Education Phase 4 Food labelling Labelling Prepackaged foods

Education Phase 4 Food labelling

Labelling Pre-packaged foods have information on their labels which can help consumers choose between different foods, brands, or flavours. Much of the information must be provided by EU law. • Directive 2000/13/EC on labelling, presentation and advertising of foods applies until 12/12/2014 • Regulation (EU) No 1169/2011 on the provision of food information to consumers will apply from 13/12/2014 (sometimes referred to as EU FIC)

Information for consumers The following information must appear by law on food labels: • the name of the food; • weight or volume; • ingredient list; • allergen information; • genetically modified (GM) ingredients; • date mark and storage conditions; • preparation instructions; • name and address of manufacturer, packer or seller; • place of origin; • lot (or batch) mark; • nutrition information (from 2016 onwards). Additional information may also be provided, such as cooking instructions or serving suggestions.

Nutrition information At the moment, foods and drinks do not have to provide nutrition information on packaging (unless they make a nutrition or health claim about the product). Where information is given, some rules have to be followed. However, provision of information will become compulsory from 13 th December 2016 and there will be some small changes to the format required.

Current labelling regulations in Europe The current rules specify the nutrients that can be included. The information has to be presented per 100 g/ml, but could also be provided per portion. Format 1: ‘Big 4’ Energy Protein Carbohydrate Fat (k. J and kcal) (g) (g) Format 2: ‘Big 4 and Little 4’ Energy (k. J and kcal) Protein (g) Carbohydrate (g) of which sugars (g) Fat (g) of which saturates (g) Fibre (g) Sodium (g) Further information can be added to labels such as the amounts of polyunsaturates, monounsaturates, starch, cholesterol, vitamins or minerals.

New labelling regulations in Europe A new Regulation covering all aspects of food labelling, including the size of the letters on packaging, came into force at the end of 2011. It will make providing nutrition information on the back of packaged foods compulsory from 2016 (previously it was optional unless a nutrition or health claim was made). The format of nutrition labels will change slightly under the new rules and companies can start to use the new format straightaway, although the old rules (known as the Food Labelling Regulations) remain legal until 2014.
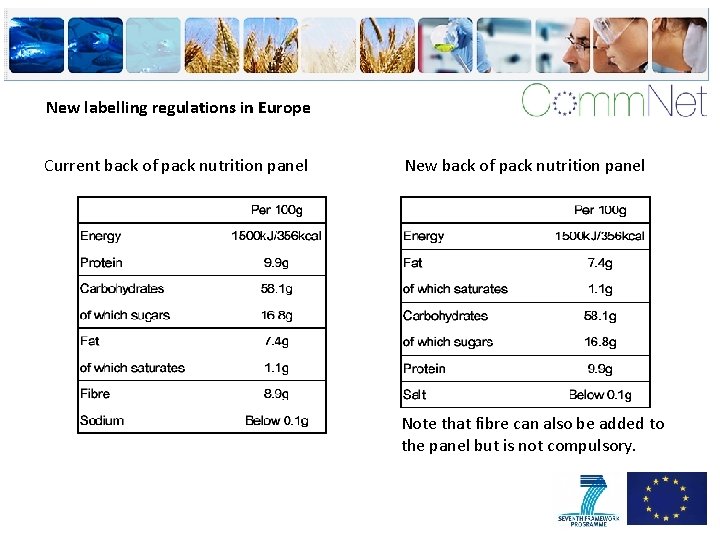
New labelling regulations in Europe Current back of pack nutrition panel New back of pack nutrition panel Note that fibre can also be added to the panel but is not compulsory.

New labelling regulations in Europe - Reference intakes The new regulations feature Reference Intakes (RIs). RIs replace what used to be called Guideline Daily Amounts (GDAs) in the UK. It should be noted that the notion of RIs is different from the notion of guideline daily amount, as the term 'reference intake' does not imply a nutritional advice unlike the term 'guideline'. There is no nutritional advice to consume, for example, 20 g of saturated fat per day and consumers should not believe it is a minimum quantity necessary to maintain health. RIs for energy and other selected nutrients are shown is the chart. Nutritional information can be provided as a percentage of the RI. Energy or nutrient Reference Intake Energy 8400 k. J/2000 kcal Total fat 70 g Saturates 20 g Carbohydrate 260 g Sugars 90 g Protein 50 g Salt 6 g

New labelling regulations in Europe Nutrients will have to be listed in a certain order – energy (both in k. J and kcal) and the amounts of fat, saturates, carbohydrates, sugars, protein and salt (in place of ‘sodium’ which is no longer permitted). The information has to be given in per 100 g/ml but can additionally be given: • Per portion or per consumption unit (e. g. per slice) • As % Reference Intakes Information of the following nutrients can be provided in a voluntary basis: mono and poly-unsaturates, polyols, starch and fibre. Information on certain approved vitamins and minerals can be listed if present in significant amounts. If a nutrition claim is made it becomes mandatory to give information about that nutrient even if it is not listed in the regulation. For example if the claim ‘high in omega 3’ was made then the amount of omega 3 per 100 g would have to be given.

Front of pack nutrition labelling Providing front of pack nutritional information remains voluntary, however many food manufacturers choose to display nutritional information on the front of prepackaged food and drinks. Front of pack labels allows consumers to: • Check what their food contains – how much energy, fat, saturates, sugar and salt the food provides, plus how much of their Reference Intake this contributes to; • Compare foods they buy with other similar foods; • Choose the foods that best suit their needs. Each serving of Edam (30 g) contains: Energy 400 k. J 95 kcal Fat Saturates Sugars Salt 7. 0 g 4. 7 g 0 g 0. 8 g 5% 10% 24% <1% 13% of an adult’s Reference Intake Typical values (as sold) per 100 g: Energy 1325 k. J/320 kcal

Nutrition and health claims The Regulation (EC) No 1924/2006 on nutrition and health claims is enforced across the EU. The regulation implements measures to ensure that any claim made on food’s labelling, presentation or marketing is clear, accurate and based on evidence accepted by the whole scientific community. Foods bearing claims which could potentially mislead consumers are eliminated by the market. In order to bear claims, foods have to have appropriate nutrient profiles which are set. This regulation helps to enhance the consumers ability to make informed and meaningful choices.

Nutrition and health claims General claims about benefits to overall good health, such as ‘healthy’ or ‘good for you’, will only be allowed to be used if accompanied by an appropriate and approved claim. This means that more general claims must be backed up by an explanation of why the food is ‘healthy’ or what makes it a ‘superfood’. Labels are not allowed to claim that food can treat, prevent or cure any disease of medical condition. These sorts of claims can only be made of licenced medicines.

Nutrition claims A nutrition claim is any claim which states, suggests or implies that a food or drink has particular beneficial nutritional properties. There are certain conditions around the usage of each nutrition claim. Examples of approved nutrition claims: • Low fat (less than 3 g of fat per 100 g food); • High fibre (at least more than 6 g of fibre per 100 g food); • Reduced sugar (30% less than the original product); • Source of vitamin C (at least 15% of the recommended daily allowance for vitamin C).

Health claims A health claim is a statement about the positive effect a product can have on health. Claims appearing on food labelling have to be approved by the European Food Safety Authority (EFSA), who evaluate the scientific evidence supporting health claims. Examples of health claims include: • Calcium is important for the maintenance of normal bones • Beta-glucans contribute to the maintenance of normal blood cholesterol levels. • Iron contributes to the reduction of tiredness and fatigue.

Types of health claim There are three different types of health claims: • ‘Function’ or Article 13 health claims. These health claims either relate to the growth, development and functions of the body, refer to psychological and behavioural functions or refer to slimming and weight control. • ‘Risk reduction claims’ or Article 14(1)(a) health claims. These are health claims on reducing a risk factor in the development of a disease. • Health claims referring to children’s development or Article 14 (1)(b) claim.

Current allergen labelling in Europe The current EU regulation establish a list of 14 food allergens, which have to be indicated by reference to the source allergen whenever they, or ingredients made from them, are used at any level in pre-packed foods. The list consists of: • Cereals containing gluten • Crustaceans • Molluscs • Eggs • Fish • Peanuts • Nuts • Soybeans • Milk • Celery • Mustard • Sesame • Lupin • Sulphur dioxide – at levels above 10 mg/kg or 10 mg/litre

New allergen labelling in Europe Following the implementation of Regulation (EU) No. 1169/2011, allergen labelling rules will be changing in December 2014. The new regulation, will build on current allergen labelling provisions for prepacked foods. It will introduce a new requirement for allergen information to be provided for foods sold nonpacked or prepacked for direct sale. Evidence suggests that most food allergy incidents can be traced back to non-prepacked food, therefore information on potential allergens should always be provided to the consumer.
- Slides: 17