Edexcel GCSE Core Science C 1 b Topic

Edexcel GCSE Core Science C 1 b Topic 6 Making Changes Foundation Tier

Chemicals in Food A natural substance in food is a substance that exists in nature, e. g. water, glucose, sodium chloride. An artificial substance in food is a chemical that has been manufactured e. g. tartrazine (yellow colouring). Sometimes there is no chemical difference between artificial substance and a natural substance (e. g. water can be made but is also in nature). Both natural and artificial foods can have health impacts. Natural vanilla pods contain vanillin mixed with other substances. Artificial vanillin is used in ice cream – it tastes different to the natural vanilla because it does not have the other substances. Sugar is a carbohydrate which the body uses for fuel. It causes tooth decay and contains calories. Aspartame and saccharin are artificial sweeteners – they taste like sugar but they are chemically different. They do not contain any calories. Cooking – Cooking food produces new substances. A chemical change has taken place – you cannot change it back as the change is irreversible. Eggs and meat contain protein. When cooked the chemical bonds change shape. Potatoes are a good source of carbohydrate but each cell is surrounded by a cell wall of cellulose. Humans cannot digest cellulose but cooking breaks down the cell walls so that we can.

Other household chemicals Sodium hydroxide (caustic soda) - Manufacture of paper, textiles, soaps and detergents Citric acid - Soft drinks (e. g. Lemonade) and laxatives Ammonia - Fertilisers, bleaches, dyes and oils/fats Carbohydrates - Fats, plastics, photographic film, emulsions and adhesives Carbon dioxide - Fizzy drinks, baking, fire extinguishers, aerosols Water - Lubricants, future energy production, living! Hydrochloric acid - Production of other chemicals, digestion Phosphoric acid - Cleaning and rust-proofing, acidifying jellies and drinks (Cola) Sodium chloride - Seasoning for foods, preservative Ethanoic acid (acetic acid) - Food preservative, condiment

Acids, Bases and Neutralisation p. H 1 2 3 4 5 6 7 8 9 10 11 12 13 14 Acids Neutral Alkalis The p. H scale is a sliding scale from very strong acids (p. H 1) e. g. stomach acid or battery acid to very strong bases (p. H 14) e. g. caustic soda, with neutral (p. H 7) in the middle. Neutral substances turn green with universal indicator (UI). Weak acids (p. H 4 or 5) such as rain turn yellow/orange. Stronger acids e. g. vinegar, lemon juice, stomach acid turn red with UI. Weak alkalis e. g. washing up liquid and soap (p. H 8 -10) turn greeny blue. Stronger alkalis e. g. oven cleaner and ammonia turn UI purple. When an acid and a base react together they neutralise. A salt + water are formed. Salts are v. useful. They can be used in fertilisers (e. g. ammonium nitrate), fireworks (e. g. potassium nitrate), colouring agents in paint (e. g. copper salts for blue/green paint) and to aid the combustion of fuels (e. g. ammonium nitrate, potassium chlorate)
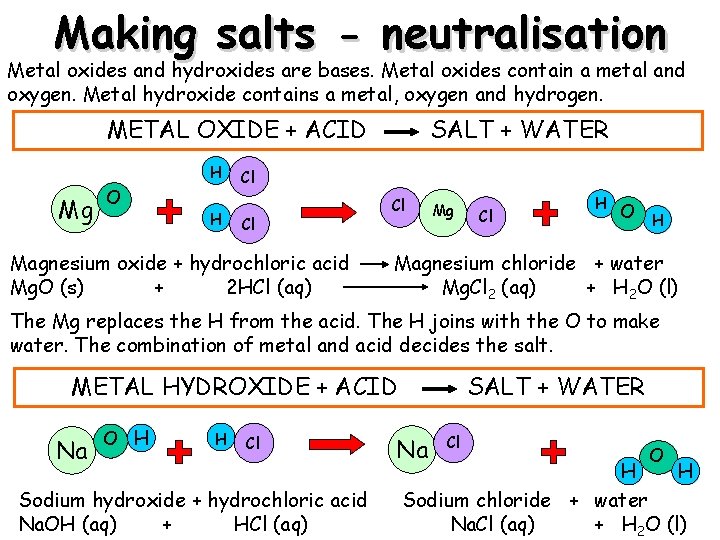
Making salts - neutralisation Metal oxides and hydroxides are bases. Metal oxides contain a metal and oxygen. Metal hydroxide contains a metal, oxygen and hydrogen. METAL OXIDE + ACID H Mg Mg O H SALT + WATER Cl Cl Magnesium oxide + hydrochloric acid Mg. O (s) + 2 HCl (aq) Cl Mg Cl H O H Magnesium chloride + water Mg. Cl 2 (aq) + H 2 O (l) The Mg replaces the H from the acid. The H joins with the O to make water. The combination of metal and acid decides the salt. METAL HYDROXIDE + ACID Na O H H Cl Sodium hydroxide + hydrochloric acid Na. OH (aq) + HCl (aq) SALT + WATER Na Cl O H H Sodium chloride + water Na. Cl (aq) + H 2 O (l)
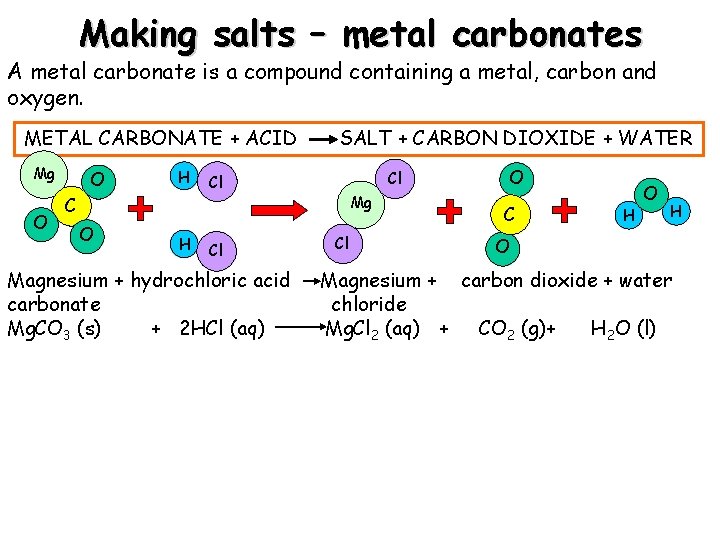
Making salts – metal carbonates A metal carbonate is a compound containing a metal, carbon and oxygen. METAL CARBONATE + ACID Mg O O H C O H SALT + CARBON DIOXIDE + WATER Cl Cl Cl Magnesium + hydrochloric acid carbonate Mg. CO 3 (s) + 2 HCl (aq) Mg Cl O C H O Magnesium + carbon dioxide + water chloride Mg. Cl 2 (aq) + CO 2 (g)+ H 2 O (l)
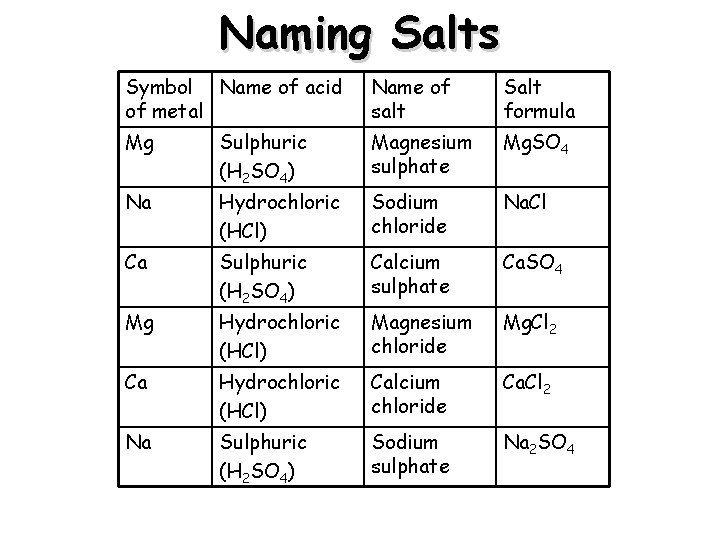
Naming Salts Symbol Name of acid of metal Name of salt Salt formula Mg Sulphuric (H 2 SO 4) Magnesium sulphate Mg. SO 4 Na Hydrochloric (HCl) Sodium chloride Na. Cl Ca Sulphuric (H 2 SO 4) Calcium sulphate Ca. SO 4 Mg Hydrochloric (HCl) Magnesium chloride Mg. Cl 2 Ca Hydrochloric (HCl) Calcium chloride Ca. Cl 2 Na Sulphuric (H 2 SO 4) Sodium sulphate Na 2 SO 4

Insoluble salts and Precipitates To make an insoluble salt you need to mix two solutions of soluble salts. All nitrate salts are soluble. Two solutions of soluble salts Lead nitrate + sodium chloride Precipitate of insoluble salt forms and eventually settles on the bottom Lead chloride + sodium nitrate Q How would you make a pure, dry sample of barium sulphate (Ba. SO 4). You can use the following: barium iodide, barium nitrate, potassium chloride and potassium sulphate. Answer : 1. Mix barium nitrate and potassium sulphate (both soluble) 2. Filter the precipitate of barium nitrate 3. Wash with distilled water 4. Dry it on filter paper

Hazard signs Flammable Corrosive h i Harmful Irritant Toxic Oxidising

Hydration, Thermal Dehydration and Decomposition Hydration reactions have water as a reactant e. g. calcium oxide (quicklime) reacts with water to produce calcium oxide (slaked lime). Calcium oxide + water calcium hydroxide Dehydration reactions have water as a product e. g. concentrated sulphuric acid is a strong dehydrating agent. It acts as a catalyst to convert sugar into water and a spongy lump of carbon Sucrose Conc. Sulphuric acid carbon + water Thermal decomposition reactions are when a substance breaks down into simpler substances when heated. Carbonates and hydrogen carbonates release CO 2 when heated Calcium carbonate Calcium oxide + carbon dioxide Sodium hydrogen carbonate Sodium carbonate + carbon dioxide + water This is in baking powder. When heated it produces CO 2 which makes your cake rise.

Metal Ores and Extraction Some metals e. g. gold are unreactive. They are found uncombined as lumps in the ground. Other more reactive metals e. g. iron react with other elements to form compounds called metal ores which can be extracted from the Earth’s crust. The more reactive the metal the harder it is to extract the metal from the ore. Lots of common metals e. g. Aluminium and Iron react with oxygen to form oxides. This reaction is called OXIDATION. The reaction that separates the metal from the oxide is called a REDUCTION reaction. Formation of a metal ore: OXIDATION Addition of O 2 Iron + Oxygen Iron Oxide 4 Fe + 3 O 2 2 Fe 2 O 3 Extraction of metal: REDUCTION Loss of O 2 Aluminium oxide Aluminium + Oxygen 2 Al 2 O 3 4 Al + 3 O 2 The most common type of reduction reaction uses carbon to separate the oxygen but this does not work for all metals.
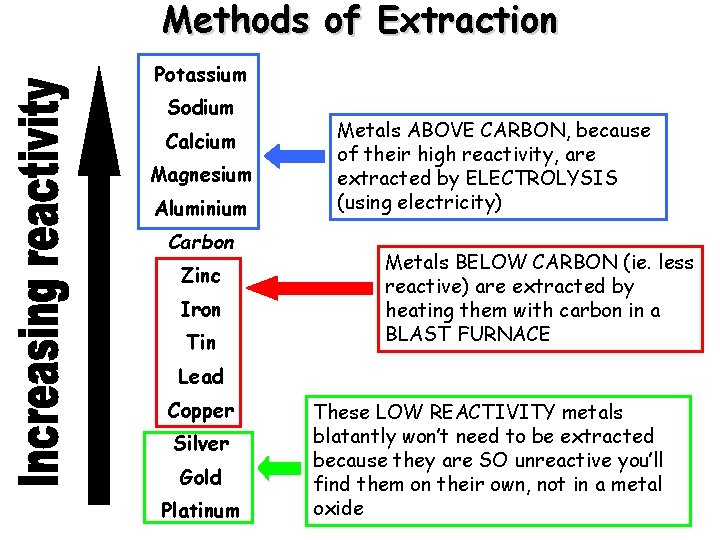
Methods of Extraction Potassium Sodium Calcium Magnesium Aluminium Carbon Zinc Iron Tin Metals ABOVE CARBON, because of their high reactivity, are extracted by ELECTROLYSIS (using electricity) Metals BELOW CARBON (ie. less reactive) are extracted by heating them with carbon in a BLAST FURNACE Lead Copper Silver Gold Platinum These LOW REACTIVITY metals blatantly won’t need to be extracted because they are SO unreactive you’ll find them on their own, not in a metal oxide
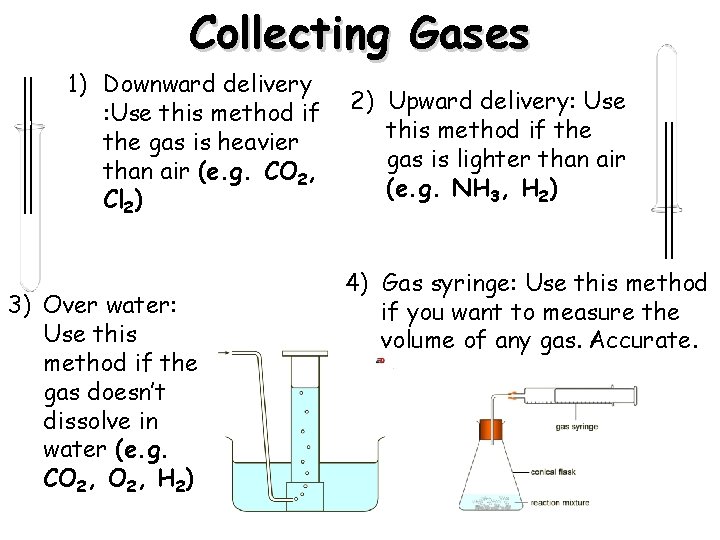
Collecting Gases 1) Downward delivery : Use this method if the gas is heavier than air (e. g. CO 2, Cl 2) 3) Over water: Use this method if the gas doesn’t dissolve in water (e. g. CO 2, H 2) 2) Upward delivery: Use this method if the gas is lighter than air (e. g. NH 3, H 2) 4) Gas syringe: Use this method if you want to measure the volume of any gas. Accurate.

Testing for Gases Hydrogen: Place a lighted splint over the mouth of the test tube – hydrogen will make a squeaky pop “POP” Oxygen: Place a glowing splint over the mouth of a test tube – oxygen will relight the splint Carbon dioxide: Turns limewater from clear to milky Ammonia: Will turn damp red litmus blue Chlorine: Will turn damp blue litmus red and then bleach it white
- Slides: 14