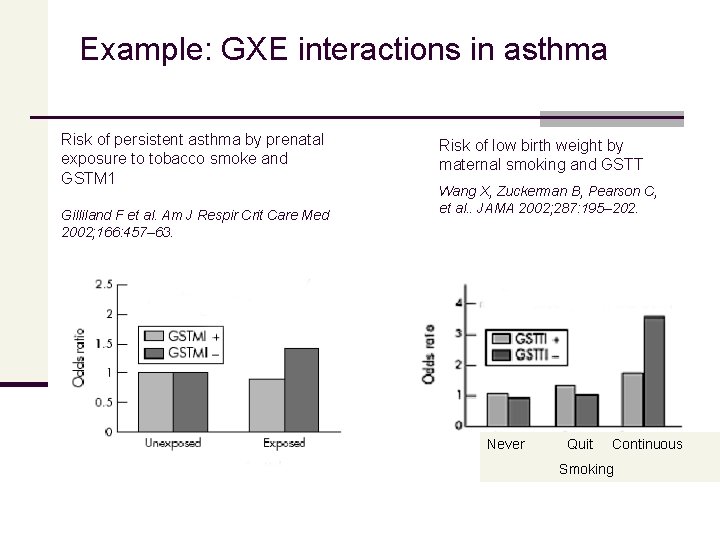
Example: GXE interactions in asthma Risk of persistent asthma by prenatal exposure to tobacco smoke and GSTM 1 Gilliland F et al. Am J Respir Crit Care Med 2002; 166: 457– 63. Risk of low birth weight by maternal smoking and GSTT Wang X, Zuckerman B, Pearson C, et al. . JAMA 2002; 287: 195– 202. Never Quit Continuous Smoking

Gene-Environment Interactions: Examples Source: Ottman R. Gene-Environment Interaction: Definitions and Study Designs. Preventive Medicine, 1996, 25: 764 -770 NOTE: The images provided to visualize the examples of ‘ geneenvironment interaction’ are NOT DAGs

Model A The genotype ‘produces’ or increases the environmental risk factor’s effect that can also be produced non-genetically Diet rich in PA environmental risk factor (ERF) genotype disease Example Phenylketonuria (PKU) – an autosomal recessive disease - can lead to mental retardation if dietary restrictions are not followed and blood phenylalanine levels become high. Infants can have PKU if • they are homozygous for PKU or • their mothers have untreated PKU leading to high phenylalanine blood levels an intrauterine exposure during pregnancy (note: fetus lacks enzyme activity ) High blood phenylalanine level (ERF) is the intervening (or intermediate) variable in Model A. The effect of the exposure to the ERF is the same in persons with and without the high risk genotype. There is no interaction in this model.

Model B The genotype exacerbates the effect of the ERF, but there is no effect of the genotype in those unexposed to the ERF genotype Example environmental risk factor (ERF) disease n Xeroderma pigmentosum - autosomal recessive disorder – in which excessive exposure to UV radiation (ERF) increases the risk for skin cancer. n Subjects with xeroderma lack an enzyme needed for DNA repair and, therefore, are at higher risk for skin cancer. n Model B shows a biologic interaction between genotype and ERF. The interaction for the xeroderma example is synergistic. This same model could be used to show antagonistic interaction where the genotype suppresses the effect of the ERF, but has no effect when acting alone

Model C The ERF exacerbates the effect of the genotype, but there is no effect of the exposure in persons with low-risk genotype environmental risk factor (ERF) disease Example n Porphyria variegata - autosomal dominant disorder – leading to skin problems ranging from sun sensitivity to a tendency to blister easily. n In individuals without the genotype, barbiturates are relatively innocuous. n If individuals with the genotype are exposed to barbiturates, they experience acute attacks that may involve death or paralysis. n Example shows an biologic interaction between genotype and ERF that is synergistic. Again, this same model could be used to show antagonistic interaction where the ERF suppresses the effect of the genotype, but has no effect when acting alone.

Model D Both the ERF and the genotype are required to increase disease risk genotype environmental risk factor (ERF) disease Example n Glocuse-6 -phosphate dehydrogenase (G 6 PD) deficiency - an x-linked recessive disorder – where most individuals are asymptomatic. n However, some develop severe hemolytic anemia if they eat fava beans. Note: Eating fava beans does not produce these reactions in people without the genotype. n Model D shows a biologic interaction between genotype and ERF that is synergistic. This same model could be used to show antagonistic interaction where the risk is reduced in persons with both ERF and genotype, but neither has an effect when acting alone.

Model E The ERF and the genotype each have similar effects on disease risk. When they occur together, risk is higher or lower than when they occur alone. genotype Example environmental risk factor (ERF) disease n Non-smokers with an α-1 -antitrypsin deficiency are at increased risk of developing chronic obstructive pulmonary disease (COPD) similar to smokers without an α-1 -antitrypsin deficiency. n Note: COPD risk is increased to an even greater extent in smokers WITH α-1 antitrypsin deficiency. n Model E shows a situation that may or may not involve an interaction between genotype and ERF. The choice of the scale of measurement will determine whether or not interaction is said to exist. For example, a report by Khoury et al. reported an additive effect of the aforementioned example. Model E can encompass antagonism, or a joint effect lower than that expected from a multiplicative or additive model.
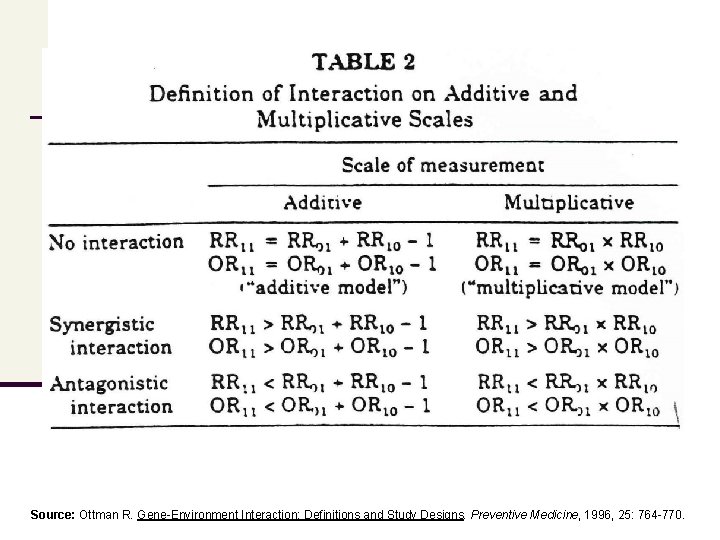
Source: Ottman R. Gene-Environment Interaction: Definitions and Study Designs. Preventive Medicine, 1996, 25: 764 -770.
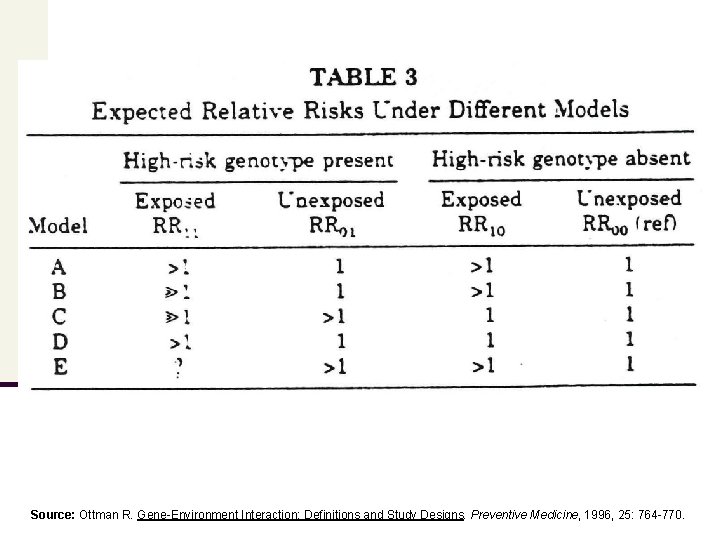
Source: Ottman R. Gene-Environment Interaction: Definitions and Study Designs. Preventive Medicine, 1996, 25: 764 -770.
- Slides: 58