EDOXABAN FOR THE TREATMENT OF VENOUS THROMBOEMBOLISM Ambulatory

EDOXABAN FOR THE TREATMENT OF VENOUS THROMBOEMBOLISM Ambulatory Journal Club Lien-Thanh Kratzke September 2013

Outline • • Case Presentation Clinical Question Intro/Background Journal Article Critical Appraisal Limitations Clinical Question Revisited Conclusion

Case Presentation

My Patient: Presentation • 54 year old female • Endometrial cancer 2008 treated with chemotherapy and TAH+BSO • 9/15 -9/20/13: GI bleed with blood in emesis and stool. Found to have bleeding duodenal mass from recurrent metastatic endometrial carcinoma. • 9/22 -9/25/13: Left leg swelling. Found to have bilateral lower extremity DVT's.

My Patient: DVT Treatment • Heparin drip, followed by coumadin and lovenox • Discharged from hospital QEC to establish care and for INR checks • Does not want to stay on lovenox because does not like injections
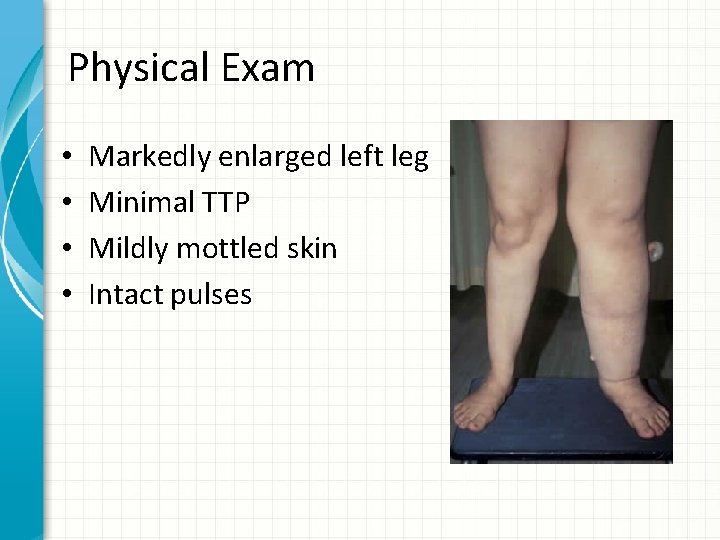
Physical Exam • • Markedly enlarged left leg Minimal TTP Mildly mottled skin Intact pulses

Clinical Question In my patient with deep vein thromboses and increased bleeding risk, what is the role of Edoxaban as a treatment option?

Background

VTE Treatment • Standard: low-molecular-weight heparin followed by vitamin K antagonists • Warfarin has slow onset of action, unpredictable dose, multiple interactions • New oral anticoagulants being studied as alternatives – Direct Thrombin Inhibitors (Dabigatran) – Direct Factor Xa Inhibitors (Rivaroxaban, Apixaban, Edoxaban)

Oral Anticoagulants: FDA Approval • Dabigatran (Pradaxa): – Nonvalvular a-fib (Oct 2010) • Rivaroxaban (Xarelto): – Postoperative prophylaxis after hip or knee replacement (July 2011) – Nonvalvular a-fib (Nov 2011) – Treatment/prophylaxis of DVT and PE (Nov 2012) • Apixaban (Eliquis): – Nonvalvular a-fib (Dec 2012)

Edoxaban • Direct Factor Xa inhibitor • Once daily medication • Approved in Japan for prevention of VTE after major orthopedic surgery (July 2011)

• Reviewed phase 3 clinical trials of new oral anticoagulants from 1/2007 -2/2013 • Apixaban, rivaroxaban, dabigatran • Favorable clinical profile in orthopedic surgery, a-fib, acute coronary syndrome • Increased risk of bleeding in critically ill • Further studies are necessary to determine long term safety

Journal Article

September 1, 2013 DOI: 10. 1056/NEJMoa 1306638
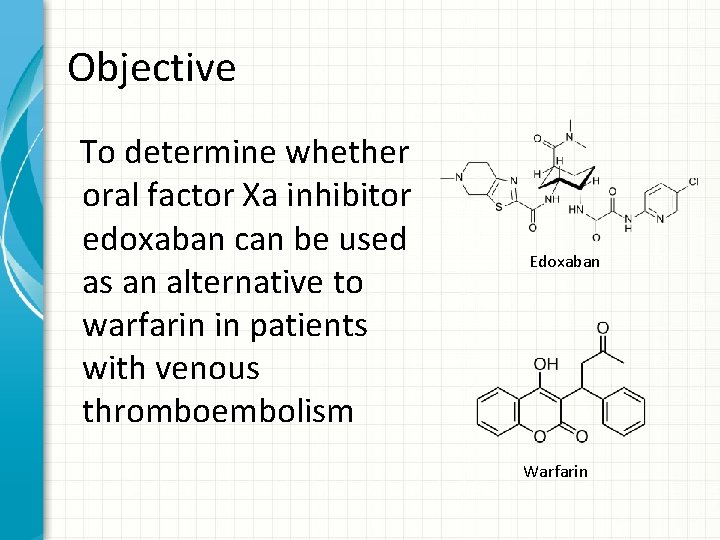
Objective To determine whether oral factor Xa inhibitor edoxaban can be used as an alternative to warfarin in patients with venous thromboembolism Edoxaban Warfarin

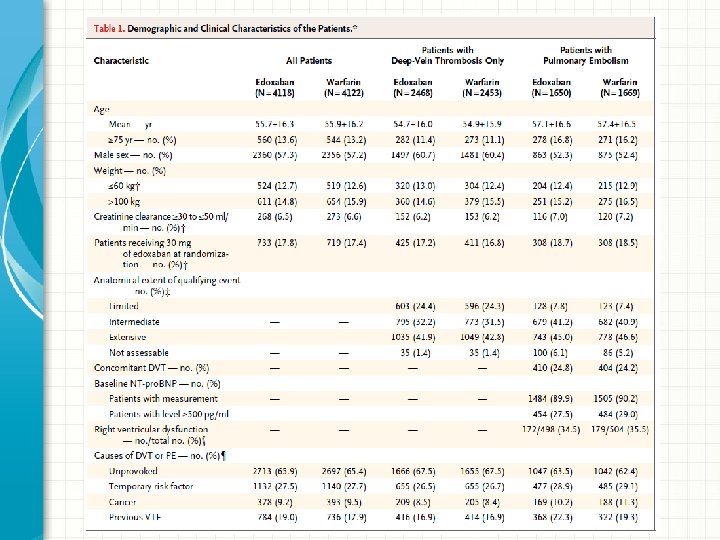
Methods: Randomization • Randomized, double-blind, non-inferiority • Assigned patients with acute VTE: • Edoxaban 30 mg or 60 mg daily (lower dose for creatinine clearance 30 -50 m. L/min or body weight <60 kg) • Warfarin (dose adjusted for INR goal 2 -3) • Stratified by diagnosis (DVT vs PE), presence or absence of temporary risk factors, and dose of Edoxaban

Methods: Treatment • Initially received heparin or lovenox for at least 5 days • Received study drug for 3 -12 months, duration depended on treating physician and patient preference • Follow up: 12 months • Assessment on days 5 -12, day 30, day 60, monthly (while taking study drug) or q 3 months (if not taking study drug)
Patient Population • 18 years or older • Objectively diagnosed with at least one of the following: • Acute, symptomatic DVT involving popliteal, femoral, or iliac veins • Acute, symptomatic pulmonary embolism

Exclusion Criteria • • Contraindications to heparin or warfarin Received >48 hours therapeutic heparin Received >1 dose vitamin K antagonist Cancer for which long-term treatment with low -molecular-weight heparin was anticipated Another indication for warfarin therapy On ASA >100 mg/day On dual antiplatelet therapy Creatinine clearance <30 ml/minute
End Points • Primary efficacy outcome: recurrent symptomatic venous thromboembolism • Principal safety outcome: major or clinically relevant nonmajor bleeding

Are the Results Valid? • Randomized controlled double-blind • Supported by Daliichi-Sankyo, which is developing Edoxaban

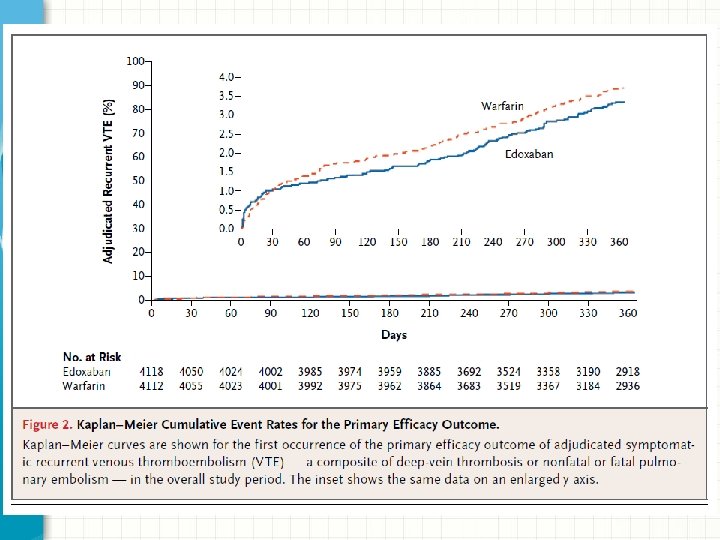
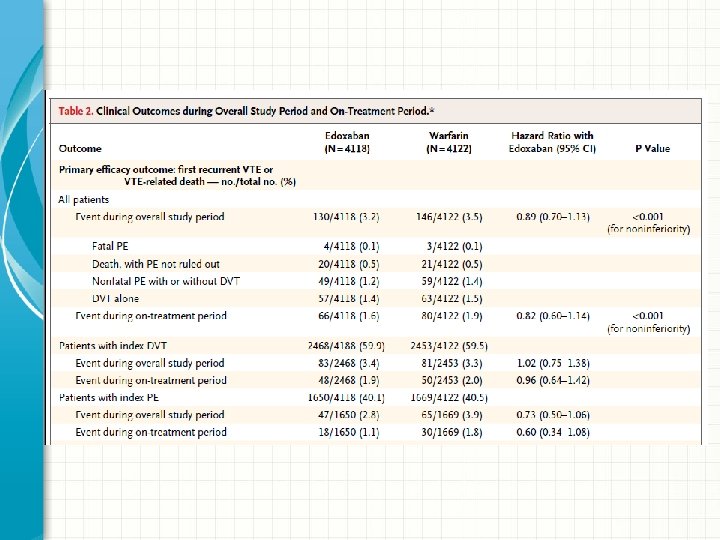
Results: Primary Efficacy • Primary efficacy outcome: recurrent symptomatic venous thromboembolism • Edoxaban was non-inferior to warfarin – Edoxaban: 130 patients (3. 2%) – Warfarin: 146 patients (3. 5%) – Hazard ratio 0. 89; 95% CI 0. 70 -1. 13; P<0. 001 for noninferiority
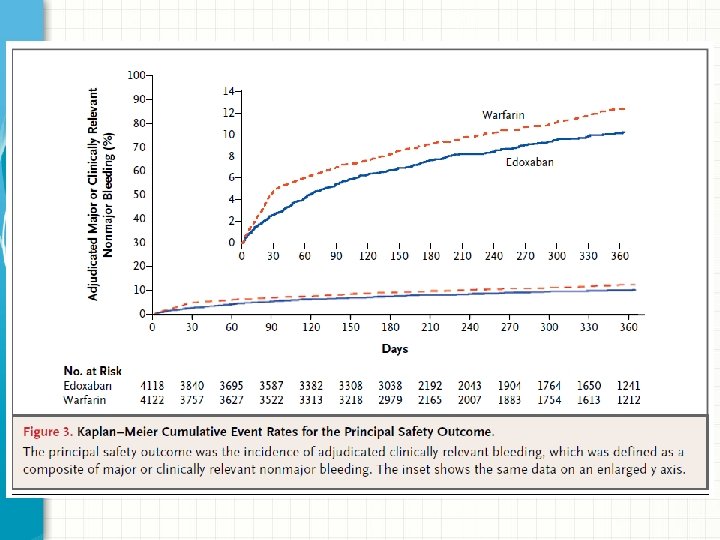
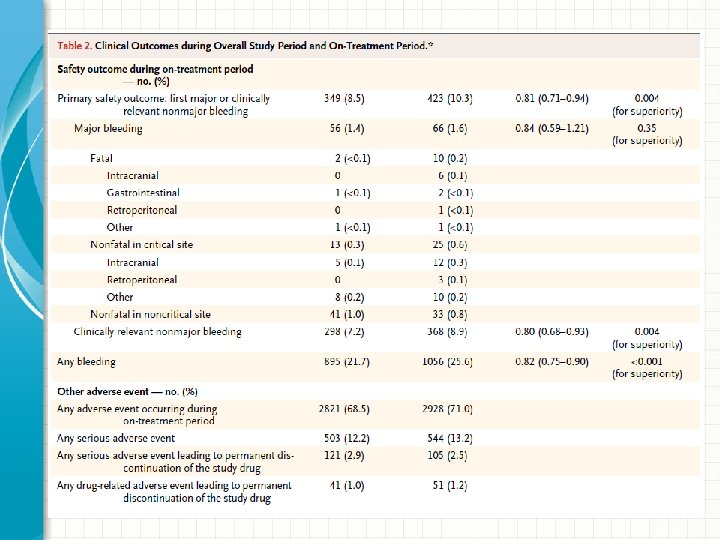
Results: Principal Safety • Principal safety outcome: major or clinically relevant non-major bleeding • Edoxaban was associated with less bleeding than warfarin – Edoxaban: 349 patients (8. 5%) – Warfarin: 423 patients (10. 3%) – Hazard ratio 0. 81; 95% CI 0. 71 -0. 94; P=0. 004 for superiority

Results: INR Checks • For patients who received warfarin, INR was checked at least monthly: – INR in therapeutic range: 63. 5% of the time – INR >3: 17. 6% of the time – INR <2: 18. 9% of the time

Limitations • Patients were initially treated with either enoxaparin or heparin, for different lengths of time • Different treatment durations ranged from 3 -12 months • INR not always in therapeutic range

Clinical Question Revisited In my patient with deep vein thromboses and increased bleeding risk, what is the role of Edoxaban as a treatment option?

Our Patients • Generalizable: maybe – Average 56 -57 – Men and women – Race/ethnicity? • Compliance: poor – Once a day oral medication – However, cost is very high

Conclusions • Efficacy and safety in real life? – No standard reliable way to measure anticoagulant effect – Can’t reverse effects • Cost is prohibitive • Not yet FDA approved

THANKS!
- Slides: 37