EE 201 SEMICONDUCTOR DEVICES MOHD ZULHILMI JAAFAR JABATAN

EE 201 SEMICONDUCTOR DEVICES MOHD ZULHILMI JAAFAR JABATAN KEJURUTERAAN ELEKTRIK POLITEKNIK KUALA TERENGGANU

SEMICONDUCTOR MATERIAL � • The term conductor is applied to any material that will support a generous flow of charge when a voltage source of limited magnitude is applied across its terminals. � • An insulator is a material that offers a very low level of �conductivity under pressure from an applied voltage �source. � • A semiconductor (SC) therefore, is a material that has a conductivity level somewhere between the extremes of an insulator an a conductor.

ATOM STRUCTURE � • Bohr Atom Structure was first introduced by Niels Bohr (1913). � • Examine the structure of an atom; atom is composed of 3 basic particles: the electron, the proton, and the neutron. � • In the atomic lattice, the neutrons and protons form the nucleus, while the electrons revolve around the nucleus in a fixed orbit. � • The Bohr models of the 2 most commonly used semiconductor (SC): Ge and Si are shown as followed: Silicon Germanium Figure 1. 1

COVALENT BONDING �Covalent bonds are formed bythe sharing of valence electrons with neighboring atoms. �Fig. 1. 1 shows how each Si atom with four adjacent atoms to form a Si crystal. A Si atom with its 4 valence e- shares an e- with each of its 4 neighbour. �This effectively creates 8 valence e- for each atom & produces a state of chemical stability.
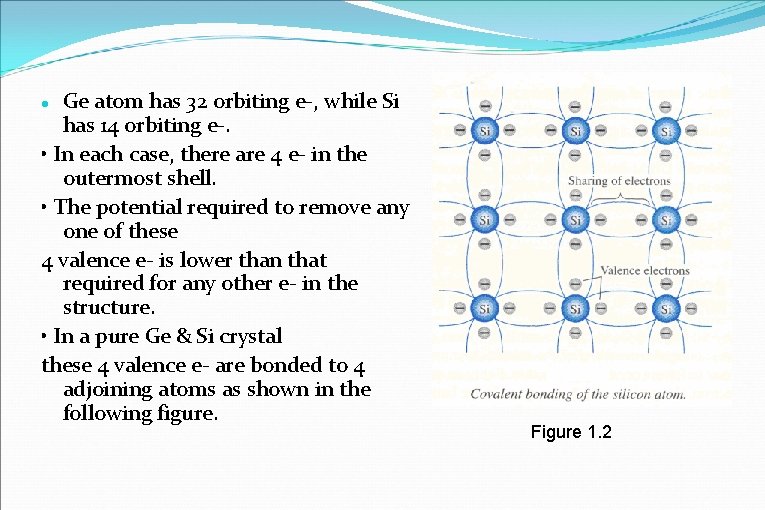
Ge atom has 32 orbiting e-, while Si has 14 orbiting e-. • In each case, there are 4 e- in the outermost shell. • The potential required to remove any one of these 4 valence e- is lower than that required for any other e- in the structure. • In a pure Ge & Si crystal these 4 valence e- are bonded to 4 adjoining atoms as shown in the following figure. Figure 1. 2

CONDUCTION ELECTRONS & HOLES �When e- jumps to the CB, a vacancy is left in the valence band within the crystal. This vacancy is called a hole. �Hole also referred as positive charge carrier. �Foe every e- raised to the CB by external energy, there is 1 hole left in the valence band, creating what is called electron–hole pair (EHP). �Recombination occurs when a CB e- loses energy & falls back into a hole in the valence band. �To summarize, a peace of intrinsic Si at room temperature has, at any instant a no. of CB (free) e- that are unattached to any atom & are essentially drifting randomly throughout the material. Also an equal no. of holes are created in the valence band when these electrons jump into the CB
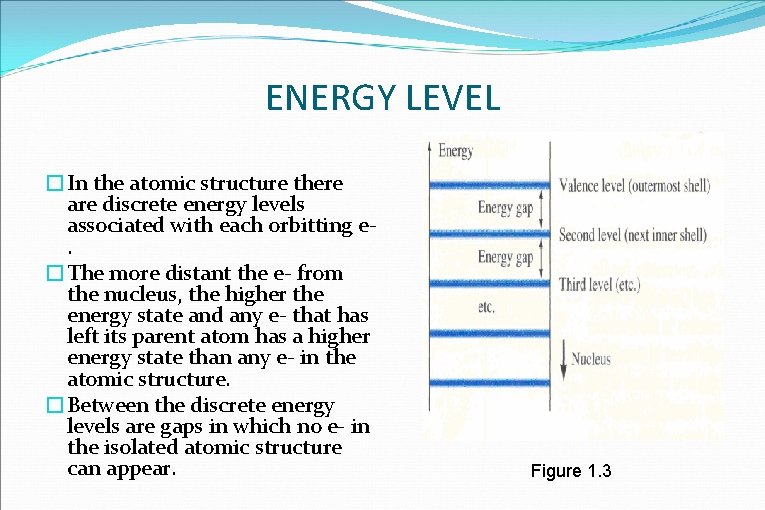
ENERGY LEVEL �In the atomic structure there are discrete energy levels associated with each orbitting e. �The more distant the e- from the nucleus, the higher the energy state and any e- that has left its parent atom has a higher energy state than any e- in the atomic structure. �Between the discrete energy levels are gaps in which no e- in the isolated atomic structure can appear. Figure 1. 3

�Note that there are boundary levels & max. energy states in which any e- in the atomic lattice can find itself, and there remains a forbidden region between the valence band & the ionization level. �Recall that ionization is the mechanism whereby an e- can absorb sufficient energy to break away from atomic structure & enter the conduction band (CB). �Energy associated with each e- is measured in e. V 4 �Since energy is also measured in joules & the charge of one e- =1. 6 x 10 -19 coulomb, W = QV = (1. 6 x 10 -19 C) (1 V) Q= charge associated with single current * 1 e. V = 1. 6 x 10 -19 J
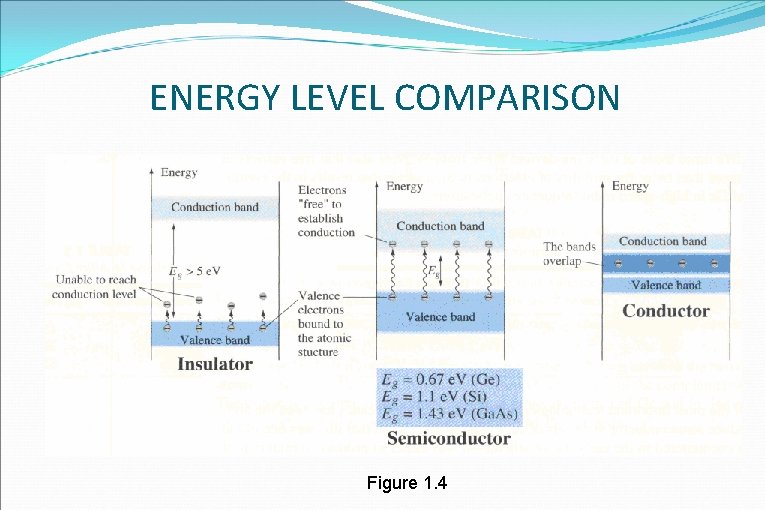
ENERGY LEVEL COMPARISON Figure 1. 4

INSULATOR �The energy diagram for the insulator shows the insulator with a very wide energy gap. The wider this gap, the greater the amount of energy required to move the electron from the valence band to the conduction band. �Therefore, an insulator requires a large amount of energy to obtain a small amount of current. The insulator "insulates” because of the wide forbidden band or energy gap.

SEMICONDUCTOR �The semiconductor, on the other hand, has a smaller forbidden band requires less energy to move an electron from the valence band to the conduction band. �Therefore, for acertain amount of applied voltage, more current will flow in the semiconductor than in the insulator.

CONDUCTOR/METAL �The last energy level is for a conductor. �Notice, there is no forbidden band or energy gap and the valence and conduction bands overlap. �With no energy gap, it takes a small amount or energy gap and the valence and conduction bands overlap. �With no energy gap, it takes a small amount easily.

INTRINSIC MATERIAL �Intrinsic material is the pure semiconductor that has no any additional elements. Examples : Silicon & Germanium. �Intrinsic Semiconductor has no advantages.

EXTRINSIC MATERIAL �Because Si is the material used most frequently as the base material in construction of solid state electronic device, the discussion to follow in this & the next few sections deals solely with Si SC. �Extrinsic Material: material that has been subjected to the doping process. �There are 2 types of Extrinsic Material: n-type & p-type. �Both materials are formed by adding a predetermined no. of impurity atoms to a Si base. �n-type: created by introducing impurity elements that have 5 valence e- (eg. As & P). �p-type: formed by doping a pure Ge or Si crystal with impurity atoms having 3 valence e- (eg. B, Al, In & Ga).

N-TYPE MATERIAL � Happens when pure semiconductor (exp: Silicon) doped with pentavalent atom (has 5 e. v)(refer Fig. 1. 3): � Four out of five electron valence from pentavalent atom formed covalent bond with silicon atoms but there is an electron that has no pair � This electron will escape from its orbit and become free electron, negative current carrier. More pentavalent atoms more free electrons, thus more negative current carriers. Therefore this material is called N-type material. � At very low temperature, majority current carriers are free electrons. � At room temperature, a few holes produced and called minority current carriers Si Si Si Ar Si Si Free Electron from arsenic atom that has no pair Figure 1. 5 Silicon doped with Arsenic

P-TYPE MATERIAL � P- type material formed when pure semiconductor doped with trivalent atom. (Figure 1. 4) � Three electron valences form covalent bond but another one has no pair. This situation creates hole, positive current carrier. � More trivalent atoms, more holes, thus more negative current carriers, therefore this material is called P-type material. � At very low temperature, majority current carriers are holes and at very high temperature, minority current carriers are electrons Si Si Si In Si Si Figure 1. 6 Silicon doped with Indium hole

PN JUNCTION �One of the crucial keys to solid state electronics is the nature of the P-N junction. When p-type and n-type materials are placed in contact with each other, the junction behaves very differently than either type of material alone. �Specifically, current will flow readily in one direction (forward biased) but not in the other (reverse biased), creating the basic diode. This non-reversing behavior arises from the nature of the charge transport process in the two types of materials.
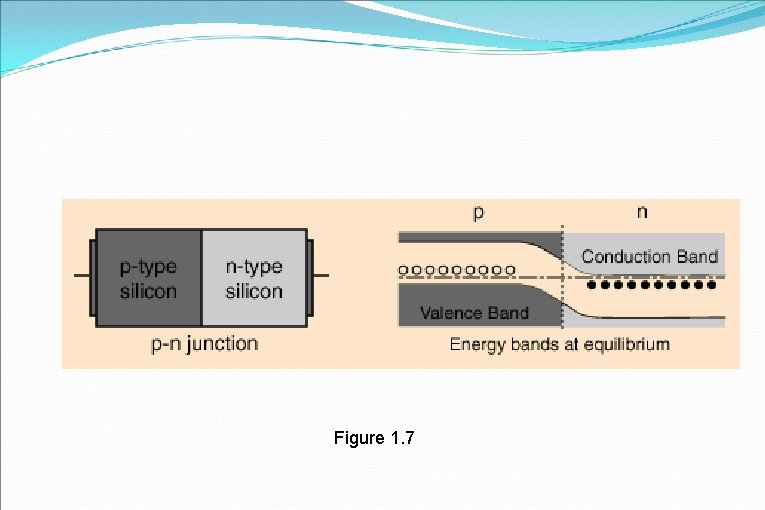
Figure 1. 7

�The open circles on the left side of the junction above represent "holes" or deficiencies of electrons in the lattice which can act like positive charge carriers. �The solid circles on the right of the junction represent the available electrons from the n-type dopant. �Near the junction, electrons diffuse across to combine with holes, creating a "depletion region".

DEPLETION REGION �When a p-n junction is formed, some of the free electrons in the n-region diffuse across the junction and combine with holes to form negative ions. In so doing they leave behind positive ions at the donor impurity sites. Figure 1. 8

THRESHOLD VOLTAGE The process of combination electrons and holes continues until threshold voltage produced, 0. 7 V for silicon and 0. 3 V for germanium, Figure 1. 9

PN JUNCTION BIAS �Voltage is supplied to both ends of the junction to give bias. �There are two types of bias, forward bias and reverse bias

FORWARD BIAS � Forward bias is shown in Figure 1. 10 where negative probe power supply is connected to n-type material and positive probe power supply is connected to p-type material. � This situation will decrease the size of depletion region. This occurs because negative probe power supply will push the electrons in n-type material and the positive probe power supply will push holes in p –type material. � The current produced is called forward current. Figure 1. 10

�Figure 1. 11 shows both current carries move and finally surpassing the voltage limit (threshold voltage) at the depletion region. �Power supply has to surpass the voltage limit, 0. 7 V for silicon and 0. 3 V for germanium before flowing Figure 1. 11

REVERSE BIAS � Figure 1. 12 shows PN junction is reverse-biased where negative probe power supply is connected to p-type material and positive probe power supply is connected to n-type material. � This situation will increase the size of depletion region. This occurs because negative probe power supply will attract the electrons in n-type material and the positive probe power supply will attract holes in p –type material. � There is a very small current produced which is called reverse current but can be ignored. � When the power supply is increased instantly, it will break the junction and will blow the component. The voltage is called breakdown voltage. Figure 1. 12
- Slides: 25